
It’s Two, Two, Two Tools in One
Technique enables simultaneous imaging and laser surgery without mechanical scanning.
If a surgeon is about to use a probe in the brain, the patient is sure to want that probe to be as small as possible. And three University of California, Los Angeles (UCLA), researchers have developed technology that could lead to tinier probes that can perform both endoscopic imaging and laser microsurgery at the same time.
The technique, called “spectrally encoded confocal microscopy and microsurgery” – SECOMM for short – uses light for both functions. And it’s the first of its kind that does not require mechanical scanning for either.
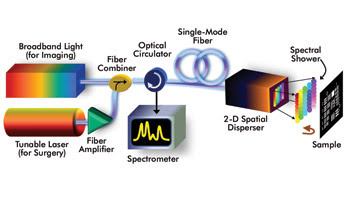
With the SECOMM method, a fiber combiner takes a broadband light source (for imaging) and combines it with a wavelength-tunable continuous-wave laser followed by a fiber amplifier (for laser ablation). The 2-D spatial disperser generates a “spectral shower,” and the spatial information about the sample is encoded into the spectrum of the back-reflected spectral shower. The optical circulator routes the back-reflected spectral shower to the spectrometer. Images courtesy of Kevin Kin-Man Tsia.
Mechanical scanning is useful because it enables acquisition of multidimensional images. But it has its downsides in endoscopy.
“Fluctuations in mechanical scanning introduce image noise and artifacts,” said recent PhD graduate Kevin Kin-Man Tsia, now an assistant professor in the department of electrical and electronic engineering at the University of Hong Kong. He added that endoscopic probes using microelectromechanical systems (MEMS) scanners are similarly limited. And MEMS scanners in miniaturized endoscopic probes require a large probe size, up to ~1 cm, which limits their usefulness in clinical settings.

UCLA researchers Dr. Kevin Kin-Man Tsia, left, professor Bahram Jalali, center, and Dr. Keisuke Goda have developed an endoscope-compatible single-fiber-based device capable of simultaneous imaging and high-precision laser microsurgery.
But SECOMM doesn’t have that problem.
How it works
“The heart of SECOMM,” Tsia said, “is an optical diffractive component: a two-dimensional spatial disperser which diffracts the different wavelengths of incident light into 2-D space, creating a 1:1 map between 2-D spatial coordinates and the optical wavelengths.”
He said the 2-D spatial disperser delivers broadband light for imaging and wavelength-tunable light for laser surgery. Two optical diffractive elements make this happen: a virtually imaged phase array (VIPA) and a diffraction grating. The figure below, left illustrates the SECOMM design.
“Both the broadband light and high-power tunable laser are coupled into the same single fiber and the same 2-D spatial disperser to perform simultaneous imaging and high-precision laser microsurgery,” Tsia said.
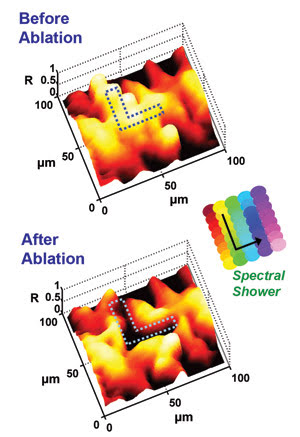
This demonstration of SECOMM’s ability to perform laser microsurgery and simultaneous monitoring shows the images captured (a) before and (b) after performing laser ablation on a bovine tissue sample. The “L” pattern (outlined in dots) is carved out of the tissue by tuning the wavelength of the CW laser in the manner shown in the inset of the figure (see arrow). The Z-axis represents the normalized reflectivity of the sample.
The disperser transforms an incident broadband light beam from a supercontinuum pulse laser or an incoherent broadband light source into a 2-D spatial spectral pattern resembling a spectral shower, which is used to illuminate the sample.
The sample’s 2-D spatial information is encoded into the back-reflected “spectral shower,” which is transmitted back to the “nondispersed” – but image-encoded – beam by the same 2-D spatial disperser.
The single-mode fiber recollects the beam, allowing transmission of 2-D images of the sample, and a spectrometer detects the image-encoded spectrum.
“Such imaging is essentially a confocal microscope,” Tsia noted, “as the aperture of the fiber that captures the reflection from the sample rejects the scattered light from out-of-focus axial planes.”
