Small semiconductors are used to detect cancer, to image the lymphatic system and to modify microscopy.
Michael A. Greenwood, News Editor
Visualizing an object can go a long way toward understanding how it works.
For growing numbers of researchers, quantum dots are becoming the tool of choice to provide the necessary light. The semiconductor nanocrystals increasingly are being used as tiny flashlights to illuminate intricate biological phenomena. The images, captured with highly sensitive cameras, are generating fresh ideas and opportunities for improved treatment and prevention of disease.
And quantum dots, which offer a host of unique optical properties compared with traditional dyes, likely will become even more commonplace in research laboratories in coming years as they are further modified and improved. The nanocrystals — usually ranging from 2 to 10 nm in size and fabricated from a host of semiconductor materials — already are tunable and have high photostability. They have broad absorption spectra but narrow emission. Colors can be excited simultaneously with a single light source, and the surfaces of the nanocrystals can be conjugated with a variety of biomolecules to perform very specific functions.
For example, a single molecule within a cell can be tagged with a blue quantum dot and tracked over long periods. Another molecule within the same cell can be tagged with another color and its peregrinations compared and contrasted with the original.
But the semiconductors are not with-out flaws. Many quantum dots contain heavy metals that could cause health problems in humans with repeated exposure. Although it is common to perform in vivo experiments with laboratory animals using quantum dots, such experiments using humans are unlikely anytime in the near future because the long-term health consequences remain in question.
Researchers, meanwhile, continue to search for less toxic or nontoxic solutions. If they are found, the appeal of quantum dots as imaging agents likely will increase even more.
Illuminating lymph nodes
Imaging the inner workings of the lymphatic system has never been clear-cut. Its many channels are tiny and not readily accessible. Tracking drainage from two or more lymph nodes becomes hopelessly confusing because it is all but impossible to tell which fluid came from where.
Researchers from the National Cancer Institute in Bethesda, Md., and from the University of Tokyo believe that quantum dots could be the key to better understanding the workings of an essential part of the body’s immune system.
The nanosize semiconductors are small enough that they can be inserted into the lymphatic system and versatile enough that their emission spectra can be tailored with near-pinpoint accuracy. Current imaging methods for the lymphatic system include those using radiolabeled colloids, which provide poor resolution and lack multiple colors and which expose both the patient and the surgeon to radioactivity.
The team used the nanocrystals to achieve simultaneous in vivo multicolor images of five different lymphatic basins in the anterior regions of mice. Lead researcher Dr. Hisataka Kobayashi of the National Cancer Institute said that imaging in vivo with five colors rarely has been performed.
Kobayashi said that quantum dots have the potential to help researchers better understand how the lymphatic system works and how cancer metastasizes into the lymph nodes. The technique potentially also could be applied to other in vivo studies, such as monitoring cellular trafficking.
“I believe that the possibilities of this method are great. This method could replace most of the current procedures for detecting cancer in sentinel lymph nodes,” he said.
Researchers used carboxyl cadmium-selenium quantum dots with peak emissions of 565 (bluish), 605 (green) and 655 nm (yellow) as well as cadmium-tellurium quantum dots emitting at 705 (magenta) and 800 nm (red). The hydrodynamic diameter of the nanocrystals — between 15 and 19 nm — was determined with a dynamic light-scattering device from UK-based Malvern Instruments and with a size-exclusion high-performance liquid chromatography system from Beckman Coulter Inc. of Fullerton, Calif. The optimal particle size for the lymphatic system is between 10 and 20 nm.
The test mice were injected with quantum dots intracutaneously in five areas — the upper extremities, the ears and the chin — to monitor drainage in the neck and in the upper trunk, sites of the most complicated lymphatic networks (Figure 1). Each of the injection sites was masked by nonfluorescent tape so that the signal emanating from it would not overwhelm the camera.
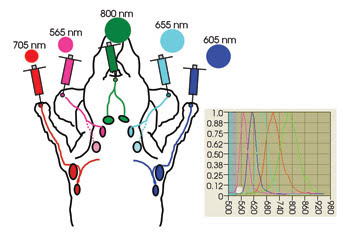
Figure 1. Quantum dots were injected into five sites in the upper body of test mice. The different colors of the quantum dots allowed researchers to image five lymphatic basins simultaneously. The graph shows the emission spectra of each quantum dot used in the experiment. Reprinted with permission of Nano Letters.
Kobayashi said that, within five minutes of injection, spectral imaging was performed with an in vivo system from CRI Inc. of Woburn, Mass. The mice were placed in a spinal position and put under anesthesia. The system is designed for multicolor whole-animal imaging. The team visualized five distinct drainages to the primary lymph nodes. All the draining lymph nodes were validated by taking ex vivo fluorescence images of the lymph nodes (Figure 2). The research results were reported in the June issue of Nano Letters.
