Surface plasmon resonance techniques show promise as the new standards of drug discovery, especially for treating cancer.
In the realm of drug discovery, the hunt is ever on for new combinations of analytes and receptors. Respectively, these are the chemicals that may become useful new pharmaceuticals and the molecules that, once bound by an analyte, direct the cells to which they are attached to perform specific actions. Some receptors, dubbed agonists, spur the cell to do something; antagonists, on the other hand, are receptors that block further action. Either agonists or antagonists can be used to restore health to patients, depending on the affliction.
The propensity of an analyte and a receptor to bond with each other, thus triggering a reaction, is called affinity; the higher a receptor’s affinity for an analyte, the keener a pharmaceutical researcher’s interest.
In most biological research, whether a study of cellular mechanics or a search for novel drug candidates, fluorescence detection is used to find out whether biological reactions are taking place. The technique is well-defined, but it takes time to set up and requires costly fluorescent tags such as green fluorescent proteins, quantum dots or other photoemitting particles.
Unlike with fluorescence detection, the technique known as surface plasmon resonance (SPR) requires no extraneous photoemitter, or label. When struck by laser light, the electrons hovering above a metal surface begin to move in a coordinated path across said surface. The waves, collectively known as surface polaritons or surface plasmons, don’t tell much about the surface material – usually thin sheets of gold or silver – but they are remarkably sensitive to items in their path. Molecules that have been attached to a gold surface, for example, can be detected and characterized because they can emit photons when surface plasmons run into them.
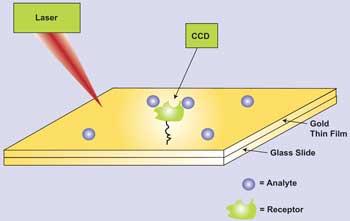
In surface plasmon resonance analysis, changes in molecules adhered to a gold substrate are highly detectable when laser light generates an evanescent wave of plasmons that move across the gold surface.
Agonists and antagonists hold mysteries besides whether they elicit a binding reaction that can be seen via fluorescence or through surface plasmon resonance. It has also been determined that various ligands can bring forth different biological reactions from cells, depending upon the intracellular pathway through which the receptor’s message translates, such as calcium-ion release or activation of cyclic adenosine monophosphate. The various forms that receptors can take also seem to play a role in cell activity.
In these recently found twists on receptor-analyte relationships, analytical techniques that require fluorescent labels can’t keep up. SPR-based methods, however, can.
In a typical SPR-based setup, ligands are immobilized onto a metal substrate, usually gold. Once in place, the ligands are then brought into contact with an analyte, and an optical reader shines laser light onto the substrate, then looks for signs of surface plasmon resonance, which indicates that binding of ligand (receptor) and analyte has occurred.
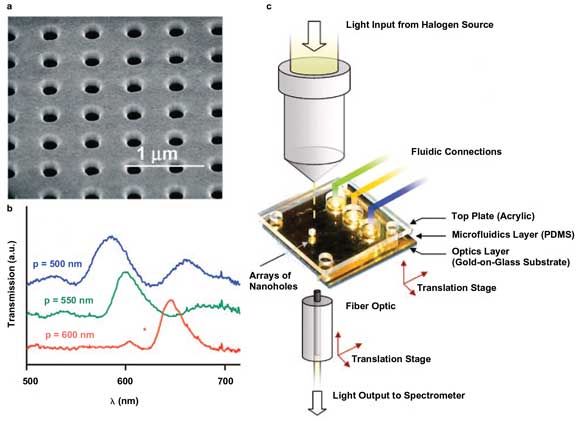
To increase the sensitivity of SPR analysis, punch some holes in the substrate. (a) A scanning electron micrograph shows a typical array of holes drilled via electron beam in a gold film. (b) Spectra from three arrays with different spacing values show how the extraordinary optical transmission effect increases with hole density. (c) The experimental setup is shown. Courtesy of Accounts of Chemical Research.
Because the industry must find possible new drug compounds fast, a multitude of ligands usually is arranged on a 96- or 384-well plate that fits into the SPR instrument. A single analyte then is introduced to the various ligands all at once, and the entire plate is scanned within a few minutes.
One of the most potentially lucrative areas of pharmaceutical research is, of course, cancer treatment. Inside most tumors, the enzyme telomerase helps perpetuate tumor growth and longevity, and many researchers are working on ways to stop telomerase’s functionality.
Véronique A. Leberre of the University of Toulouse and her colleagues at INRA, CNRS and the Curie Institute, all in France, wanted to find the effect of certain ligands on tumor cells.
Their proteins of interest, known as G4 ligands, are small molecules that bind to DNA. Telomerase activity is involved in cell immortalization and tumorigenesis, Leberre said, but it is inhibited by the stabilization of telomeric DNA when the G4 ligands force the normally loose ends of the DNA segment into the G-quadruplex structure – a four-stranded path of self-contained loops.
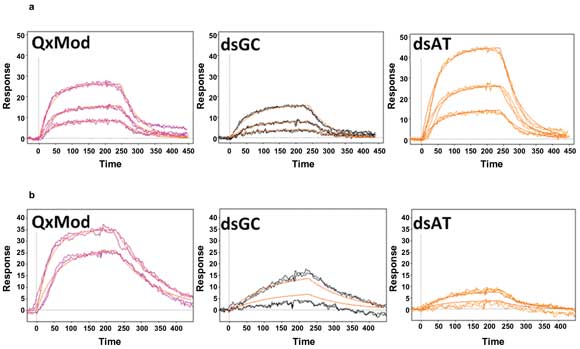
Examples of kinetic curves generated by SPR imaging of interactions between ligands Mn-TMPyP (a) and 360D (b) with various DNA structures.