Folding the 1-D spectral data into a 2-D matrix that represents the image allows for digital reconstruction of the sample’s image.
Tsia said that a high-power wavelength-tunable laser beam will follow the same wavelength-to-spatial-coordinate mapping as the spectral shower performing the imaging if it is coupled with the imaging optics via a beam combiner and passes through the same spatial disperser.
“By tuning the wavelength of the laser,” he said, “the beam can be directed to any arbitrary position on the sample to perform laser surgery [ablation], without any mechanical movement of the probe to steer the laser beam or the movement of the sample.
“Hence, high-precision microsurgery can be performed by computer-controlled tuning of the laser wavelength according to a preprogrammed pattern.”
Advantages and challenges
Most currently employed endoscopes rely on a CCD or a fiber bundle to capture images, sometimes combined with a scalpel or other surgical instrument to provide simultaneous imaging and surgery.
For minimally invasive procedures, an endoscope must be very flexible, and it must have a very small diameter. “In CCD-based probes, the size of the chip placed at the distal tip limits the minimum diameter to about a few millimeters,” Tsia noted, “and their electrical cables limit their flexibility.” He added that fiber-bundle technology is limited because obtaining a high pixel count requires a large number of fibers, resulting in mechanical rigidity. Because SECOMM uses a single fiber, it is both small and flexible.
Right now, SECOMM is hindered by its spatial resolution – around 4 to 10 μm – and by the number of pixels it can capture.
“Nevertheless,” Tsia said, “these are not the inherent limitations of this technique because the number of pixels can significantly be increased by using an optical source with larger bandwidth or a spectrometer with higher spectral resolution.”
First of its kind
Others have been working on designing similar probes in recent years. A Harvard Medical School group demonstrated a 1-D spectrally encoded endoscope, but the technique requires mechanical scanning to capture a whole 2-D image. Another group demonstrated an endoscopic probe capable of both imaging and laser surgery through the use of MEMS scanners for beam steering. “SECOMM has the advantages over such endoscopic probes,” Tsia said, “as it eliminates the need for mechanical scanning.”
Grating-VIPA arrangements have been used in the past for demultiplexing in telecom applications and for spectroscopy, he added. But his team used the 2-D spatial disperser for imaging and microsurgery.
A winning team
Tsia worked on the SECOMM project with Keisuke Goda, a postdoctoral researcher in the electrical engineering department at UCLA, and with Bahram Jalali, a professor of electrical engineering at the university.
These same three researchers recently made headlines when they developed “the fastest camera in the world.” That camera is known as STEAM, which stands for “serial time-encoded amplified microscopy.” It allows real-time imaging with up to 6 million fps, thanks to ultrashort laser pulses; “optical” image gain enables high detection sensitivity. STEAM, according to Tsia, can overcome the trade-off between speed and sensitivity that occurs in existing CCD/CMOS cameras. And its speed makes it useful for capturing rapid biological processes and events such as the firing of a neuron. It also can be used with SECOMM for laser surgery.
In fact, Tsia said, SECOMM originated from the work they did on STEAM.
“One of the key features in STEAM is spectrally encoded imaging, which maps the spatial information onto the spectrum of an ultrashort laser pulse. We borrowed this idea for SECOMM and realized that laser surgery can also benefit from the same wavelength-space mapping idea.”
The two can be combined to perform simultaneous ultrafast real-time imaging and laser surgery. “This is particularly useful to monitoring the laser ablation dynamic in the tissue,” Tsia said. “The optical amplification feature in STEAM can also be applied to SECOMM, in which the detection sensitivity can be greatly enhanced.”
Concept proved
The next step for the SECOMM project is to design and build the miniaturized SECOMM probe. This will consist of off-the-shelf miniaturized optics, including a diffraction grating and a gradient-index lens.
The preliminary design showed that SECOMM can be miniaturized into a submillimeter-diameter probe with spatial resolution of 1.4 × 2 μm, with a field of view of 280 × 70 mm. “The present imaging technique can also be further extended to three-dimensional volumetric imaging by employing an interferometric configuration, which enables the acquisition of the depth information of the sample,” Tsia said, which will be good news for future patients undergoing delicate procedures.
“SECOMM can be applicable to any area where high-precision, small and flexible probes are required, such as brain tumor, pediatric and endovascular surgeries,” he said.
Published: September 2009