Photodynamic therapy (PDT) is still a fresh-faced kid in the realm of medicine. Primarily used to destroy various cancerous tumors, it is being tested for use in wet macular degeneration, in acne and in other conditions. The technique has gained a lot of ground in Europe but is just beginning to win over doctors and hospital staff in the US and only slowly trickling into the public consciousness everywhere.
In PDT, photoreactive substances are brought into contact with tumor cells or other materials one wishes to remove. Once the light-sensitive molecules are given enough time to bind to their targets, light of a particular visible or near-infrared wavelength is shone on the target. As the energy of the illuminated photosensitizer is raised, it reacts with nearby oxygen to form singlet oxygen molecules. Singlet oxygen is very reactive and will proceed to destroy the target cells via necrosis or apoptosis.
PDT is mostly notable for its ability to use photosensitive dyes that selectively target tumors and other unwanted cells – leaving healthy cells alone. The technique was first developed several years ago, but ongoing research is focused on finding the most effective sensitizer/wavelength combinations for particular targets as well as proving the method’s overall clinical benefits.
Research also is under way in finding novel targets that will expand PDT’s use into new fields with new targets.
Not playing around with spores
Fungal infections don’t get a lot of respect from the public, who generally laugh off athlete’s foot and similar inflictions – as long as someone else gets them. Medical professionals, however, know better. Spores from fungal species such as yeast can enter the body through the mouth, wounds or any other available opening. Fungi then can spread into the lungs and blood, causing illnesses such as urinary tract infections. Fungal infections even can lead to death, especially among people with suppressed immune systems.
Antifungal drugs that act against the most prevalent fungal species, such as Candida spp., are common, but as when one battles any fungus, virus or bacterium, evolutionary forces get in the way, and the microorganisms adapt to become increasingly resistant to whatever tactic you try to use against them.
PDT is a relatively new weapon in the antifungal arsenal. A group of investigators representing Massachusetts General Hospital’s infectious diseases division and its Wellman Center for Photomedicine, both in Boston, is going after fungi such as Candida albicans and Cryptococcus neoformans with an all-out effort.
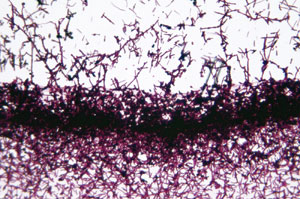
Researchers at Massachusetts General Hospital are using plant-derived saponins in tandem with photosensitizers to kill yeast cells.
Led by Eleftherios Mylonakis, the team has been testing the potential of the plant-derived substance saponin as a potential fungicide. Saponin, a bitter foaming agent that may keep insects and animals from eating the parent plant, is notable because it increases pore permeability in some cells, including those of fungi.
After identifying 12 types of saponin that exhibit various degrees of antifungal behavior, Mylonakis and his colleagues used them in conjunction with three types of photosensitizers: rose bengal, chlorin(e6) and a conjugate of polycationic chlorin(e6) and polyethyleneimine (PEI-ce6). For illumination, they used a LumaCare LC-122 noncoherent light source made by Ci-Tec UK Ltd., a division of MBG Technologies Inc., along with bandpass filters designed for 540 ±15 nm and 660 ±15 nm. The irradiance level was 100 mW/cm2.
The scientists found that the rose bengal and chlorin(e6) were not readily taken up by yeast samples but that, because of its polycationic charge, PEI-ce6 self-sufficiently penetrated the yeast cells. The addition of any of the saponins, however, increased the ability of both rose bengal and chlorin(e6) to permeate the yeast cell membranes, thereby increasing the destructive capacity of PDT.
Mylonakis and his team caution that it is still unclear whether C. albicans and other fungi can develop resistance to saponins, but they otherwise seem confident that the natural product holds great promise as a new class of antifungals.
Switching perceptions
Although much attention on photodynamic treatments focuses on drug activation, there are other ways in which photoswitching can come in handy. One such possibility is to use light to open and close the channels that regulate the flow of ions through live cells.
Controlling the stream of ions through a cell effectively gives you power over the operating state of the cell. If you block calcium ions for heart tissue, for example, you might be able to rein in heart damage during a myocardial infarction; control other ions, and you can choose which neurons fire in an afflicted brain.
Ion channels are gated by ligands – molecules that alter their shape under certain conditions, thus blocking ions or letting them pass through the cell membrane. When exposed to light in the 300- to 500-nm range, so-called photochromic ligands open or close the gate by switching between their trans and cis forms.
At the University of Munich in Germany and at the University of California, Berkeley, scientists have teamed to investigate another group of molecules that ultimately may act as neuromodulators. Led by Dirk Trauner of the University of Munich, the group is working with a family of azobenzene molecules that target tetrameric voltage-gated ion channels, such as potassium-ion channels. These molecules include acrylamide-azo-quaternary ammonium (AAQ), benzoyl-azo-QA (BzAQ) and propyl-azo-QA (PrAQ). They are among the first molecules that can bind to naturally occurring, rather than genetically altered, potassium channels.
Using a family of azobenzene molecules and 380-nm light, investigators have found a way to block and unblock the voltage-gated potassium-ion channels in neurons.
Trauner and his team have found that 380-nm light – generated with a xenon lamp and narrowed with a bandpass filter – puts the AAQ into cis configuration, completely unblocking the potassium channels of neurons. Conversely, filtered 500-nm light puts the AAQ into trans mode, reblocking the channels.
Blocking more than 95 percent of potassium ions, BzAQ works as well as AAQ, the group reported in the Nov. 19, 2009, issue of Angewandte Chemie, but does so at a far smaller concentration – 25 µM vs. 400 µM. PrAQ blocks about half the ions at 40-µM concentration, but preferentially blocks while in cis mode.
More work must be performed to determine the selectivity of this new class of photochromic ligands as well as to investigate the molecules’ potential use with calcium channels, but Trauner and his team foresee using them to tackle vision problems and other neurobiological afflictions.