While careful study of the effects of G4 ligands on tumor cells is promising, there are unfortunately also other molecules that the proteins may target, such as other quadruplex nucleic acids that may be present. To help clarify these effects, Leberre and her colleagues evaluated the usefulness of SPR imaging in discerning G4 ligands as they bind to immobilized G-quadruplex nucleic acids.
The investigators immobilized the test receptors to a 50-nm-thick gold substrate. For the G4 ligands, they used manganese porphyrin (Mn-TMPyP), terpyridine platinum complex (Pt-ctPy), phenanthroline derivative (PhenDC3) and 2,6-pyridine dicarboxamide derivative (360D).
Using an SPR imaging system made by Horiba Scientific-Genoptics, Leberre’s group passed the ligands over the immobilized receptors at a flow rate of 50 µl/min. They obtained kinetic information – the relative spectral peaks showing activity (kon) or inactivity (koff) – and derived an affinity constant (M—1) from kon/koff. Among their findings, they showed that the affinity of Mn-TMPyP for quadruplex forms was far higher than for single-stranded DNA fragments or for double-stranded forms abundant with guanine and cytosine (“GC-rich”). The affinity of Mn-TMPyP for double-stranded DNA rich in adenine and thymine (“AT-rich”) was also fairly high. Table 1 contains more affinity data.
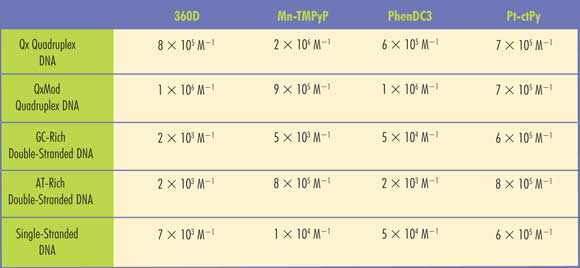
Table 1. SPR imaging provided the binding affinities of several known drugs (360D, Mn-TMPyP, PhenDC3 and Pt-ctPy) for various quadruplex DNA strands (Qx and QxMod), double-stranded DNA abundant in guanine and cytosine (dsGC) or in adenine and thymine (dsAT) that might be associated with various types of tumor cells. Adapted from Sensors and Actuators B, Vol. 157 (2011), pp. 304-309.
Ultimately, the higher the affinity, the more quadruplex DNA is present, and therefore a greater likelihood that the potential drug candidate will have antitumor properties.
The group found that SPR imaging let them quickly screen up to 1000 ligand-receptor interactions in one scan – and discriminate easily between the binding properties of all the G4 ligands they tested.
“SPR imaging allows discrimination between low and high specific interactions,” Leberre said. The investigators reported their work in the April 11, 2011, issue of Sensors and Actuators B: Chemical.
Next, Leberre’s group will try to increase the sensitivity of its approach and, outside of cancer studies, examine the interactions of DNA and proteins inside the bacteria Escherichia coli.
“In our lab, we are interested in the interaction between bacteria and glycoproteins,” she said. “So we try to fix glycoproteins on the prism surface and run the entire bacteria in the flow.”
The problem that must be addressed with the SPR imaging technique, she said, is that it is difficult to immobilize glycoproteins onto standard SPR surfaces because they have weak interactions with the bacteria. Therefore, the team must develop a new surface that can induce immobilization and yet still produce an adequate surface plasmon resonance effect.
Holes in theory
In addition to finding novel substrate materials, you can try poking holes into the ones you have at hand. That is the approach being taken by collaborating researchers at the University of Victoria and Simon Fraser University in Burnaby, both in British Columbia, Canada.
Instead of Mn-TMPyP and other drug candidates, the Canadian groups are exploring monoclonal antibodies, molecules that bind to antigens related not only to cancer but also to infectious and autoimmune diseases. Specifically, the hunt is on for cells that secrete antigens whenever disease has started to progress. Again, fluorescence labeling typically is used to locate monoclonal antibodies, but the labeling process slows the procedure, reducing the amount of screening tries in any given block of time.
To get around that, Reuven Gordon of the University of Victoria and his colleagues are developing a system that exploits a different sensing-friendly phenomenon, extraordinary optical transmission (EOT).
As Reuven’s group explains it, EOT is a result of surface plasmon excitation. If you drill an array of tiny holes into a gold or silver substrate and then immobilize a target molecule to the surface, you are set up for a strange reaction. When you excite the surface with a laser beam, the plasmons generated travel across the surface until they encounter one of the immobilized molecules.
Counterintuitively, perhaps, the light generated by this encounter not only passes through a nearby hole, but also comes out the other side at a rate higher than the input. That is, the substrate appears more transparent by up to three orders of magnitude. The extent of the effect changes with the number of holes and their distance apart, as well as with the choice of materials – gold and silver remain best, however.
For its work with monoclonal antibodies, Reuven’s group used gold substrates with targets immobilized to them. Via an FEI Co. ion-beam instrument, the substrates were perforated with 200-µm holes spaced 3.77 mm apart. More tightly spaced holes would provide better EOT performance, but the larger spacing helps reduce diffusion of the cellular material.
Over the top of each pierced surface, the researchers laid a film of poly(dimethyl siloxane) (PDMS) that included divots, or wells, that trapped the cells they wished to test against the targets. Running their tests with a spectrometer made by Burnaby-based Photon Control to collect spectra, the group detected binding of secreted antibodies to the immobilized targets via shifts in the EOT spectra.
The results produced by Reuven’s group thus far are proving to be similar to those gained with fluorescence labeling, but still lacking a bit in sensitivity. The team continues to work on increasing both selectivity and sensitivity of the system, primarily by reducing the density of the cells collected in each PDMS trap.