Compiled by Photonics Spectra staff
A novel cellular research platform uses nanopillars that glow in
such a way as to allow a deeper and more precise look into living cells.
A Stanford University team led by chemist Bianxiao Cui developed
the precise system of illumination.
Earlier forms of molecular imaging shined light directly on the
subject area rather than using backlighting, as is used in this approach. With the
earlier methods, the focal area was physically limited; the minimum observation
volume was subject to the diffraction limit, whereas individual molecules are much
smaller than visible light’s typical limit of about 400 nm.
To overcome these hurdles, the team used quartz nanopillars that
glow just enough to provide light to see by, but are weak enough to punch below
the 400-nm barrier. The field of light that surrounds the glowing nanopillars, known
as the “evanescence wave,” dies out within about 150 nm of the pillar,
thus acting as a light source smaller than the diffraction limit. Cui estimates
that they have shrunk the observation volume to one-tenth the size achieved by previous
methods.
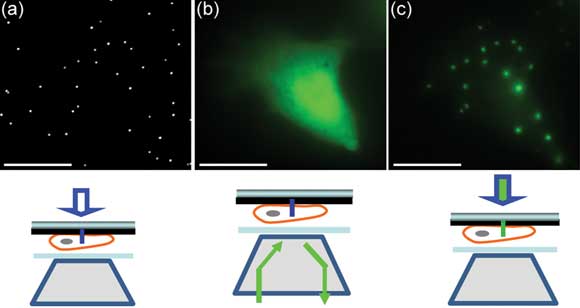
Fluorescence imaging using nanopillar illumination in live cells. (a) White-light imaging reveals the locations of nanopillars. (b) Fluorescence imaging by epi-illumination shows the shape of a cell transfected with green fluorescent protein. (c) Nanopillar illumination excites only those fluorescence molecules that are very close to nanopillars inside the cell, giving rise to fluorescence spots perfectly colocalized with the nanopillars. Courtesy of Stanford University.
The technique does not harm cells under observation – a
drawback of some earlier technologies. For example, living neurons can be cultured
on the platform and observed over long periods of time. In addition, the nanopillars
essentially pin the cells in place, which is promising for the study of neurons
in particular because they tend to move as a result of the repeated firing and relaxation
necessary for study.
The scientists also discovered that by modifying the chemistry
on the surface of the nanopillars, they could attract specific molecules they wanted
to observe. This enabled them to handpick molecules to study, even within the crowded
and complex environment of a human cell.

(Left) A scanning electron microscope image of a cell grown over and interacting with nanopillars. Arrows indicate three nanopillars. (Right) Image of the interface of cell (purple)
and nanopillar shows cell membranes wrapped around the pillar. (Image: Bianxiao
Cui, Stanford University)
“We know that proteins and their antibodies attract each
other,” Cui said. “We coat the pillars with antibodies, and the proteins
we want to look at are drawn right to the light source – like prima donnas
to the limelight.”
To create the nanopillars, the researchers used a sheet of quartz,
which they sprayed with fine dots of gold in a random pattern. Using a corrosive
gas, they etched the quartz. The gold dots shielded the quartz directly below from
the etching process, leaving behind tall, thin pillars of quartz. The nanopillars’
height can be controlled by adjusting the amount of time the etching gas is in contact
with the quartz; their diameter, by varying the size of the gold dots. After the
etching process was completed, a layer of platinum was added to the flat expanse
of the quartz at the base of the pillars.
The scientists then shined a light from below their creation.
The opaque platinum blocked most of the light, while a small amount traveled up
through the nanopillars, which glowed against the dark field of platinum and looked
much like a forest of tiny light sabers.
The team has created a platform for culturing and observing human
cells. The platinum is biologically inert, and the cells grow over and closely adhere
to the nanopillars. The glowing spires then meet with fluorescent molecules within
the living cell, causing the molecules to glow – providing the researchers
just the amount of light they need to peer inside the cells.
The work appeared in the Feb. 22, 2011, issue of PNAS (doi: 10.1073/pnas.1015589108).