Compiled by BioPhotonics staff
Three-dimensional plasmon rulers that can measure nanometer-scale spatial changes in macromolecular systems could provide scientists with unprecedented details on critical dynamic events in biology – including the interaction of DNA with enzymes, the motion of peptides, the folding of proteins or the vibrations of cell membranes.
The new measurement technique, developed by researchers at Lawrence Berkeley National Laboratory in collaboration with the University of Stuttgart, is based on coupled plasmonic oligomers in combination with high-resolution plasmon spectroscopy. The 3-D plasmon ruler enables scientists to retrieve the complete spatial figuring of complex macromolecular and biological processes, and to track the dynamic evolution of these processes, said Paul Alivisatos, Berkeley Lab director and head of the research.

The spatial freedom afforded the 3-D plasmon ruler’s five nanorods enables it to measure the direction as well as the magnitude of structural changes in a macromolecule sample. Images courtesy of Paul Alivisatos’ research group, Berkeley, Calif.
As human machines and devices shrink to the size of biomolecules, scientists have found a need to develop tools that can precisely measure minute structural changes and distances on the nanometer scale. To meet their needs, researchers have developed linear rulers based on plasmons.
Compared to other types of molecular rulers – based on chemical dyes and Förster resonance energy transfer – plasmon rulers neither photobleach nor blink, and they offer exceptional brightness and photostability. Until recently, plasmon rulers could be used to measure distances only along one dimension, making it difficult to understand the biological and other soft-matter processes that take place in three dimensions.
To combat this problem, the scientists used dipolar and quadrupolar modes to create sharp spectral features in plasmon-coupled nanostructures. Typical dipolar plasmon resonances are broad because of radiative damping. As a result, the simple coupling between multiple particles produces indistinct spectra that are not readily converted into distances. The scientists overcame this problem with a 3-D ruler constructed from five gold nanorods of individually controlled length and orientation.
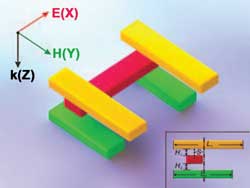
A 3-D plasmon ruler is constructed from five gold nanorods, one of which (red) is placed perpendicularly between two pairs of parallel nanorods (yellow and green).
The strong coupling of the nanorods suppressed radiative damping, allowing the excitation of two sharp quadrupolar resonances that enabled high-resolution plasmon spectroscopy to occur.
To prove their concept, the scientists fabricated a series of samples using high-precision electron beam lithography and layer-by-layer stacking nanotechniques. They then embedded these with the 3-D plasmon rulers in a dielectric medium on a glass substrate. The end result agreed with the calculated spectra.
The collaborators say that the 3-D plasmon rulers could someday be attached – through biochemical linkers – to samples of macromolecules such as strands of DNA or RNA, or at different positions along a protein or peptide, and the samples’ optical responses could be measured via dark-field microspectroscopy upon exposure to light.