Volker Pfeufer, Coherent, Inc.
OPSL technology has powered a paradigm shift in orange lasers for use in the life sciences by enabling wavelength and power to be customized to exactly match specific applications, instead of the other way around.
For many years, applications in life sciences were optimized to match the available laser output characteristics – particularly wavelength – that most nearly met their needs. This situation completely changed with the advent of optically pumped semiconductor laser (OPSL) technology, an all-solid-state laser architecture that is freely scalable in terms of wavelength and output power. As a result, lasers can now be fit to specific applications, instead of vice versa.
The use of multiple fluorophores significantly extends the utility and enhances the power of many techniques based on laser-excited fluorescence. For example, it enables multiple structure types to be simultaneously imaged in microscopy, multiple cell types to be individually counted in flow cytometry, and multiple expression pathways to be screened against potential drug candidates.
Extending flow cytometry
Simple, reliable lasers in the orange region of the visible spectrum enable fluorescence applications such as flow cytometry, a cell analyzing and counting technique that relies on laser-excited fluorescence. Cells pass singly through the interaction zone, where one or more laser beams excite fluorescent probes: fluorescent dyes covalently bonded to ligands that preferentially attach to specific antigens on the surface (or sometimes structures within) of the cells being counted.
The resultant fluorescence from each cell is separated into several wavelength bands by dichroic beamsplitters, prior to detection using photomultiplier tubes, or PMTs (Figure 1). Two or more broadband PMT detectors are also used to measure forward scatter for information on cell size and side scatter for information on cell complexity. Cells are counted according to the relative intensity in the various detectors – i.e., via their fluorescence and scatter signatures.
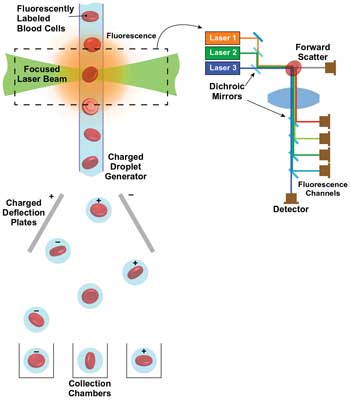
Figure 1. In a flow cytometer, cells are counted and/or sorted according to their scatter and fluorescence signatures. Images courtesy of Coherent Inc.
Users of both clinical and research instruments have a need to count and/or sort for an ever-greater number of parameters, which in turn requires a larger number of fluorophores. The problem is that emission from a single fluorophore type is usually broad and will cover more than one of the instrument’s detection bands. Ultimately, the limit on the number of cell types is determined by the overlap of the emission spectra of the fluorophores used and the working bandwidth of the instrument. Together, these factors determine how many fluorophores can be uniquely identified.
There are two ways that instrument designers and users can increase the number of cell types that can be counted in a single instrument run. One approach is to use fluorophores with narrower emission bands – i.e., less spectral overlap – so that more fluorophores can be uniquely identified by the instrument. This is enabled by the use of quantum dots (QDs): fluorescent nanocrystals in the 10- to 20-nm size range. The emission spectra of these QDs are quite narrow depending on their size, and quantum dots are now available from the blue through the deep red; they also can be bound to a variety of ligands (probes). At present, the use of QDs is limited, but it certainly will increase as the technology for linking them to different probes improves and the price decreases.
