Precisely applied heat from the touch of a laser not only can hide blemishes, but can also save lives.
For many, laser surgery brings to mind highly refined incisions made by coherent light pulses. Cutting, however, is not the only action a laser can perform: Sometimes, all you need is a little heat.
Aiming a properly chosen laser onto a blood vessel or other tissue section adds enough thermal energy that the tissue’s properties alter. When heat is applied to a blood vessel, the vein or artery deforms, or even withers away if enough energy is used.
The ability of a laser to attain the right amount of photothermal effect on veins and arteries is controlled by the absorptive capacity of the molecules that comprise the tissue. The constituents of the blood within veins and arteries are important targets of research behind photocoagulation improvement.
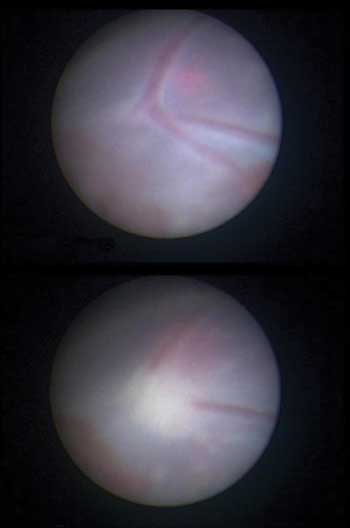
Top: Blood vessels cross the vascular equator in a case of twin-twin transfusion syndrome. Bottom: The same vessels are shown after laser photocoagulation. Reused with permission of Early Human Development.
Dr. Iris K. Rubin of the Children’s National Medical Center in Washington and her colleagues at the Wellman Center for Photomedicine in Boston became determined to find the best way to treat port-wine stains – birthmarks experienced by about 3 in 1000 infants caused by malformations of blood vessels just under the skin.
Named for the color and spread of the abnormality’s appearance, the effect is caused by unusually dilated capillaries. Port-wine stains can be located anywhere on the body, but up to two-thirds of cases are found on the head and neck. For many years, the condition has been treated by covering the area with tattoos, or through surgery or freezing, but laser therapy has proved to be the most efficacious method of reducing the appearance of these marks.
Laser-based photocoagulation of port-wine stains has become routine, even though it requires multiple treatment sessions and completely clears the birthmark only a minority of the time.
“Photocoagulation of malformed blood vessels using laser pulses based on the theory of selective photothermolysis is the current treatment of choice because blood vessels can be damaged while preventing injury to the normal overlying epidermis,” said Wangcun Jia of the Beckman Laser Institute and Medical Clinic in Irvine, Calif.
“The most important factor for causing photocoagulation of veins is reaching an adequate temperature in the vessel to result in closure,” Rubin added. “The temperature must be adequate, though not too high as to cause nonspecific thermal damage to surrounding structures.”
Adjusting the laser to the task of photo-thermolysis – the breaking down of tissue with a precise wavelength and pulse length – is the key objective of the clinician performing the treatment. Choosing correctly means knowing your molecular target.
Oxyhemoglobin (HbO2) is the traditional target for vascular lesions, Rubin said, because it is specific to blood, allowing selective photothermolysis of the blood vessels.
By selecting the right wavelength and pulse duration, Jia said, the laser beam is absorbed preferentially by the hemoglobin present in the blood vessel. The buildup of heat causes thermal damage and thrombosis.
“The laser wavelength is chosen to maximize the difference in light absorption between blood and other tissues such as epidermis and dermis,” Jia said. The pulse duration is selected so that resulting heat is confined to the targeted blood vessel during the pulse.
To settle one part of the equation, Rubin and her colleagues used a series of lasers to test the effects of several popular wavelengths used for port-wine stain treatment. They tested a 694-nm ruby laser, a 755-nm alexandrite laser and a 1064-nm Nd:YAG laser, and pulsed-dye lasers set to 585, 590, 595, 600 and 633 nm. The ruby laser was made by Palomar Medical Products of Burlington, Mass., the others, by Candela Inc. of Wayland, Mass.
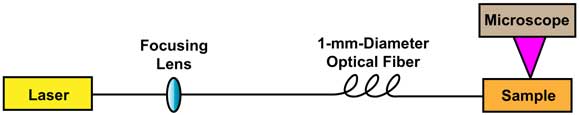
A schematic representation of a benchtop laser system used to assess which wavelengths are best suited for photocoagulation.
