PROVIDENCE, R.I., Dec. 23, 2010 — For the first time researchers have documented how single-walled carbon nanotubes are cut, a finding that could lead to producing more precise, higher-quality nanotubes.
According to the researchers at Brown University and in Korea, such manufacturing improvements likely would make the nanotubes more attractive for use in automotive, biomedicine, electronics, energy, optics and many other fields.
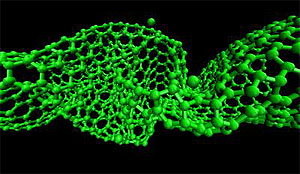
High-intensity atomic-level sonic boomlets cause nanotubes to buckle and twist at “compression-concentration zones.” (Image: Kyung-Suk Kim Laboratory, Brown University)
"We can now design the cutting rate and the diameters we want to cut," said Kyung-Suk Kim, professor of engineering in the School of Engineering at Brown and the corresponding author on the paper that was published in the British journal Proceedings of the Royal Society A.
The basics of carbon nanotube manufacturing are known. Single-atom-thin graphene sheets are immersed in solution (usually water), causing them to look like a plate of tangled spaghetti. The jumbled bundle of nanotubes is then blasted by high-intensity sound waves that create cavities (or partial vacuums) in the solution. The bubbles that arise from these cavities expand and collapse so violently that the heat in each bubble's core can reach more than 5000 °K, close to the temperature on the surface of the sun. Meanwhile, each bubble compresses at an acceleration 100 billion times greater than gravity. Considering the terrific energy involved, it's hardly surprising that the tubes come out at random lengths. Technicians use sieves to get tubes of the desired length. The technique is inexact partly because no one was sure what caused the tubes to fracture.
Materials scientists initially thought the superhot temperatures caused the nanotubes to tear. A group of German researchers proposed that it was the sonic boomlets caused by collapsing bubbles that pulled the tubes apart, like a rope tugged so violently at each end that it eventually rips.
Kim, Brown postdoctoral researcher Huck Beng Chew, and engineers at the Korea Institute of Science and Technology decided to investigate further. They crafted complex molecular dynamics simulations using an array of supercomputers to tease out what caused the carbon nanotubes to break. They found that rather than being pulled apart, as the German researchers had thought, the tubes were being compressed mightily from both ends. This caused a buckling in a roughly 5-nm section along the tubes called the compression-concentration zone. In that zone, the tube is twisted into alternating 90°-angle folds, so that it fairly resembles a helix.
That discovery still did not explain fully how the tubes are cut. Through more computerized simulations, the group learned the mighty force exerted by the bubbles' sonic booms caused atoms to be shot off the tube's lattice-like foundation like bullets from a machine gun.
"It's almost as if an orange is being squeezed, and the liquid is shooting out sideways," Kim said. "This kind of fracture by compressive atom ejection has never been observed before in any kind of materials."
The team confirmed the computerized simulations through laboratory tests involving sonication and electron microscopy of single-walled carbon nanotubes.
The group also learned that cutting single-walled carbon nanotubes using sound waves in water creates multiple kinks, or bent areas, along the tubes' length. The kinks are "highly attractive intramolecular junctions for building molecular-scale electronics," the researchers wrote.
Huck Beng Chew, a postdoctoral researcher in Brown's School of Engineering, is the first author on the paper. Myoung-Woon Moon and Kwang Ryul Lee, from the Korea Institute of Science and Technology, contributed to the research. The US National Science Foundation and the Korea Institute of Science and Technology funded the work.
For more information, visit: www.brown.edu
How nanotubes are cut from Brown PAUR on Vimeo.
Compression causes nanotubes to buckle and twist and eventually to lose atoms from their lattice-like structure. (Credit: Huck Beng Chew/Brown University)