For the greatest reward, you sometimes have to take the most risk. Even though balancing risk against reward can be difficult, the federal government does it all the time in funding basic life sciences research.
A look at several agencies reveals some of the challenges and potential of funding riskier, yet possibly more rewarding, projects. It also shows what may be an emerging new funding source.
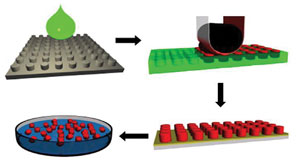
High-risk/high-reward funding has been awarded to help scale up the fabrication of nanoparticles for the life sciences in four easy steps. In the upper left, a proprietary material (green) and a silicon master template (gray) create a mold (green, upper right), which is filled with a drug formulation (red, upper right). These almost identical particles are then harvested (array, lower right; solution, lower left).
Aiming for the moon
The mission of the Bethesda, Md.-based National Institute of Biomedical Imaging and Bioengineering (NIBIB), part of the National Institutes of Health (NIH), is to accelerate the discovery and development of biomedical technologies that prevent or treat illness. In talking about high-risk, high-reward research, NIBIB Deputy Director Belinda Seto invoked two examples to describe what the institute is trying to accomplish. One is the US Department of Defense’s DARPA, which bankrolled the development of such originally far-out, but now commonplace, technologies as computer networking and micromachines.
A second example Seto cited comes from NASA. That is particularly apt because one of NIBIB’s aims is to create a medical moon shot. “You make this quantum leap in discovery, and the discovery is in a major health disease area,” she said in explaining the concept.
In 2007, NIBIB awarded four Quantum Grants, totaling $12 million and spanning three years. One of these involves a project at the University of Michigan in Ann Arbor to develop nanoparticles that will highlight brain cancer cells. The fluorescent nanoparticles will bind to the cells, allowing them to be seen and more effectively removed.
The same technology also could be used to photoinactivate cancer cells. Although aimed at brain cancer, the technique could be extended to other diseases.
Other Quantum Grants were for the development of insulin-producing islets at Wake Forest University in Winston-Salem, N.C.; for a miniaturized implantable renal assist device at the Cleveland Clinic Lerner College of Medicine in Ohio; and for a lung cancer-detecting point-of-care micro-fluidic device at Massachusetts General Hospital in Boston. The latter technology can separate specific circulating tumor cells in the blood at concentrations as low as one in a billion.
Seto noted that any research carries risk. Operationally, the NIBIB selects high-risk projects for funding by looking at those requests that do not get money through regular review procedures. The process includes a peer review, which results in scores that ultimately determine which projects get funded.
The NIBIB has adopted a policy to encourage high-risk research by re-examining – and possibly funding – those projects that don’t make the cut. “Each year, we will fund up to two high-risk research projects that are not above the payline,” Seto said.
She noted that this does not mean that this is the only high-risk research done by the NIBIB. It’s just that, under the existing criteria, only those that scored above the payline were going to get money anyway.
Finding the right balance
The NIH is the second-largest supporter of research and development in the federal government, surpassed only by the Defense Department. The FY 2009 NIH budget was $30.5 billion, with nearly all of that classified as R&D. The corresponding FY 2010 figure is almost $31 billion.
In 2009, a total of $348 million went out in the NIH Director’s Transformative R01, Pioneer and New Innovator Awards programs, all of which emphasize innovation and risk taking. In 2008, the figure for the latter two categories was $138 million. Both numbers are estimates of funds disbursed over a five-year period from the time the awards are announced.
Lana Skirboll, acting director of the program coordination, planning and strategic initiative division at NIH, explained that there is no formula for deciding the level of funding for high-risk/high-reward projects. She noted, however, that it is vital to fund some riskier proposals that could be fundamentally important but that lack preliminary data showing feasibility.
Although all proposals go through peer review by a panel of scientific experts, the riskier ones are evaluated differently. “High-risk/high-reward review panels are asked to weigh the potential impact of the research more heavily in their scores than feasibility,” Skirboll said.
She also noted that high-risk research that doesn’t succeed is not necessarily a failure. Sometimes finding out that something doesn’t work can be as important as finding out that it does.
Taking a risk on manufacturing
Not all high-risk research involves basic scientific breakthroughs. A case in point can be found in a December 2009 Technology Innovation Program (TIP) award from the National Institute of Standards and Technology (NIST) to Liquidia Technologies Inc. of Research Triangle Park, N.C. The startup company received $3 million over three years, in part because its technology might address a critical biomedical manufacturing need.

Applying a semiconductor-derived process, Liquidia Technologies Inc. can stamp out nanoparticles of various sizes and shapes (top, pollen; middle, posts; bottom, filaments) that can carry cancer drugs, inhalation products and other therapeutic agents. The company won a NIST award to help scale the technology for manufacturing. Images courtesy of Liquidia Technologies.
Joseph DeSimone, Liquidia’s co-founder and a professor of chemistry and chemical engineering at the University of North Carolina at Chapel Hill, noted that the company borrows techniques from the semiconductor industry to do something new.
“We have developed a roll-to-roll process that starts with semiconductor-grade master templates to make precision particles that have controlled size and shape and can be made out of any chemistry, including the very delicate molecules associated with biology,” DeSimone said.
The technology can produce nanoparticles that are completely absorbable yet can carry a drug within them. The technique has been used to make filaments that measure 80 nm in diameter and that are hundreds of nanometers long.
Liquidia plans to use these and other nanoparticles to deliver vaccines, cancer drugs, inhalation products and other therapeutic agents. Because the particles are all the same size and shape, the dosage and the timing of drug delivery can be controlled precisely. This also may mean that less of the therapeutic will be wasted and less needed overall.
Research projects carry a variety of risks. In the case of Liquidia’s project, the jeopardy resides in the scaling up of the manufacturing process.
A single 8-in. silicon wafer can produce enough vaccine-containing particles for hundreds of human doses. Testing a cancer drug requires 10,000 times that volume.
“We can get to clinical trials in vaccines with where we’re at today. But this technology could stop there if we can’t provide the scale-up required in a cost-effective way for these other products,” DeSimone said.
Successfully solving such problems, however, is important to future competitive manufacturing, which is why NIST funding makes sense, he added. He said one nice aspect of the NIST grant is that, unlike some other government programs, it is available to companies that have received venture capital funding, as is the case with Liquidia.
A new source?
The TIP grant to Liquidia is a new funding program, having made its first awards in 2008. The program has yet to launch a grant competition in life sciences research, said NIST spokesman Michael Baum. “The TIP competitions to date, focused on infrastructure and advanced materials in manufacturing, are not generally in that field.”
That may be changing, however, as shown by the Liquidia example. Another place where such a change can be found is in a NIST white paper open for comments as of early January 2010. It outlines a potential future area of funding titled “Advanced Technologies for Proteomics, Data Integration and Analysis, and Biomanufacturing for Personalized Medicine.”