Compiled by BioPhotonics staff
A new fluorescence labeling technique that can monitor the reversible plasma membrane
association of protein domains without the need for scanning, optical splicing or
imaging has been identified by researchers. The method uses fluoxetine, an active
ingredient in Prozac that suppresses the activity of the potassium channel protein
TREK1, activity associated with mood regulation.
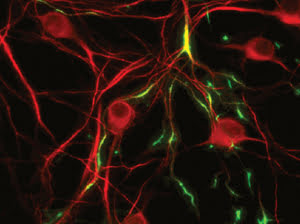
TREK1 potassium ion channels, fluorescing green in these cultured
neurons, have been linked to the regulation of emotions. Images courtesy of Isacoff
group, UC Berkeley.
Investigators at the University of California, and at the US Department
of Energy’s Lawrence Berkeley National Laboratory found that the previously
discovered inhibition of TREK1 by fluoxetine is accompanied by an unbinding of the
protein’s C-terminal domain from the membrane. This is the first observation
that TREK1 might be regulated by antidepressant drugs.
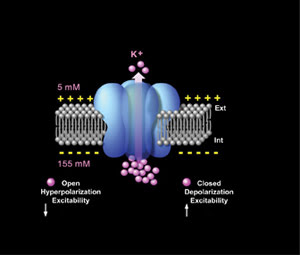
The TREK1 ion channel, shown in blue, controls the passage of potassium ions (pink) through the plasma membrane
(gray) of neurons, which sets neuron excitability.
In the brain, TREK1 acts as a gate to the passage of potassium
ions through the neural membrane, which sets the excitability of the neuron. Previous
studies conducted in mice showed that, when the TREK1 gene was “knocked out,”
the mice displayed a depression-resistant phenotype that mimicked the behavior of
mice treated with fluoxetine. It also showed that the antidepressant inhibited the
activity of the TREK1 channel. Although the results indicated a possible role of
the TREK1 ion channel in response to fluoxetine, the mechanism behind the activity
was unclear.
After developing techniques to label the channel proteins with
site-specific fluorescent dyes, the investigators detected structural rearrangements
of the labeled sites in the channel through changes in the fluorescence. They separated
the C-terminal domain from the rest of the protein, tagging it with a GFP (green
fluorescent protein). Unlike the TREK1 domain, in which the pore is embedded in
the plasma membrane of a neuron, the C-terminal is a short tail that protrudes into
the surrounding cytoplasm.
Using voltage clamps to measure electrical currents through the
channel and fluorescence to monitor the disposition of the terminal’s domain,
the scientists discovered that, when the C-terminal tail was fully bound to the
plasma membrane, the TREK1 channel opened more, but when it was unbound from the
membrane, the ion channel tended to close. They concluded that fluoxetine causes
the isolated C-terminal domain to unbind from the membrane and also causes an inhibition
of current from the full TREK1 channel.
The new assay has potential for applications in pharmaceutical
research and could be used to follow changes in lipid composition that results from
membrane signaling events or to study the binding of membranes by cytoplasmic regulatory
domains of ion channels. Future research will be conducted to see how the C-terminal
tail is affected by the presence of fluoxetine when it is still attached to the
rest of the TREK1 protein.
The research, which appeared in the journal Proceedings of the
National Academy of Science (PNAS), Jan. 24, 2011 (doi: 10.1073/pnas. 1015788108),
was supported primarily by the National Institutes of Health.