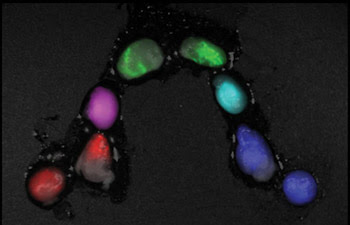
Figure 2. Spectral fluorescence imaging of lymph nodes was performed after they were surgically removed from the test subject. Reprinted with permission of Nano Letters.
Separating five drainage patterns by color could provide researchers and surgeons with a quicker and vastly improved picture of what is happening inside a complex and vital network, whereas imaging the lymphatic system with a single color allows little distinction of the traffic patterns or of the interplay between the system’s various parts. Taking one image at a time and merging the results is time-consuming and laborious.
One problem encountered by investigators was that the 565-nm quantum dots exhibited poor brightness and penetration in comparison with the others. They overcame this by using a double dose of the 565-nm variety. In the future, substituting a nanocrystal that emits at 750 nm might be preferable, they said.
The potential toxicity of the quantum dots, meanwhile, remains a concern. Those used in the research contain heavy metals encapsulated in a nontoxic coating. Even though the concentrations are infinitesimal, the long-term health effects from exposure must be determined before the technique can be extended to humans. “This is still an unanswered question,” Kobayashi said.
Indeed, the human lymphatic system is many times more intricate than that of a mouse, so many more quantum dots, and possibly even more colors, would be needed to acquire precise readings. If the quantum dots are determined to be unsuitable for use in humans, other sources, such as organic dyes, could be used. Dyes, however, have a wider bandwidth and are less suitable for the research conducted by the group.
Despite these issues, Kobayashi said that the procedure is promising enough to go to clinical trial. Moreover, the upper limit probably is not five simultaneous images: Technically, it is possible to image 10, and possibly more, sites. He predicted that it is only a matter of time before that is accomplished.
Evolution in imaging
The ever-growing world of microscopy may be getting a little larger.
Quantum dots have enabled researchers in the Netherlands to combine two common microscopy techniques long believed to be incompatible with one another. By employing the tiny semiconductor nanoparticles — instead of traditional fluorescent labels — the researchers created a hybrid form of fluorescence and Raman microscopy that can be performed on a single optical setup. The method could become another tool in a researcher’s arsenal, allowing versatility while gathering more information on single cells in less time.
The technique consists of fluorescence microscopy scanning (revealing quantum dots targeted to specific intracellular sites) and then focusing on particular regions of interest within the cell and performing chemical analysis with Raman microspectroscopy. Correlating the fluorescence emission with the Raman signal provides information that is difficult or impossible to obtain using each method individually.
The hybrid technique works because the fluorescence of the quantum dots is spectrally separated from the Raman signals, so it does not overwhelm the much weaker cellular vibrations that are the signature of Raman readings. Simply put, the fluorescence signals are far enough apart on the spectrum that they do not compromise Raman microscopy on the same cell.
“Our goal is to use fluorescent molecules as flashlights,” said researcher Henk-Jan van Manen of the Biophysical Engineering Group at the University of Twente. “We can then zoom in on those interesting regions and perform detailed biochemical analysis.”
Van Manen and lead researcher Cees Otto demonstrated two applications of the technique. In the first, the team used light with a wavelength of 413.1 nm to excite quantum dots in white blood cells. The resonant Raman signal from the cells was between 420 and 445 nm, whereas the one-photon excitation fluorescence emission was detected between 590 and 650 nm. Both signals could be visualized separately (Figure 3).
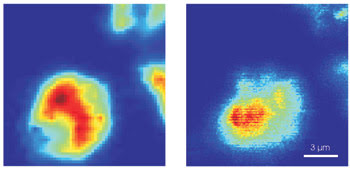
Figure 3. Shown are the combined results of resonant Raman (left) and one-photon excitation (right) microscopy using 605-nm quantum dots excited by UV light with a wavelength of 413.1 nm. Reprinted with permission of Nano Letters.
In the second application, single-cell nonresonant Raman and continuous-wave two-photon excitation fluorescence microscopy were performed together. A 647.1-nm excitation wavelength was applied to visualize cellular proteins and lipids in cells incubated with quantum dots. The Raman signal ranged from 660 to 730 nm. The fluorescence of quantum dots in endocytic vesicles was detected below 640 nm. As with the previous application, both signals could be visualized separately (Figure 4). Details of the research were published in the June issue of Nano Letters.
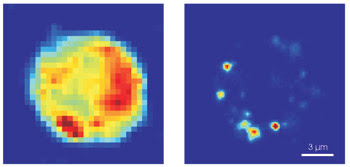
Figure 4. The combined results of nonresonant Raman (left) and two-photon excitation (right) microscopy using 585-nm quantum dots excited with a 647.1-nm wavelength are shown. Reprinted with permission of Nano Letters.
The researchers said that both techniques should be applicable to any cell that can be labeled with a quantum dot. Other excitation wavelengths, including 488 and 514 nm, also might be used to excite quantum dots and to perform Raman measurements.
