
Easy, visual detection with multicolor SPR imaging
Spatial resolution and setup simplicity may make the technique useful for handheld SPR sensors
Gary Boas
Surface plasmon resonance (SPR) sensing offers label-free detection for a wide range of applications —immunosensing, proteomics and drug discovery, detection of DNA hybridization as well as other biologically relevant processes — and for this reason has gained currency in both research and industry.
Most SPR methods use single-color detection, which takes advantage of measurements either of changes in the intensity of reflected light recorded at a fixed angle of incidence or of reflectivity as a function of incident angle. Investigators also have reported use of two-color SPR sensing to measure simultaneously the refractive index and thickness of adsorbed films and, thus, to determine the optical thickness of the film.

Researchers have developed a multicolor SPR imaging method that enables easy, visual detection of biomolecular binding events basedon changes in color, using wavelength and the angle of incidence to determine the thickness of films on a microarray. Shown here are SPR images of a protein microarray using 632-nm light at an angle of incidence of 43.8° (left), white light at 43.8° (middle) and white light at 45° (right).
In the July 15 issue of Analytical Chemistry, Bipin K. Singh and Andrew C. Hillier of Iowa State University of Science and Technology in Ames reported multicolor SPR imaging of protein microarrays. Coupling of surface plasmons leads to absorption of specific wavelengths of incident white light, producing distinct reflected colors. The formation of thin molecular films on micro-arrays yields various colors — depending on their thicknesses — thus allowing easy, visual detection of biomolecular binding events.
“We wanted to develop a simplified SPR readout method,” said Singh, now of Intel Corp. in Hillsboro, Ore. “We have shown that one can differentiate between films of different thicknesses by just looking at the surface and not having to analyze the complete spectral response.”
Creating an array with traditional patterning methods proved challenging, however. The researchers therefore chose to use ink-jet printing, which delivers precise amounts of protein at the surface. “The ability to have precise control over the volume of the droplets was a significant advantage,” Singh said. They used a chemical ink-jet printer, the printing nozzle of which comprised a glass capillary fused to a piezoelectric crystal. Applying a voltage pulse to the crystal caused the nozzle to dispense a droplet of ink — in this case, with a diameter of 48 ±2 μm.
They imaged the surface plasmons’ resonance response to both wavelength and angle of incidence using a custom-built optical system with a tungsten-halogen lamp made by Ocean Optics Inc. of Dunedin, Fla., as a 360- to 2500-nm white light source. Light from the source illuminated a hemicylindrical prism coupler with a focal length of 25 mm; a microarray on a gold-coated glass slide was kept in optical contact with the flat side of the prism coupler. The reflected light was sent through a transmission diffraction grating made by Edmund Optics of Barrington, N.J. A digital camera with a high-sensitivity and high-resolution CMOS sensor, made by Canon Inc. of Tokyo, captured the first-order dispersion after it passed through the grating.
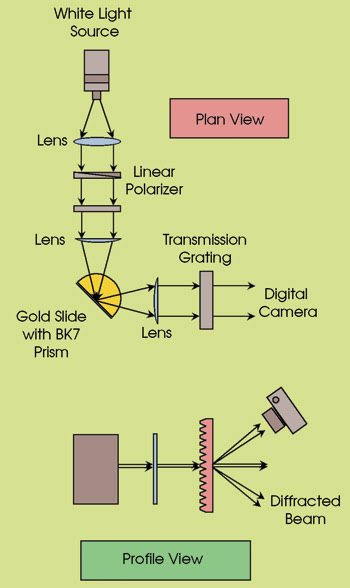
They used a custom-built system to characterize the SPR response of glass slides coated with gold films. With the system, the reflected light is sent through a transmission diffraction grating, after which a digital camera captures the first-order dispersion.
The researchers demonstrated the potential of multicolor SPR imaging and determined its efficacy using a microarray made of bovine serum albumin. They used another custom-built system to collect the SPR images.
With this system, the prism was placed in optical contact with the SPR sensor chip and the back side of the sample, illuminated by a collimated beam of white light. A 10x objective made by Mitutoyo Corp. of Kawasaki, Japan, magnified the reflected image, which was then captured by the same Canon digital camera. After printing the microarray on the gold-coated slide, they imaged it at 43.8° and 45°. At 43.8°, they performed the experiments with and without a 632-nm bandpass filter so that they could compare the color SPR method with monochromatic SPR imaging, which was made possible by using 632-nm light.
In the images acquired with the filter, the bare gold regions of the slide appeared as a dark background because the incident light was totally absorbed when exciting the surface plasmons. The areas of the slide with deposited protein showed an increase in the reflected light intensity.
When they removed the 632-nm filter, thus illuminating the slide with white light, the microarray appeared as yellow spots on a green background. Because the color of the spot depends on the angle of incidence, film-covered regions of different thicknesses will appear in the image as different colors. They acquired another multicolor SPR image, with an angle of incidence of 45°, and observed green spots with a yellow rim — confirming the color change with angle of incidence.
Singh noted that the overall sensitivity of the technique is lower than that available with monochromatic SPR imaging, but that the spatial resolution and detection limits that it offers — besides the easy readout and fewer optical components required — recommend it for applications in SPR microscopy and in portable, handheld SPR sensors. Development of inexpensive “dipstick”-type sensors is another possibility, especially because the color changes are much easier for users to detect than the more subtle changes in light intensity found with monochromatic SPR imaging.
The technique also could help to determine simultaneously the refractive index and thickness of adsorbed films, much like the previously reported two-color SPR methods. Multicolor SPR imaging would offer more flexibility than two-color methods, though, because any wavelength of light in the red and green regions of the spectrum can be coupled to surface plasmons.
Published: September 2007