Compiled by BioPhotonics staff
Using an extremely thin sheet of light, a new
type of microscope reveals the 3-D shapes of cellular landmarks in never-before-seen
detail. The technique images live cells at high speed, creating movies of biological
processes such as cell division.
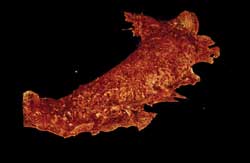
The ruffled membrane of a monkey
kidney cell, as observed by Bessel beam plane illumination microscopy. Images courtesy
of Eric Betzig, Thomas Planchon and Liang Gao.
Known as Bessel beam plane illumination microscopy, the method
was developed by researchers at Howard Hughes Medical Institute’s Janelia
Farm Research Campus. Their findings were published online March 4, 2011, in Nature
Methods (doi: 10.1038/nmeth.1586).
Their goal was to design a microscope that would reveal the dynamics
of living cells while minimizing the light damage and fading of the fluorescent
labels associated with live-cell imaging.
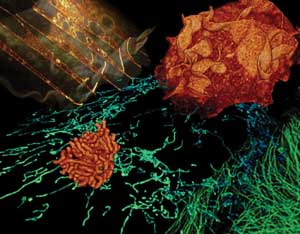
A montage of live cells imaged by Bessel beam plane illumination microscopy. Clockwise
from upper left: internal architecture and vacuoles in a monkey kidney cell (gold);
membrane ruffles at the surface of a monkey kidney cell (orange); microtubules in
a pig kidney cell (green); mitochondria in a human osteosarcoma cell (blue); and
chromosomes during mitosis of a pig kidney cell (orange).
Although plane illumination has been used to study multicellular
organisms, previously the sheets of light have been too thick to image effectively
within single cells only tens of microns in size. The wide swath of light also exposed
more of the cell than the scientists desired. To circumvent this, the group used
a Bessel beam – a nondiffracting light beam used in applications such as bar-code
scanners – to create a thinner light sheet across the sample.
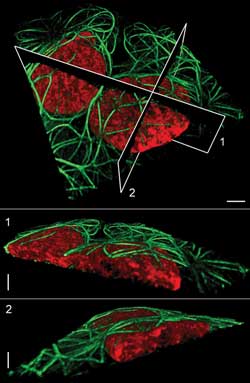
Microtubules
(green) surround nuclei (red) in a pair of live human osteosarcoma cells. Planes
at top show the location of the cutaway views below. Scale bars = 5 μm.
The team noticed that the Bessel beam behaved a bit strangely,
however. Besides a very narrow light beam, the beams also created a weaker light
that flanked the focal point, creating an illumination pattern that looked like
a bull’s-eye. To counteract this, the team used two tricks: two-photon microscopy
and a concept called structured illumination, which turns a beam on and off rapidly
over a sample.
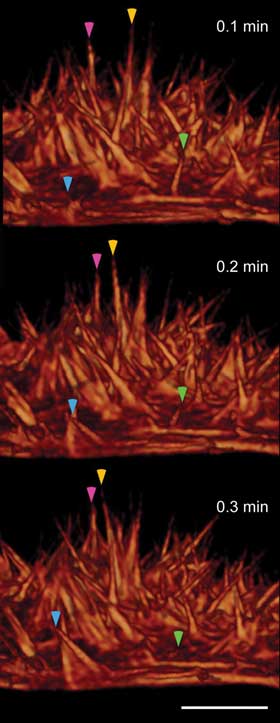
Dynamics of actin-based filopodia at the surface of a live HeLa cell,
at 6-s intervals, show filaments that wave (magenta and yellow arrowheads), extend
outward (cyan arrowhead) or retract inward (green arrowhead). Scale bars = 5 μm.
To image a sample as fast as possible, they used a Bessel beam
to sweep quickly through the sample, taking nearly 200 images per second; almost
simultaneously, in 1 to 10 seconds, a three-dimensional stack was built from hundreds
of two-dimensional images. The scientists took hundreds of such 3-D image sets without
harming the cell, generating movies of mitosis, for example.
The new instrument may be used in the future to apply superresolution
microscopy to thicker cells. In any case, this technique’s ability to image
the rapidly evolving 3-D complexity of cells noninvasively should make it a powerful
tool, the team said.