
Delivering drugs with light
Technique enables precise control
David L. Shenkenberg
Although numerous drug delivery devices have been developed, few offer the precise control that light can provide. The dose and the time and location of release can be controlled by adjusting the properties and location of emitted light, as researchers from Queen’s University Belfast in the UK have demonstrated.
Colin P. McCoy, the leader of the team, said that the group is interested in applying light-mediated drug delivery to treating infection in medical devices such as catheters. Such a technique in principle could be used to deliver any drug anywhere light has access, such as to stents and catheters as well as to external areas such as the skin or eyes.

Figure 1. Researchers attached a light-activated compound to drugs, resulting in this solution (left). Once 365-nm light was focused on the solution, it changed color (right), corresponding to the separation of the drug from the light-activated compound. Courtesy of Colin P. McCoy.
He explained also that traditional oral tablets, for example, are therapeutically active only within a small concentration range. Above this range, they may be toxic; below this range, they may produce side effects. “The level of control that we offer is potentially down to the level of a single molecule.”
To enable light-controlled drug delivery, the researchers used organic chemical synthesis to attach a light-activated compound to the drugs. This compound, 3,5-dimethoxybenzoin, has been used extensively for synthesis but not for drug delivery. It dissociates from other compounds when hit with 365-nm (UV) light. The researchers joined 3,5-dimethoxybenzoin to three model drugs: acetyl salicylic acid (aspirin), ibuprofen and ketoprofen.
In the first experiment, the scientists determined whether the photoreaction would liberate most of the drug molecules from the light-activated compound and whether any additional chemicals were produced in the photoreaction. They wanted most of the drug molecules to be released, and they did not want to produce additional chemicals, McCoy said. They employed a 15-W mercury discharge lamp for UV excitation, and they measured the reaction using various methods. When exposed to the light source, the light-activated compound dissociated from the drugs with a high yield and no side products, as desired.
Molecular-scale control
In the second experiment, the researchers investigated the extent to which light could control the drug dose, the amount of drug released. To do so, they placed the solution containing the drugs and light-activated compound in the dark, and they exposed it to bursts of light followed by darkness, using the same UV light source as before.
They found that the reaction started when the first burst of light occurred, stopped completely in the dark and continued with subsequent bursts, resulting in a step-shaped graph (Figure 2). The dose of the drug correlated with the duration of light exposure, and the researchers achieved molecular-scale control of dosing.
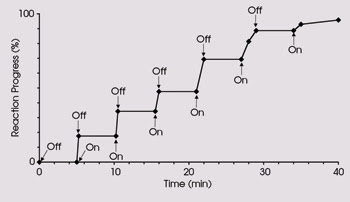
Figure 2. This step-shaped graph shows that light precisely controlled the amount of drug released. When the light source was turned off, the drug no longer was released, and drug release resumed when the light source was turned on again. Reprinted with permission of the Journal of the American Chemical Society.
In the final experiment, they dissolved the drug and light-activated compound in a polymer. McCoy said that catheters and stents are made of polymers; thus, this experiment was a test to see how activation of the drug would proceed in a real medical device. The researchers excited the drug with the UV light source and measured the reaction with a PerkinElmer fluorimeter because the reaction product left in the polymer was fluorescent. Importantly,
McCoy said that the polymer absorbed water and became soft and flexible like a contact lens, allowing the drug to diffuse out of the polymer. The reaction proceeded efficiently. A side product was produced in this medium, but it was retained within the polymer. The researchers reported these findings in the Aug. 8 issue of the Journal of the American Chemical Society.
In the future, they would like to use longer wavelengths because UV is harmful to tissue and because longer wavelengths enable greater penetration depths, McCoy said. They plan to use the technique in a working medical device and with drugs that are clinically useful.
Published: September 2007