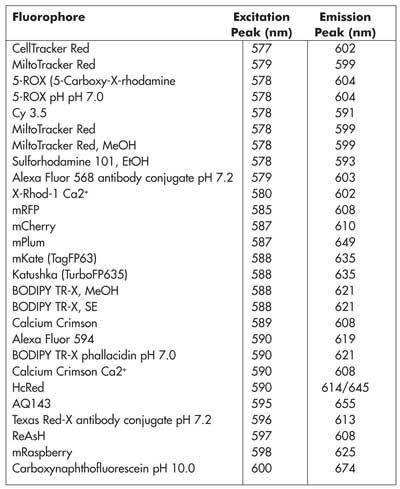
The other way to increase the multiplex detection capabilities of flow cytometry is to increase the total spectral bandwidth of the instrument. This can be done by addressing the wide gap in the arsenal of available excitation wavelengths, particularly those produced by compact laser technologies that can be economically integrated within an instrument such as a flow cytometer. Specifically, even a quick look at the Photonics Spectrum Reference Wall Chart from Laurin Publishing reveals that the orange region of the visible spectrum is virtually empty of such options, all the way from 561 to 630 nm. However, there is a growing list of useful fluorophores in this spectral region, including Alexa 594, mPlum, mCherry and BODIPY TR-X, to name a few (see Table 1).
 Reaching new wavelengths
Reaching new wavelengths
The optically pumped semiconductor laser is a relatively new type of laser that is wavelength-scalable and can be optimized for any target wavelength over most of the visible spectrum. And because OPSL technology is also power-scalable, it can be the basis of lasers for diverse applications such as medical diagnosis and therapy, light shows and materials processing.
In an OPSL, the gain medium is a large-area vertical-cavity surface-emitting laser (VCSEL) chip that is optically pumped by one or more diodes or diode arrays (Figure 2). The output wavelength is determined by the stoichiometry and dimensions of the quantum wells in the VCSEL structure, and thus can be optimized for nearly any wavelength in the near-infrared. Very efficient intracavity doubling then enables the final output to be set anywhere throughout the visible spectrum. And frequency-tripled ultraviolet OPSLs provide true-CW output for applications such as cell sorting at 355 nm.
A significant advantage of OPSL technology is that a VCSEL has a much broader absorption bandwidth than crystalline materials, such as Nd:YAG or Nd:YVO4. This substantially relaxes the pump diode wavelength tolerance, thus reducing diode cost. It also eliminates the requirement for tight temperature control, lowering system complexity and cost. These are important benefits for life sciences applications, particularly in original equipment manufacturer instruments.
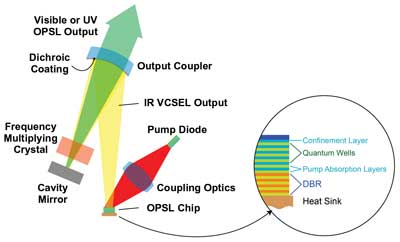
Figure 2. Schematic illustration of the main components of an optically pumped semiconductor laser (OPSL). DBR = distributed Bragg reflector.
In an OPSL, power scaling is enabled simply by increasing the output power of the pump diode or by increasing the number of these diodes. As a result, OPSLs covering a wide range of output powers and wavelengths can all be packaged in the same compact platform, which is a tremendous advantage for designers of instruments that require multiple wavelengths or for those upgrading existing instruments to new wavelengths. And, because rear-surface cooling of the thin gain chip ensures that there is almost no thermal lensing in OPSLs, they can deliver excellent TEM00 beam quality, a circular output beam profile (Figure 3) and superior beam-pointing stability.
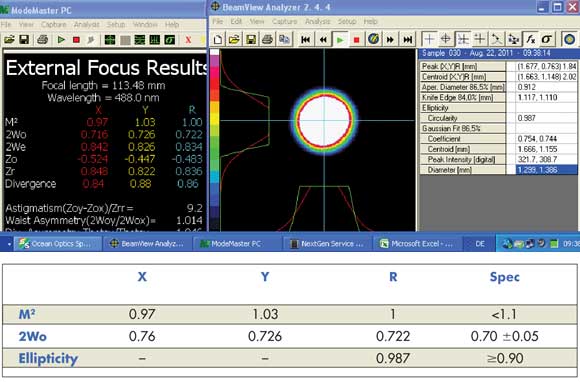
Figure 3. OPSL technology is characterized by a circular beam profile that can easily deliver a TEM00 output mode.
