Observing what happens when atoms interact or proteins fold requires picosecond or faster imaging. Recent innovations promise to allow researchers to capture previously invisible events. That capability could have industrial and medical uses, as can be seen with examples of imaging below and above the visible spectrum.
For ultrafast
imaging at wavelengths well below the visible, consider the work being done by a
team led by professors Henry Kapteyn and Margaret Murnane. The married couple holds
appointments in physics at the University of Colorado at Boulder and JILA, a physical
science research institute jointly operated by the university and the National Institute
of Standards and Technology. Their research focuses on generating and using coherent
laserlike x-rays.

An ultrafast laser frequency upshifted into a rainbow of laserlike x-rays spans
the entire soft x-ray region of the spectrum. This enables any element in a sample
to be identified chemically, or a movie of any reaction to be made in real time.
Courtesy of Tenio Popmintchev, JILA.
As described in the December 2010 issue of Nature Photonics, they
start with a femtosecond laser pulse and send it into a gas cloud inside a hollow
waveguide. The resulting high harmonic generation produces an output that can be
thousands of times shorter in wavelength.
The laser and gas determine the output wavelength and its brightness.
For instance, a Ti:sapphire beam at 800 nm and argon yield a 29-nm wavelength output,
while the use of helium leads to a 13-nm output. Recently, the group has figured out how to produce
light of nanometer wavelengths, which somewhat paradoxically will need mid-IR lasers
operating at 1.3 μm and longer, Murnane said.

An x-ray converter developed at JILA takes an ultrafast laser and converts it into
laserlike beams at much shorter wavelengths and pulse duration. The laser accelerates
electrons within an atom that then radiate a rainbow of laserlike x-rays. Reprinted
from Nature Photonics. Courtesy of Tenio Popmintchev and Brad Baxley, JILA.
With shorter wavelengths, the investigators will improve imaging
resolution, since the classical diffraction limit is about half the wavelength.
The push down from extreme-ultraviolet, at 13 nm, into x-rays as short as 1 nm should
allow an equivalent enhancement in resolving object details.
There’s another effect of this approach, Kapteyn said. “If
you do it under the right conditions, you generate an attosecond pulse.”
Like the wavelength, the pulse duration is thousands of times
smaller than the original. Results indicate that pulses of about 5 as can be generated.
At 10−18 seconds, an attosecond is so short that light travels only one-third
of a nanometer. These brief bursts of light should allow the capture of electron
dynamics in materials and molecules.
Combined laser and x-ray beams can excite and probe a molecule
to follow how the electron density and atoms move during chemical reactions. Courtesy
of Greg Kuebler, JILA.
For imaging, the researchers illuminate an object with a coherent
beam and collect the scattered light. Visually, this looks like a mess, but it contains
information from which spatial data can be extracted.
What’s more, light below 4 nm is absorbed by elements such
as carbon and nitrogen and so can provide elemental information. In particular,
water is relatively transparent in this region, but carbon is strongly absorbing,
leading to an interesting possibility.
“You can image carbon content in an x-ray image with 10-nm
resolution for a field of view that’s about the size of a single cell,”
Kapteyn said.
Nonbiological uses of the technique could include tracking the
dynamics of semiconductors by following transistor heat dissipation. For hard disks,
changes in data bit magnetization could be measured, an important topic as the industry
strives to make higher-capacity disks.
Imaging is done by having the energetic photons directly strike
a CCD sensor, which causes some chip damage. The technique is also limited to imaging
depths of only a few microns, and the beam itself has to travel in vacuum. The sample
being imaged can sit in atmosphere, pressed up against a transparent window.
The key to the latest advance has been a better understanding
of the nonlinear optics that transform an infrared beam into x-rays. Decades ago,
the first x-ray lasers were powered by stadium-size sources, but with this breakthrough,
that situation has changed.
“We can generate very useful amounts of x-ray light, and
it’s on a tabletop,” Kapteyn said.
So far, the group has done extreme-ultraviolet imaging in three
dimensions in space and in one dimension with time resolution. The investigators
plan to combine these and lower the wavelength, thereby enabling nanoscale imaging
with subfemtosecond resolution. Kapteyn and Murnane formed a company more than a
decade ago to commercialize their research and innovations.
In addition to ultrafast imaging below the visible, there’s
work under way to do the same at wavelengths that are much longer. Take, for example,
research being done at Imperial College London by physics professor Chris Phillips.
His group demonstrated ultrafast infrared chemical imaging of live cells, reporting
on the work in the January 2011 issue of Chemical Science.
They were able to capture an image in about 100 ps, roughly 100
billion times faster than current mid-IR spectroscopic imaging techniques can. They
achieved this through two innovations. The first was a new source, and the second
was a new detector.
Of the two, the source was the more important, Phillips said.
“Other than large scientific installations like synchrotrons and free-electron
lasers, there’s really nothing in that part of the spectrum that tunes across
the wavelength range of interest and gives anything like the time resolution we
can manage.”
