
Molecular probe may signal cure for Alzheimer’s
Understanding Alzheimer’s, a neurodegenerative disease that robs its sufferers of their personalities and normal brain functioning, has eluded the scientific community. What is known is that the condition involves the misfolding of a protein called amyloid beta, which originates from the amyloid precursor protein during its formation. This results in deposits of incomplete proteins, called amyloid plaques, outside the neurons of the central nervous system. Later, hyperphosphorylated tau proteins form tangles inside the neurons.
However, treatment for Alzheimer’s should be a little nearer, thanks to a new molecular probe that can do what previous “detective” molecules could not: cross the blood-brain barrier, the physiological mechanism that protects the brain from harmful substances. Called an LCO, or luminescent conjugated oligothiophene, this molecule will allow scientists to see amyloid plaques in the brains of live mice through a cranial window, enabling treatment to be followed in real time. This is a crucial step toward investigating the disease in humans. The study was published online July 22, 2009, in the ACS journal Chemical Biology.
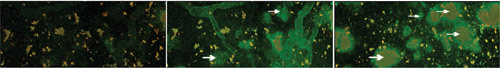
This multiphoton image, taken through a cranial window, shows LCO labeling of amyloid-beta deposits, which are characteristic of Alzheimer’s disease, in the brain of a transgenic mouse. Scientists anticipate that the development of LCO, a new molecular probe that can cross the blood-brain barrier, will lead to live imaging of Alzheimer’s in the human brain. (Left) Background shot; (middle) 12 minutes after injection of LCO; (Right) 100 minutes after injection. The white arrows indicate LCO-labeled amyloid plaque. Courtesy of Mikael Lindgren.
Physicist Mikael Lindgren of the Norwegian University of Science and Technology, one of the authors, describes the potential of the new probe: “Here we designed new oligomeric thiophenes that have superior performance compared to the previous polymeric ones. For instance, these smaller oligomers pass the blood-brain barrier and can be used for in vivo investigations in Alzheimer mouse models using a cranial window and multiphoton absorption.”
This advance will build on previous Alzheimer’s research carried out in Scandinavia and published in the Aug. 3, 2007, issue of Chemical Biology. It describes how Lindgren and others studied amyloid plaques at various stages by using a molecular probe called a luminescent conjugated polyelectrolyte (LCP) developed by Peter Nilsson, chemistry professor at the University of Linköping in Sweden.
The LCP, which has fluorescent properties, can selectively target and bond to amyloid proteins in such an inflexible configuration that the emission spectra of plaques at similar stages are consistent. The researchers’ goal is to create color maps that will not only allow early diagnosis of Alzheimer’s in humans but also monitor its therapy.
To understand the dynamics between LCP and its target proteins, the scientists analyzed the excitation and emission fluorescence spectra of four variations of LCP both alone and bound to amyloid plaque. To simulate the human brain, amyloid plaques of various stages and sizes were studied in vivo as well as in laboratory conditions (the former in recently sacrificed mice).
The proteins were classified into various morphologies, or shapes, including microplaque, compact-core with diffuse exterior, and diffuse deposit. Histological staining of microplaque, an early-stage formation that is very dense, fluoresced an intense orange, as did histological staining of a compact-core formation, suggesting a close relationship; both have the same LCP signal profile.
The majority of compact plaques with diffuse exterior had an orange periphery that fluoresced in the 639- to 661-nm spectral region and a yellow-green center that fluoresced most intensely in the 543- to 564-nm region. These color emissions suggest that the exterior is denser, while the center is more loosely structured. Spectroscopic analysis raised the question as to whether the proteins form from the center outward or vice versa.
The researchers anticipate answering these questions and others as they use the breakthrough LCO probe to study Alzheimer’s disease in live mice. Eventually, to study the disease in humans, researchers would like to create a magnetic probe so that the disease process can be tracked with magnetic resonance imaging.
Published: September 2009