Experiments were performed by point-scanning the excitation beam over the sample. A foldable mirror was placed in the optical detection path to switch between Raman and fluorescence readings. A PerkinElmer avalanche photodiode point detector was used to detect fluorescence, while a Horiba Jobin Yvon spectrograph, coupled with a CCD camera from Princeton Instruments, was used for the Raman signals. A Coherent krypton ion laser provided the excitation light. Much of the team’s instrumentation for the Raman-fluorescence microscopy was home-built and is not commercially available. However, it would not be difficult to integrate a Raman system with additional detection parts for fluorescence microscopy.
During experiments, the researchers noticed that the quantum dots were prone to photobleaching at 413-nm excitation. After they performed resonant Raman imaging, the fluorescence of the quantum dots disappeared. They overcame this by keeping an eye on the illumination dose and by performing fluorescence microscopy first.
Van Manen said that, now that the hybrid technique has been proved to work, the group plans to test it on more complex biological phenomena, including phagocytosis and stem cell differentiation.
Revealing cancer
A diagnosis of pancreatic cancer is almost always bad news. In many cases, the disease is fatal. One of the few hopes is to catch it in the earliest stages, when treatment options still have a chance of success, said Paras N. Prasad, the executive director of the Institute for Lasers, Photonics and Biophotonics at the University at Buffalo in New York. He believes that quantum dots can provide the critical warning signal, even when the disease is in its nascent stages.
Prasad and a team of investigators, including others from the institute, from Johns Hopkins University School of Medicine in Baltimore and from Zhejiang University in Hangzhou, China, found that quantum dots have a host of exceptional properties — spectral stability, tunability, a flexible surface chemistry — that allow them to be conjugated with a wide variety of task-specific biomolecules. Quantum dots also can be tuned to emit in the near-IR region, where light penetrates tissue more effectively, thus providing a better picture of whatever is taking place.
During recent experiments, the team took advantage of these properties to create conjugated quantum dots capable of targeting and illuminating human pancreatic cancer cells in vitro.
Although pancreatic cancer can be imaged with a variety of other techniques — including abdominal CT and MRI or endoscopic ultrasound — often it proves to be too little too late. The disease often has quite progressed by the time these techniques are used.
“Our final aim is to engineer bioconjugated quantum dots that will recognize the signatures of pancreatic cancer at its incipient stage,” Prasad said. The team’s research was outlined in the June 28 issue of The Journal of Physical Chemistry B Letters.
The investigators worked with quantum dots made of cadmium selenide, cadmium sulfide and zinc sulfide, a combination that offers particularly good photoluminescence efficiency and stability. These quantum dots were then conjugated with either transferrin or anti-Claudin-4 as targeting ligands. The quantum dots used in the experiments emitted at 624 nm.
Three human pancreatic cancer cell lines (Panc-1, MiaPaCa-2 and CoLo-357) were incubated with the transferrin-conjugated quantum dots. Unconjugated quantum dots also were incubated as a control. After one hour, the cancer cells were washed and imaged with a laser scanning confocal module attached to a Nikon upright microscope. This setup allowed for single- and two-photon imaging and for spectrally resolved or lifetime imaging.
The researchers found that quantum dots coated with transferrin successfully located and robustly illuminated the cancerous cells (Figure 5). Cells exposed to unconjugated quantum dots remained dark. Similar results were observed for all three cancer cell lines.
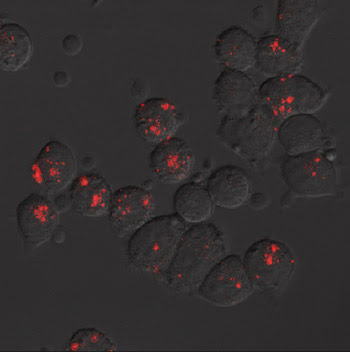
Figure 5. Pancreatic cancer cells treated with antibody-conjugated quantum dots emitting reddish fluorescence are shown. Researchers used transferrin and anti-Claudin-4 as ligands.
Besides transferrin, the team tested the technique with quantum dots conjugated with anti-Claudin-4, a monoclonal antibody that is highly specific for pancreatic cancer. The results were similar to those with transferrin. Researchers found a strong uptake of the conjugated quantum dots in the cancer cells. Conversely, cancer cells exposed to the control quantum dots exhibited virtually no uptake and, hence, no fluorescence.
One issue still to be worked out is the safety of the quantum dots. Many that have desirable photoluminescence, including those used by Prasad’s team, potentially contain less-desirable heavy metals. An option is to develop relatively nontoxic quantum dots made from indium phosphide and silicon. Safety concerns must be resolved before studies can be extended to humans.
Still, Prasad sees the preliminary findings as a foundation for further work with conjugated quantum dots as imaging agents for pancreatic cancer. Future work will focus on the in vivo detection of primary and metastasized tumors in mice. Prasad said that quantum dots that emit deeper into the NIR region will be used to enhance illumination.