By Karen Newman, Group Publisher
On the way to conjoining two well-established but disparate imaging modalities, a FLIM (fluorescence lifetime imaging) and CARS microscope was achieved along with the development of an adapter that could bring real change to the way microscopists use their instruments.
Working on a grant to study the hepatitis C virus, scientists at National Research Council of Canada, Steacie Institute for Molecular Sciences, in Ottawa, along with colleagues from National Yang-Ming University, Taipei, and Queen’s University, Ontario, sought a modality to study lipids. They decided to combine FLIM and CARS (coherent anti-Stokes Raman scattering) as a step toward combining FRET (Förster resonance energy transfer) and CARS (for an explanation of CARS, see CARS Microscopy Made Simple, BioPhotonics, October 2009.
“In the field of HCV infection, there are models which suggest that the interface of a lipid body may be an important step in viral assembly,” said Albert Stolow, principal research officer and program leader, molecular photonics at Steacie Institute for Molecular Sciences. “The lipid bodies are imaged by CARS, whereas tagged viral RNA can be imaged by fluorescence. The combined FLIM-CARS technique could help validate these models.” Knowing if such an interaction is, in fact, a key step in HCV infection will lead researchers to develop new approaches to stopping its progress.
“An advantage of multimodal microscopies is to spatially and temporally correlate images obtained using different methods, for example CARS and SHG microscopy. The ability to combine CARS with other methods such as SHG, two-photon fluorescence, FLIM, etc., enhances researcher's abilities to determine mechanisms,” said Stolow. “For example, a co-localization study using fluorescence and CARS may allow researchers to determine that two molecules are within the same focal volume. Unfortunately, they could still be 100 nm apart since the spatial resolution in nonlinear microscopy is not better than, say, 300 nm radially.
“Two molecules that are 100 nm apart know nothing about each other; co-localization within the laser focal volume is not proof of chemical interaction,” said Stolow. “However, we know from the fields of FLIM and FRET microscopy that these effects are sensitive to short-range electric interactions (say, within 5nm) which alter fluorescence lifetimes or energy transfer processes. By combining FLIM + FRET with CARS, we can use the CARS to determine if two molecules are within 300 nm of each other and, simultaneously, using FLIM-FRET to determine that they are also within 5 nm of each other. You can think of it as being analogous to a vernier technique.”
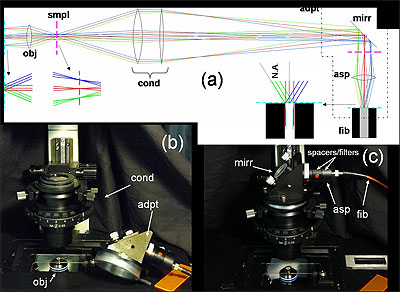
(a) Fiber-coupling geometry (not to scale): obj: objective; smpl: sample plane; cond: condenser; adpt: fiber coupling adapter; mirr: mirror; asp: aspheric lens; and fib: multimode fiber. Cyan dashed line and magenta dashed line denote objective back aperture conjugate planes and sample conjugate planes, respectively. (b) and (c) Annotated photos of microscope with fiber-coupling adapter beside and affixed to the condenser housing, respectively.
Before showing full FRET + CARS on the HEPC lipid virus, the researchers wanted to show FLIM in the forward direction, according to Aaron Slepkov, the group’s lead communicator. This required adapting a commercially available microscope from detecting in the backward direction to the forward for the experiment.
In a paper published by Journal of Biomedical Optics in February 2011, Stolow and his team describe the “simultaneous collection and separation of femtosecond-laser-based F-CARS and two-photon-excitation-induced FLIM using time-correlated single photon counting (TCSPC).”
The demonstration, as outlined in the paper, was achieved “in a nondescanned geometry using a single multimode fiber without significant loss of light, field of view, and most importantly, TCSPC timing fidelity,” indicating the acknowledgement and resolution of several significant challenges to combining FLIM and CARS, and bringing researchers a step closer to conjoining FRET and CARS.
