
Virus tracking without labeling
Technique could be useful for following particles over long periods
Hank Hogan
Researchers at the Swiss university ETH Zurich have shown that, with the right light and equipment, it is possible to image and track virus particles, or virions, without fluorescently labeling them first. Because the virions aren’t labeled, they don’t photobleach into invisibility, no matter how long the exposure, which makes it possible to track and study single particles for extended periods, perhaps even days. What’s more, the equipment required to implement the technique is fairly standard.
At its heart, the new method uses interferometry. Vahid Sandoghdar, head of the nano-optics group at the university, recalled that the approach evolved from work done several years ago involving detection and spectroscopy of gold nanoparticles. The contrast mechanisms of the gold nanoparticles, the researchers realized, were very simple and held true for other nanoparticles. Differences in particle polarizability accounted for small differences in signal.
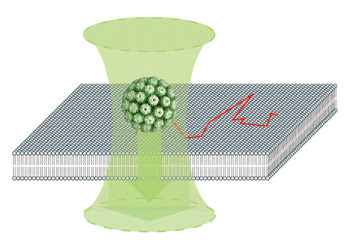
Using an interferometric optical detection scheme (represented by the two arrows), researchers tracked the movement of unlabeled single simian virus virions, shown here as a sphere, as they moved across a supported membrane bilayer. The track is represented by the red line. Because the virions are not labeled, they can be tracked for long periods. Images courtesy of Vahid Sandoghdar, ETH Zurich.
In their detection scheme, a laser beam strikes and is scattered by a nanoparticle and reflected by the substrate. The interference of the scattered and reflected beams produces a signal that pinpoints the location of the particle. A 45-nm-diameter virus — which is about 85 percent protein and has a refractive index of about 1.5 — in an aqueous medium produces a signal contrast of about 2.6 percent.
The technique is fairly simple. “Anyone with a confocal microscope could do it. The only thing you have to worry about is the intensity noise on your laser,” Sandoghdar said. The low signal contrast drives the need for a relatively noise-free laser. Although most have a 1 to 2 percent intensity noise, which is too large, he noted that there are inexpensive lasers that have noise an order of magnitude lower.
The simplicity of the method may have contributed to its not having been demonstrated previously. “I believe the reason no one had done it before is that one didn’t expect such a simple thing to work so well,” he said.
As detailed in the August issue of Nano Letters, the researchers implemented the method using a custom-built microscope with a diode-pumped 532-nm Nd:YAG laser as a light source, galvanometric mirrors to steer the beam, assorted optics and a Hamamatsu photodiode for detection. To demonstrate the technique, they used simian virus 40, a tumor virus that is 45 nm in diameter and whose human counterparts are associated with cancer and other diseases. The structure of the virion is known, and its cellular receptor has been identified as GM1. They put a dilute solution of these virions on a clean cover glass on the stage and imaged the glass-water interface from below using an oil-immersion objective from Zeiss.
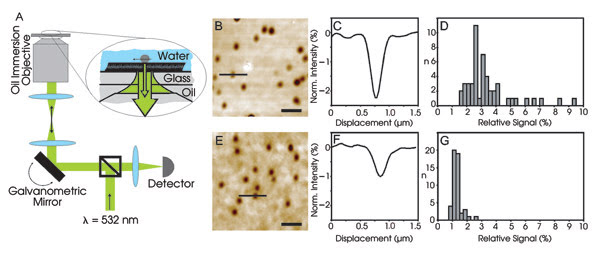
In the setup on the left, a 532-nm beam strikes a galvanometric scanning mirror and is focused onto an aqueous solution containing virus particles. The reflected and scattered light interferes, giving rise to the images in B and E for a virus and viruslike particle, respectively. The data in C and D show that each spot is a single virus particle, while F and G are for viruslike particles, again with one per spot.
Using the mirrors, they scanned the 532-nm beam across the field of view, with the reflected light captured by the optics, passed through a beamsplitter and focused onto the photodiode. Within minutes after adding the virus solution, spots appeared in the scanning image, and the researchers showed that each contained a single virion.
The average relative contrast was 3.00 ±0.87 percent, about what was calculated for a virus of this size and makeup. As further proof, they also fluorescently labeled virions and imaged them using a Zeiss confocal laser scanning microscope. The interferometric and fluorescent images overlapped completely.
Comparison studies
Using the same setup, they imaged viruslike particles in solution. These particles were not intact virions and lacked the dense DNA-histone core of the complete viruses. Their relative contrast averaged only 1.28 ±0.25 percent. The researchers attributed the lower signal — as compared with the virions — to the structure of the viruslike particles. By way of comparison, they noted that a 20-nm gold nanoparticle would yield relative contrast numbers similar to those of the virions and viruslike particles under the same conditions.
The researchers also demonstrated their detection technique using supported membrane bilayers containing GM1 and choleratoxin, which uses GM1 as a receptor, bound to 20-nm colloidal gold nanoparticles. When imaged, the bare membranes essentially showed nothing. When the choleratoxin-gold was added to the solution, spots with an average relative contrast of 2.61 ±0.65 percent appeared. However, when gold nanoparticles functionalized with another biomolecule — one that doesn’t have the GM1 receptor — were added, no spots appeared.
Finally, they tracked a single virion as it moved in the supported membrane, capturing its location at fixed intervals via consecutive images. From the data gained by tracing random movement, they calculated the diffusion constant of individual virions. This figure of about 0.0088 square microns per second was in agreement with previous values they had obtained in studies tracking single fluorescent viruses.
The technique has its limitations. It won’t work, for example, in strongly scattering media such as living cells, and it needs a light source that meets the required noise intensity. But because it allows the tracking of particles for long periods of time, it also has advantages. One of these is the collection of data on a single particle, as opposed to an approach in which many particles are imaged and a statistical average of some parameter is extracted. Another is an investigation of the effect of changing biochemistry through the alteration of some condition, with tracking of a single virion before and after the change providing clues as to the consequences.
Finally, according to Sandoghdar, the technique can be used in conjunction with other methods. “Another powerful feature is that we could combine this mechanism with single-molecule fluorescence detection.”
The researchers, for example, are using this approach to simultaneously detect single virions and the single dyes attached to them in a study of three-dimensional motion of receptor-bound virions.
Sandoghdar noted that this work is well under way but that not enough data has yet been collected to comment on any results.
Published: September 2007