Using a custom-built optical parametric amplifier, the source
can deliver peak power of 100 W to the sample, starting with a 2.78-μm beam
generated from a doped yttrium scandium gallium garnet rod. This pulsed beam passes
through a zinc germanium phosphide crystal, shifting the wavelength of the pulse.
By altering the crystal angle, the output can be tuned over the range from 3 to
more than 10 μm.
The detector is a commercial mercury-cadmium-telluride focal plane,
modified by removing thermal background-suppressing filters. That allows the system
to image across the entire biologically important 2- to 9.7-μm range. Those
wavelengths are sensitive to chemical bonds and so highlight the dynamic chemistry
of a sample.
The researchers have started to use the setup to study cancer
biopsy specimens, with the hope that the chemical maps thus produced will allow
rapid differentiation of diseased from healthy tissue.
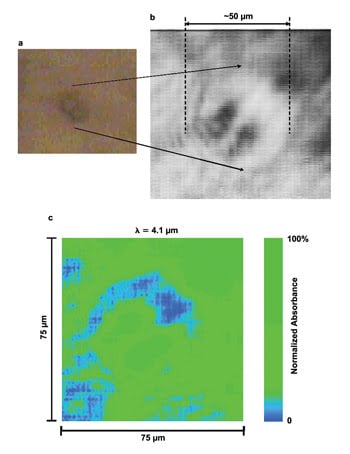
A mid-infrared
image of a single live SK-OV-3 human ovarian cancer cell undergoing mitosis. Visible
image (a) and IR transmission image (b) at 4.1 μm, along with false-color IR
image (c). Dividing nuclei of the cell are clearly visible. The image was acquired
in 100 ps at a 1.9-μm pixel size. Reprinted from Chemical Science. Courtesy
of Chris Phillips, Imperial College London.
Aside from possible diagnostic applications, Phillips noted that
other uses will include tracking the way chemicals move around in a cell. Of particular
interest will be how cells respond to external triggers, such as necrosis factors
and drugs.
Recent source advances also promise to make ultrafast imaging
a reality at even longer wavelengths, said Daniel Mittleman, a professor of electrical
and computer engineering at Rice University in Houston. He is involved in terahertz
imaging, which covers wavelengths from 100 μm to 1 mm.
Ironically, although femtosecond lasers have long been a preferred
method for generating and detecting terahertz radiation, the field has not employed
ultrafast imaging. In fact, there has been virtually no time-resolved imaging at
all, Mittleman said.
The issue has been a lack of high-intensity ultrafast terahertz
pulses. The situation has changed in the past year or so, and Mittleman expects
that terahertz ultrafast imaging is on the horizon. When it does arrive, the long
wavelengths will allow the tracking of phonons, the quantized vibrations found in
a crystal lattice, or the folding of a protein.
However, those long wavelengths also pose a challenge. They’re
much bigger than a cell or other items of interest, which renders conventional imaging
impossible. Researchers are working to overcome this limitation, turning to near-field
imaging and other techniques to resolve small objects.
They’ve had some success, and that could bode well for future
ultrafast terahertz imaging. As Mittleman said, “People have been able to
image hundred-nanometer objects with terahertz radiation that is hundreds of microns
in wavelength.”
Not ultrafast, but faster
Although researchers have been
focusing on sources, detectors have also been improving. One example comes from
Teledyne Dalsa, the Waterloo, Ontario, Canada-based camera maker. In December 2010,
the company announced a CMOS line-scan camera with 1146-megapixel-per-second throughput.
The data rate is high enough that the camera requires a new interface. Dubbed HSLink,
it forms the foundation for Camera Link HS, a proposed successor to the interface
standard Camera Link.
Another example comes from Fairchild Imaging, a Milpitas, Calif.-based
company – currently being acquired by British defense contractor BAE Systems
– that makes both CMOS- and CCD-based imaging systems. The company realized
a few years ago, said Colin Earle, vice president of sales and marketing for Fairchild
Imaging, that the scientific community could use a faster, higher sensitivity sensor
with more dynamic range than CCDs offer. The company therefore developed its scientific
CMOS sensors. Fairchild Imaging partnered with both Andor Technology plc of Belfast,
UK, and Kelheim, Germany-based PCO in bringing to market systems that are based
on the technology.
Fluorescent-labeled BPAE cells, captured with a Fairchild Imaging
SciMOS camera. Courtesy of Fairchild Imaging.
Earle said that the sensors and imaging systems based on them
have been well received. They offer capture rates of up to 100 fps, about five times
faster than comparable CCD sensors, but do so without sacrificing what’s important
to the target market.
In particular, researchers want a low-noise sensor, Earle said.
“A scientist cares about low noise because he wants to be able to measure
faint signals, and he’s got to ensure that his noise floor is below what he’s
trying to measure without resorting to multiplicative gain techniques that introduce
further uncertainty.”