
Luciferase-based cancer drug screen tested
David Shenkenberg
Because the p53 gene has mutated and does not function properly in more than half of all
human cancers, researchers developed a luciferase-based bioluminescent assay that
detects restored p53 gene expression upon the administration of an anticancer drug.
Dr. Wafik S. El-Deiry and colleagues at the University
of Pennsylvania School of Medicine in Philadelphia transfected p53-deficient cells
with a reporter construct that contained the luciferase gene so that restoration
of the p53 gene’s function resulted in luciferase production. They detected
its luminescence using a Xenogen Corp. workstation with a CCD camera so that they
could use the image to visually determine whether a drug worked. Imaging began
at the start of transcription, 12 hours after drug administration, and ended no
more than 72 hours after drug administration, when the effects of the drug appeared.
Using this system, the researchers assessed the effects of 2000 compounds from the
National Cancer Institute.
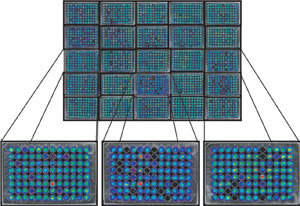
In this 5 x 5 grid of 96-well plates, human colon cancer cells deficient
in p53 carry a luciferase reporter that causes them to become bioluminescent upon
restoration of p53 function. Reprinted with permission of PNAS.
The screen was highly automated and
capable of measuring more than 200 cells per plate and thousands of cells in vivo,
but the investigators had to perform manual procedures to determine whether the
anticancer agent acted on p53 or on a closely related gene. They needed this information
because they believe that it will be easier to discover small molecules that target
related genes. In most cases, they determined which gene the anticancer drug affected,
but they could not identify the mechanism of two compounds.
Although El-Deiry said that he would
like to make his method more automatic, he maintained that some aspects, especially
simulating a tumor’s microenvironment, are best handled manually by experts.
He has a patent pending for the drug
screen and is in the market for developers. He said that genetic screens such as
his have not found their way into industry yet because the pharmaceutical companies
traditionally have followed the blockbuster drug model. However, he believes that
the screens eventually will become routine because targeted drug therapies could
save companies billions of dollars on clinical trials.
PNAS, July 18, 2006, 11003-11008.
Published: September 2006