OPSLs also provide excellent power stability and low noise because the near-zero excited-state lifetime of the semiconductor gain material eliminates the potential of “green noise.” This is the chaotic output noise caused by intracavity doubling and frequency mixing in most CW solid-state and gas lasers. Green noise in those lasers can be rigorously eliminated only by resorting to the complexity and cost of stabilized, single-longitudinal-mode operation. But a simple (low cost) OPSL cavity operating on multiple longitudinal modes can now deliver superior noise characteristics (Figure 4).
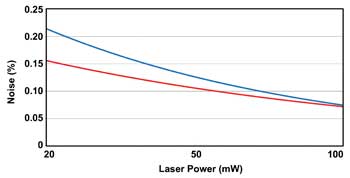
Figure 4. OPSLs such as the new Sapphire 588 from Coherent offer very low output noise because intracavity doubling does not produce the chaotic “green noise” that plagues many other solid-state laser types.
In addition, OPSL technology is simpler to assemble than earlier laser technologies and does not require the manual adjustment typically required when building diode-pumped solid-state (DPSS) and other lasers. Thus, OPSLs lend themselves to automated production methods with their related economies of scale and high unit-to-unit consistency. And, lastly, because OPSL technology uses all-solid-state components and PermAlign rigid mounts for the cavity optics, OPSLs feature low service/repair returns.
Orange OPSL at 588 nm
Based on dialogues among engineers, end users and instrument builders, the optimum orange wavelength for most applications appears to be 588 nm. This is ideal for exciting the new mfruit series of red luorescent proteins – e.g., mPlum, mKate (TagFP63), mCherry and mRFP, as well as popular labels such as Texas Red, Alexa Fluor 594, Katushka (TurboFP635), MitoTracker Red and CellTracker Red. As a result, Coherent launched its series of 588-nm Sapphire lasers at the SPIE BiOS 2012 conference and exhibition in San Francisco. And, because of the ease of power scaling of their OPSL technology, these lasers are offered with a choice of output power ranging from 20 to 100 mW.
Of course, several other types of orange lasers have been developed in the past couple of years. It’s useful to briefly compare the new 588-nm OPSLs with these other lasers as well as with the venerable orange HeNe. Starting with the HeNe at 594 nm, there are two reasons that this laser has never been widely adopted in the life sciences market, even though it delivers excellent beam quality and reasonably low noise. Most importantly, the low gain of this medium (especially at 594 nm) makes it difficult to obtain more than 2 mW without resorting to an unwieldy long laser head. In addition, most life sciences applications have now switched to solid-state lasers because they offer much higher electrical efficiency, superior reliability and smaller packaging than gas lasers.
Fiber lasers are now available at 580 and 592.5 nm, with powers up to 2 W. While these are adequate for some illumination-type applications, they are not well suited to demanding applications in life sciences because their output is highly asymmetric; more importantly, they produce high output noise (up to 2 percent rms), which is much higher than the 0.25 percent of a typical 588-nm OPSL (Figure 4). In addition, they are surprisingly bulky compared with OPSLs and other all-solid-state lasers.
DPSS lasers have also been developed around 594 nm based on intracavity frequency doubling and intracavity frequency mixing of the near-IR output of a Nd:YVO4 laser crystal. While all-solid-state, this laser is much more complex than an OPSL. Specifically, frequency mixing requires the generation of two laser beams at different wavelengths at sufficient power, and with high beam quality and matched beam parameters. This adds complexity and cost to the DPSS laser and negatively affects its reliability and stability.
In addition to the challenge of phase matching two intracavity nonlinear crystals (for doubling and mixing), both of these nonlinear processes cause green noise. So, avoiding multiplicative noise and reaching a noise level that is acceptable for cell counting require operating and stabilizing the laser on a single longitudinal mode. However, this introduces yet more system complexity and cost. This is in marked contrast to the robust and simple OPSL cavity design.
Meet the author
Volker Pfeufer is product line manager at Coherent Inc. in Lübeck, Germany; email: [email protected].