Angela Goodacre, Dennis Donley and Jiwei Jai, Olympus America Inc.
Second-harmonic generation (SHG) is a label-free method of imaging structures in tissue using a pulsed near-infrared laser. It is generated by the nonabsorptive interaction of two photons with an orderly array of molecules that all point in the same direction. Known as inversion asymmetry, this property of directionality is found in certain types of collagen and elastin fibers, in myosin in muscle myofilaments and in microtubules of the mitotic spindle. Second-harmonic imaging is coherent, meaning that the signal propagates in a fixed direction and is sensitive to excitation light polarization relative to the sample.
The process occurs when two photons simultaneously interact with the sample, generating a scattering photon of exactly twice the incoming energy and therefore half the wavelength. It is nonlinear, occurring only at the focal plane where there is sufficient photon flux, so that it has inherent optical sectioning and thereby provides information on three-dimensional structures. The signal has a very sharp spectral peak at exactly half the wavelength of the incident light; it requires the use of a short-pulse laser system to deliver the intense photon density at the focal plane that is needed for imaging.
Combining methods
The technique can be combined with various methods of fluorescence imaging to deduce molecular organization and other key aspects of intracellular function; it is most often used with multiphoton excitation of either exogenous labels or of intrinsic fluorescence in cellular constituents such as flavins, NADH and retinols. Because SHG encodes information on molecular orientation, researchers can, for instance, image molecules labeled with GFP and then compare the fluorescence image, which shows all the molecules, with the corresponding image, where only molecules with a certain orientation will give rise to signal.
For example, in a mitotic spindle, the microtubules extend from both poles and interdigitate at the metaphase plate in the center. The GFP signal will be bright in the center, but no SHG signal will be generated because the perpendicular orientation of the microtubules from opposing poles will interfere with signal generation. At the spindle poles, where microtubules are radially arrayed around the centrosome, the intensity of the SHG signal varies with the coincidence of the laser polarization and the orientation of the microtubules, giving rise to crescent shapes.
The signal is strongest when detected in the forward or transmitted mode, using a high-numerical-aperture condenser or water-immersible “dipping” lens. The only molecule whose signal is strong enough to be detected in the backscattered (epi) direction is collagen. Thick tissue and in vivo imaging limit SHG to backscattered detection and thus to collagen imaging only. Rayleigh scattering of both the incident and SHG wavelengths can limit the depth of imaging through thick tissue, but researchers have now been able to achieve depths of greater than 500 µm through muscle tissue.
SHG, alone and in combination with multiphoton (usually two-photon excitation fluorescence, or TPEF) imaging, offers novel opportunities for investigating the three-dimensional structure of macromolecules within living tissue. SHG has several advantages over TPEF, including the ability to determine molecular orientation. Even more important, it offers reduced toxicity to cells because the photobleaching of fluorescent dyes creates toxic free radicals, whereas SHG uses no fluorophore and therefore shows no bleaching or associated toxicity. However, it has disadvantages as well – particularly its lack of absolute molecular specificity.
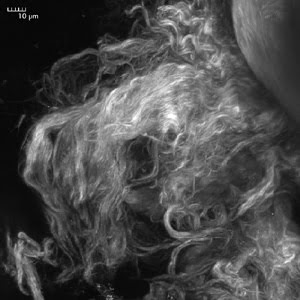
Figure 1. The SHG signal from 870-nm incident light is generated by collagen in a rat’s tail. The signal is collected using a 420- to 460-nm bandpass filter. The image is a maximum intensity projection of a Z-stack of 50 images spaced 2 μm apart and was captured using the Olympus FluoView FV1000-MPE with second-harmonic generation. Images courtesy of Claudio Vinegoni and Ralph Weissleder, Massachusetts General Hospital, Boston.
The process is now being used alongside multiphoton imaging in laboratories around the world to study a wide variety of phenomena in living tissue. Dr. Claudio Vinegoni of professor Ralph Weissleder’s Center for Systems Biology at Massachusetts General Hospital uses SHG to visualize collagen (Figures 1 and 2) using an Olympus system built around the Fluo-View FV1000 multiphoton system. In one version of the Olympus system, the transmitted light detector has a built-in option for forward collection of the signal.
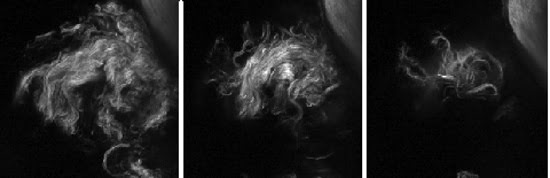
Figure 2. Three images show representative Z-sections spaced 10 μm apart, highlighting the optical sectioning inherent in SHG imaging. The SHG signal from incident light of 870 nm is generated by collagen in a rat’s tail. In other experiments, the SHG signal was collected simultaneously with multiphoton excitation of GFP at 495 to 540 nm using a 485DM beamsplitter.
Selecting the right wavelength
In use, SHG can be accomplished using a range of excitation wavelengths. The emission will vary accordingly, at half the excitation wavelength, with a sharp peak. Researchers select the optimal wavelength based on the following:
• Depth of penetration, as longer wavelengths are able to penetrate deeper
• Laser power available at each particular wavelength
• Whether other fluorophores will be imaged using TPEF
• Other intrinsic fluorescence to be imaged by TPEF, such as elastin, NADH or FAD. The SHG signal is first reflected into the detector light path, and then short-pass filters are used to isolate the signal from the laser and any fluorescence.
Among the assemblies and features frequently imaged using SHG are coiled-coil complexes within connective tissues and thick muscle filaments, matrices of collagen fibrils, sarcomeric repeats in the myo-filament lattice, microtubule arrays within interphase and mitotic cells, and starch granules in plant seeds. Other applications include studies in which fibrillar species such as collagen are altered in tissue.
Still another type of research involving the technique includes instances where providing a reference image of underlying tissue organization is beneficial. This includes studies of connective tissue, skin and muscle pathology, cornea, wound-healing, and invasion and metastasis in cancer. The effect of matrix metalloproteinases on collagen structure has been investigated in the development of cancer therapeutics. Myofilament assembly can be studied in muscle development and disease, and microtubules can be observed in the division cycles of normal and tumor cells. The advantage of SHG lies in the ability to image tissue in situ, without adding any dyes.
Third-harmonic generation (THG) also has been reported. It derives images from wavelengths exactly one-third of the excitation wavelength, typically collected in the forward direction. Unlike SHG, THG is the result of local material index of refraction difference, and it is not related to molecular spatial symmetry. To achieve the longer incident wavelengths needed for this process, an optical parametric oscillator is pumped by a Ti:sapphire laser to produce femtosecond laser pulses centered at 1100 to 1300 nm. Because of its low signal level, THG is applicable only to thin specimens, but a proportion of the signal may get reflected back as a bulk scattering process so that epidetection is possible in certain model systems.
THG microscopy has been used to image lipid bodies by virtue of the lipid-membrane interface. Starch granules in plant material and needle-shape crystals of malarial pigment in infected red blood cells also have been imaged using this technique.
Multiphoton excitation and SHG can be used in tandem for simultaneous imaging of both intrinsic fluorescence of cellular molecules and the array of macromolecules in the cell. These imaging modalities differ only in the placement of the detectors and filters in the optical path.
Meet the authors
Angela Goodacre is marketing manager, imaging systems applications, at Olympus America Inc.; e-mail: [email protected]. Dennis Donley is group manager for confocal laser scanning microscopes; e-mail: [email protected]. Yiwei Jia is a product manager; e-mail: [email protected].