The effectiveness of blue-light therapy for human skin is assessed in vivo using Raman spectroscopic biomarkers.
Dr. Nihal DeSilva and Dr. Simon Yu, Prevention & Healing Inc.
The goal of preventive medicine is to stop
illness before it begins, or to discover and treat disease before it spreads and
becomes serious. There are many approaches to preventive medicine today, including
screening for high blood pressure and diabetes, prescribing drugs to lower cholesterol
and recommending lifestyle changes, such as exercise and a balanced diet.
However, the fundamental understanding of health and disease is
dependent not so much upon genetic factors but more on epigenetic changes that are
reversible. These play a unique role in holistic medicine.1 It has been said that
epigenetic changes may be the root cause of disorders such as cancer, cardiovascular
disease, diabetes and more, and alternate therapies are being sought to address
these.
Color therapy can induce subtle yet dramatically beneficial health
effects in humans.2,3 In this study, color therapy using blue light was used on
subjects with a previous history of health conditions that had been monitored by
Raman vibrational spectroscopy.4 Our preliminary results indicate that blue light
significantly affected the skin meridian vibrational Raman spectral shifts, indicating
that the Raman technique with the attached fiber optic laser system is ideal for
studies of this nature. This report is the first investigation of in vivo acupuncture
meridian systems of the human skin using Raman spectroscopy.
Color therapy was administered on the patient by the Mora Color
instrument from Med-Tronik of Friesenheim, Germany. Blue light was applied to the
left hand as the subject held onto the color transfer probe for two minutes. Immediately
following this therapy, the right-hand finger meridian points (minimum of three)
of the subject were monitored by Raman spectroscopy.
Briefly, the Raman system used in this study consisted of a ProRaman-L
B-785 high-performance Raman analyzer from Enwave Optronics of Irvine, Calif. It
was equipped with a 785-nm frequency-stabilized, narrow-line diode laser and fitted
with a high-sensitivity CCD spectrograph cooled up to –60 °C. The spectral
range was between 250 and 3000 cm–1. Initially, the skin meridian points on both
hands were imaged using a 20x lens attached to the fiber optic probe, providing
the background information before the color therapy.
For all experiments, the laser power on the skin was kept to about
75 mW and the exposure time to 40 seconds per accumulation. The spectra were analyzed
using Raman reader data collection software fitted to a laptop computer for real-time
reaction monitoring; for spectral postprocessing, the software directly interfaced
with Grams/AI and Spectral ID by Thermo Scientific. Two spectral-range regions were
used to monitor the Raman shifts: (a) 250 to 2500 cm–1, referred to as the fingerprint
region, and (b) 250 to 3500 cm–1.5
The possibility of spectral artifacts caused by changes in pressure
on, and angle of contact with, the subject’s skin was considered in the collection
of data. Before real-time measurements were taken, two consecutive random scans
with a mild, moderate or firm probe measured skin tissue pressure with a 10-second
recovery period, with the probe lifted from the tissue surface between measurements.
From these determinations, mild pressure scans were deemed suitable
for obtaining optimum in vivo scan results for this study. Detailed principal component
analysis of spectra (for a larger subject-based study) will be considered later
to determine whether any systematic or significant differences exist due to probe
pressure or angle.6 In this study, three consecutive scans were taken for each meridian
point, and the mean intensity did not differ more than ±3 percent.
Results
Representative Raman spectra of Subject 1 (R.S.): Figure 1a shows
representative spectra of precolor therapy of the subject’s right meridian
points, while Figure 1b shows the corresponding spectra of postcolor therapy. Similar
scans were recorded for other subjects (data not shown). The spectral shifts can
be divided into two main regions, such as the mid-frequency fingerprint region and
the high-frequency region.
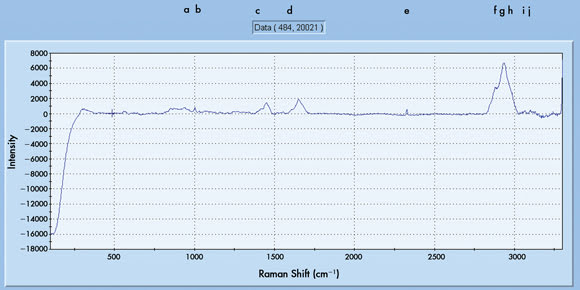
Figure 1. (a) Raman scan of skin monitored in vivo before color therapy
(major Raman shifts are labeled from a to h).
While specifically considering the mid-frequency region, the following
shifts (cm–1) were observed: 557, 871, 1000, 1072, 1343, 1443, 1557, 1614 and 1657.
However, the major shifts in relation to blue-light therapy were confined mainly
to 1443 and 1657 in this fingerprint region. These spectral shifts are assigned
mainly to skin proteins, sugars and some lipids. For example, the 800-cm–1 region
may represent C-H deformation due to beta linkages in proteins; similarly, the 1000-cm–1
shift region may represent the C-O and C-C stretch vibrations.7,8 The 1300- to 1550-cm–1
region may represent amide III vibrations, COO- stretch, CH3 bend and CH2/CH3 vibrations
derived from skin lipids and collagen II. Finally, the 1600- to 1700-cm–1 region
may indicate the amide I vibrations due mostly to skin collagen III.
It is generally known that the high-frequency region of the Raman
spectrum contains the lipid acyl chain methylene and methyl C-H stretching modes.
