
Single-cell elasticity measured by atomic force microscopy
Lauren I. Rugani
The effect of external forces on living cells holds importance in
areas of study such as tissue engineering and cancer research. Forces induce biological
transformations on the cell that can alter functionality or mechanical behavior.
Two techniques that were combined and utilized to study these transformations, atomic
force microscopy and confocal optical microscopy, each have disadvantages when
used alone. However, when used together, they are sensitive to biological properties
and structural changes and enable the investigation of single-cell mechanics.
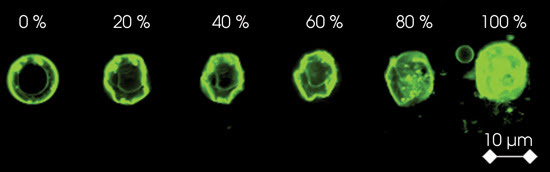
A single cell is exposed to compression
by atomic force microscopy. Membrane deformation followed by the rupture of individual
cellular components is evident as the load increases.
Researchers at the University of California in
Davis and in Sacramento have combined atomic force microscopy with optical microscopy
techniques to explore single-cell compression and its effect on the mechanical behavior
of cells. “Investigation of single-cell mechanics is essential for the characterization
and control of the mechanical properties and functions of reconstituted tissues,
an important task for practical application of tissue engineering,” said Gang-yu
Liu, a chemistry professor at the Davis campus.
To avoid staining the cells, most of
the experiments were conducted under bright-field optical microscopy with an inverted
optical microscope from Olympus America. An Olympus laser scanning confocal microscope
was used to image stained cells when the researchers needed a high-resolution visualization
of the three-dimensional cellular structure. “Confocal microscopy provides
accurate initial imaging for AFM investigation, including the positioning of AFM
probes to the designated cell location and simultaneous monitoring of cell deformation,”
Liu explained.
Cell deformation was measured with
an atomic force microscope from Asylum Research Corp., equipped with a nanopositioning
sensor. Silicon cantilevers with a force of either 1.0 N/m or 40 N/m, depending
on single-cell elasticity, were used for single-cell compression, force sensing
and high-resolution imaging. A 40-μm glass bead was attached to a cantilever,
and optical microscopy positioned the cantilever over the center of the cell to
monitor the deformation of the cellular components as force was applied.
The researchers created force versus
deformation curves for living, dead and fixed cells. For low applied loads, live-cell
deformation was found to be elastic and fully reversible, suggestive of an impermeable
cell membrane. As the load increased, most cells burst at roughly 30 percent deformation,
followed by the rupture of components within the cell, including the nucleus. These
specific events correlated with stress peaks in the force-deformation curves.
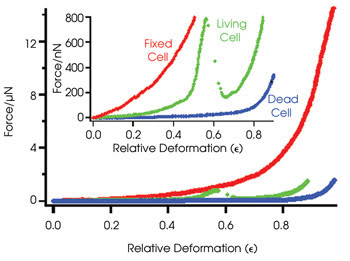
Force versus deformation curves demonstrate
the relative deformation — calculated as cell height change/initial cell height
— of living, dead and fixed cells. The stress peak of the living cell is evidence
of the membrane rupturing, while dead and fixed cells exhibit smooth curves. Fixed
cells require much higher forces than living and dead cells for relative deformation.
In contrast, dead cells exhibited a
smooth force-deformation curve, indicative of a uniform process that became irreversible
beyond 20 percent deformation resulting from high membrane permeability. Cells fixed
to the surface of the slide also exhibited smooth deformations; however, they required
much higher forces for compression and displayed full reversibility even after 60
percent deformation because of the cross-linking of membrane proteins.
The researchers found that AFM-based
positioning combined with optical monitoring improved accuracy for cell positioning,
mechanical perturbation and force measurements. “Combination of the two methods
enables high-resolution applications of AFM to be properly directed and utilized
in molecular and cellular biology investigations,” Liu remarked. However,
the instrumentation is expensive and complex. The team plans to improve the instrumentation
and methodology for easier use in biomedical research.
Langmuir, published online July 18, 2006.
Published: September 2006