Enabling technology
A small technical development that was critical to the success of the experiment and is described in the paper will make a big difference in the life of the microscopist, according to Slepkov. The adapter developed is attached to the condenser of an inverted laser-scanning microscope, where it couples light into a standard multimode fiber. From there, the light is routed to off-board detectors.
By employing a single, multimode fiber instead of a bundle, signals can be sent to any off-board detector with almost perfect fidelity -- with the room lights on. “Scientifically it is almost trivial, but when it comes to how scientists use their equipment, it is quite an interesting advance,” said Slepkov. “It really changed the way I use my microscope.”
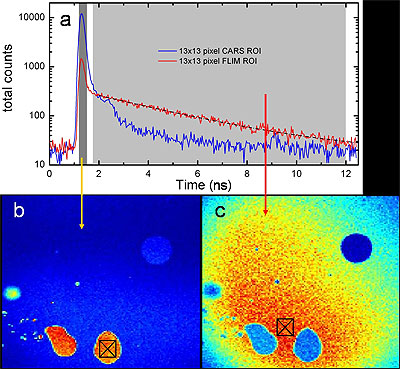
Time-gated F-CARS and FLIM imaging on a single detector. The sample is comprised of oil droplets in an aqueous fluorescein solution. (a) TCSPC timing histogram of the two 13x13 pixel regions of interest marked in (b) and (c). The two time gates are shown in gray regions. (b) Image of all photons arriving in the vicinity of the IRF peak, representing CARS signals from the oil. (c) Image of all photons arriving between 500 ps after the IRF peak and the end of the laser repetition period, representing the TPEF signal from fluorescein. A FLIM analysis in (a) yields a relaxation lifetime of 3.4 +/- 0.3 ns for fluorscein. The blue circle in the upper right corner of both images is identified as an air bubble. For (b) and (c) color scales from blue (low) to red (high) with the figures independently scaled for contrast.
In a further advance from the research, the team developed what Slepkov called a clever coupling geometry that involves NOT focusing light into the face of the fiber but rather sending a very particular imaging plane into the fiber.
Making it simple
Stolow and his team are looking for applications for using CARS for real-world problems. CARS has been promoted for many years as the modality of the future, said Slepkov, but the equipment necessary to run it has kept it on the edge of use and acceptance.
The team had already developed a CARS microscope label-free chemical modality and integrated it into a nonlinear microscope platform that is widely available.
“We integrated CARS into it driven by a philosophy of doing this as simply and effectively as possible. That led to some success. Since then we’ve been looking to expand capability of this scope to do more things – use CARS microscopy to solve real-world problems,” said Slepkov.
Stolow said his laboratory specializes in laser physics, ultrafast molecular dynamics and nonlinear optics, and that he and his team have no difficulties in implementing complex techniques in that stable lab environment. He acknowledges that not all researchers have access to a lab like his.
“Biomedical researchers should not be required to have expertise in femtosecond nonlinear optics before they can explore new imaging modalities,” said Stolow. “The challenge is to make things simple enough that anyone can use them.”
The simplified approach his group employed was commercialized by Olympus Corp. in October 2009, producing the world's first commercially available CARS microscope and the joint launching of a CARS Microscopy User Facility. “We also demonstrated the first live-cell CARS microscopy imaging using all fiber lasers,” Stolow said. “In the not too distant future, all nonlinear microscopy will be achieved using turn-key fiber lasers.”
The next step for the team in reaching its goal of FRET +CARS is demonstrating that it can be used to solve a real problem, such as the mechanism of HCV infection.
For Stolow, the ultimate in multimodal imaging is inexpensive, turn-key, telecom-stability-fiber-based ultrashort pulse lasers which will be widely available to microscopy facilities in hospitals and clinics.
In thinking about his work and the results of his team’s recent efforts, Stolow added that nonlinear optics is a very well established field of physics, having begun in 1961. The CARS effect was first discovered in 1965. He believes there are many well-known nonlinear optical spectroscopy methods and techniques that can apply to nonlinear optical microscopy. But which of these is the most useful and important for biomedical imaging remains yet to be seen.
[email protected]