In this study, marked changes were also observed in this region with blue-light
therapy (compare Figure 1a with Figure 1b). The Raman shifts around 3000 cm–1 consist
of a complex of three interrelated peaks of about 2833, 2865 and 2930 cm–1. The
shifts at about 2833 and 2865 cm–1 refer to the acyl chain methylene (CH2) symmetric
and asymmetric stretching vibrations, respectively. The higher shift at about 2930
cm–1 represents a complex spectral interval that contains spectral components from
Fermi resonance interactions involving the chain methylene moieties and, separately,
the C-H symmetric stretching modes of the chain methyl termini of the skin lipids
(ceramides). The spectral intensity ratio m = I (2833 cm–1) / I (2865 cm–1) reflects
the pure lateral interchain-chain interactions.7 In comparison, the intensity ratio
n = I (2930 cm–1) / I (2865 cm–1) reflects the order-disorder arising from hydrogen
chain-chain interactions with some contributions from intrachain trans-gauche isomerizations.8,9
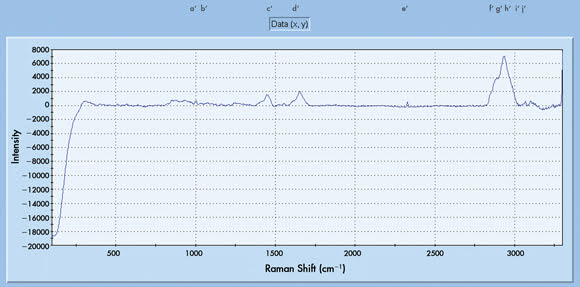
Figure 1. (b) Raman scan of skin monitored in vivo after color therapy
(major Raman shifts are labeled from a’ to h’).
The current results show that these ratios change with blue-light
color therapy. The overall augmentation of blue-light therapy on skin meridian Raman
shifts is denoted by ratio ABL:BBL, or after blue light to before blue light, as
indicated in Table 1. The Raman shifts attributed to the region between 1400 and
1600 cm–1 showed nearly a twofold increment compared with the region between 2800
and 2950 cm–1. This may indicate that the effect of blue light is more pronounced
on Raman vibrations due to protein structural components such as the amide III,
C-O and C-C, and due to lipidic structures (ceramides, phospholipids) such as COO-,
CH3 and CH2 vibrations.9 As indicated in Table 1, the high-frequency (2800 to 2950)
Raman spectral augmentation by blue light is more than one order of magnitude compared
with the control.
Similarly, the ratio m is low, as the Raman vibrational shift
at 2833 cm–1 before color therapy is of low intensity (0.42 to 0.46, Table 1). However,
with blue light, the increase in intensity of this vibrational shift is evident
(Figure 1b). This indicates that the m value does not change a great deal with the
blue-light therapy in this subject.
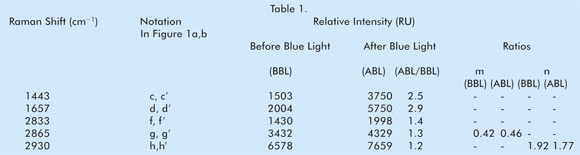
Table 1. Comparison of Raman vibrational shifts
monitored in vivo before and after blue-light color therapy (Raman shifts with notations
such as a, b, e and so on were omitted when they did not show a significant change
in intensity).
However, the n values did change with blue-light therapy, the
ratio being about 1.92 before the color therapy compared with 1.76 after. This data
taken together means that the same lateral chain-chain ordering, but with a different
intrachain structure, may take place with a different degree of perturbation at
varying depths of the skin cell bilayer for this particular meridian point in this
subject. Whether this would mean a dampening effect of overcoming adverse conditions
by blue light on skin cellular pathways needs further work. We are continuing this
work with more subjects so that we can continue to use Raman spectral biomarkers
to understand blue-light color therapy.
Conclusions
1. Blue-light therapy has a definite augmentation effect on the
relative intensity of Raman vibrational spectral shifts as investigated in vivo
on the thumb skin of a human subject.
2. The augmentation effect was elevated by a 2.5-fold increase
in the low-frequency Raman shifts compared with a nearly 1.4-fold increase in the
high-frequency region. Minor changes in lipidic molecular chain-chain interactions
and order-disorder perturbations were also evident.
3. The data indicates that the effect of blue light is more pronounced
on certain specific Raman shifts due to protein and lipidic biomolecular structures
in the skin. Further work is being pursued to decipher the exact physiological effects
of blue light on the skin meridians in health and disease.
Meet the authors
Dr. Nihal DeSilva and Dr. Simon Yu work at Prevention & Healing
Inc. in St. Louis; e-mail: [email protected].
References
1. S. Yu (2010). Accidental Cure.
2. D.C. Holzman (January 2010). What’s in a color? The unique
human health effects of blue light. Environ Health Perspect, A22-A27.
3. L. Marshall (April 2010). Mouse model shines light on locomotor
circuits. Biophotonics, p. 8.
4. P. Lasch and J. Kneipp, eds. (2008). Biomedical Vibrational
Spectroscopy. Wiley-Interscience.
5. X. Nan et al (August 2004). CARS microscopy lights up lipids
in living cells. Biophotonics Int., p. 44.
6. M.G. Shim et al (July 2000). In vivo near-infrared Raman spectroscopy:
demonstration of feasibility during clinical gastrointestinal endoscopy. Photochem
Photobiol, pp. 146-150.
7. N.S. DeSilva (September 1999). Structural changes in the lipoprotein
complex of bronchoalveolar lavage (BAL) fluid detected by Raman spectroscopy. Spectros
Int J, pp. 21-24.
8. N.S. DeSilva et al (June Suppl. 2004). Identifying macromolecules:
The authors review a surface enhanced laser Raman spectroscopy study of lung surfactant
protein interaction with bacterial lipopolysaccharide. Spectros Int J, pp. 48-52.
9. G. Zhang et al (2008). Interplay of univariate and multivariate
analysis in vibrational microscopic imaging of mineralized tissue and skin, In:
P. Lasch and J. Kneipp, eds. Biomed Vibrat Spectros, Wiley Interscience, pp. 357-378.