In general, pulsed-dye lasers are known for introducing a low risk of scarring after the port-wine stain treatment. However, they also have less efficacy because they cannot penetrate deeply enough into tissue to reach every abnormal blood vessel – no farther than about 2 mm.
The investigators tested each laser within a common system using venous or arterial blood at various fluences until photocoagulation occurred. They reported their work in the February issue of Lasers in Surgery and Medicine.
The group found that energy from the dye lasers emitting in the 630- to 780-nm range was absorbed best by venous blood samples, indicating that they are the best candidates for port-wine stain reduction. The 1064-nm beam emitted by the Nd:YAG laser, on the other hand, was absorbed preferentially by arterial blood, which is not optimal for this treatment; lasers emitting below 630 nm did not have strong efficacy.
Being able to selectively target veins rather than arteries, the researchers say, enables better port-wine stain reduction while sparing healthy arteries that aren’t responsible for the condition.
The next step for the group, Rubin said, is an animal study to test the concept that vein-selective photothermolysis is possible in vivo as well as ex vivo, on blood samples; this stage will be followed by a human clinical trial.
Because standard laser photocoagulation of port-wine stain blood vessels is performed with single pulses, Jia’s group looked for improvement on that part of the technique. In the same issue of Lasers in Surgery and Medicine, Jia and his colleagues describe their process of determining whether multiple laser pulses could be more efficacious than single pulses.
The group – which included members from the University of California, Irvine (of which the Beckman Institute also is part), and from the Jozef Stefan Institute and Fotona d.d., both of Ljubljana, Slovenia – used two to five 1-ms pulses from a 532-nm Nd:YAG laser supplied by Fotona on the blood vessels of living animals.
The investigators varied the fluence of the pulses from 1.4 to 7 J/cm2 to test both the number of pulses and total applied energy on the overall photocoagulation effect.
Among their findings, they reported, single pulses required 7 J/cm2, whereas multiple pulses could require as little as 2 J/cm2. The reduction of thermal energy is important, because heat accumulates within the blood vessel. Too much heat buildup could result in adjoining tissues becoming injured.
“[The multiple laser pulse] approach distributes the laser energy into several pulses of lower energy, which avoids the inherent risk of damaging the epidermis due to nonspecific light absorption by epidermal melanin, especially for patients of darker skin phototypes,” Jia said.
The researchers are now working on translating their results into a multiple-pulse technique suitable for clinical practice. Technological upgrades to increase the energy output of the 532-nm laser or the repetition rate of the pulsed-dye laser are part of their plans to facilitate the translation from laboratory to clinic, Jia said.
Beyond birthmarks
The transport of blood and nutrients is of vital importance to the health of all fetuses, but can be especially problematic in twins who share the same placenta. In up to 20 percent of monochorionic twin pregnancies – where two or more fetuses inhabit the same chorionic membrane and possibly the same amniotic sac – the blood flow is unbalanced.
One of the most troubling complications, this imbalance is called twin-twin transfusion syndrome (TTTS), and it occurs after the formation of reconnecting arteries and veins, called anastomoses. An anastomosis, which looks like the lanes of a two-way road diverging around a traffic island, connects the umbilical cords of the twins, making their circulations a single entity.
In TTTS, one of the twins becomes an unwitting donor, with all the available nutrient-carrying blood diverted to the second fetus. This leaves the donor twin with lower blood volume and, in cases where the twins have separate amniotic sacs, lower amniotic fluid volumes, leading to anemia, malnourishment, growth limitation and other serious problems. The recipient twin receives too much blood (and amniotic fluid), which strains the fetus’s heart to the point where cardiac failure becomes likely. Also, the buildup of amniotic fluid in the second twin’s sac can cause it to burst, forcing premature labor.
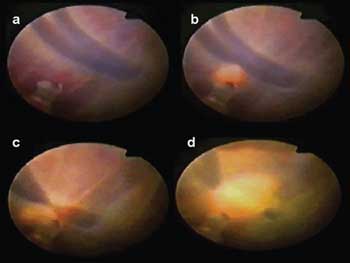
(a,b) A fetoscopic view shows inter-twin vascular anastomoses (mergers of veins or arteries) present during twin-twin transfusion syndrome. (c,d) The same regions are shown after laser photocoagulation of the anastomoses. Reused with permission of Clinics.
Untreated, 80 to 100 percent of both donor and recipient twins die before or just after birth. Even if only one dies prenatally, the trauma often seals the fate of the other.
There are a few treatments available to forestall danger, including removal of excess amniotic fluid via syringe. However, the latter therapy does not solve the underlying condition, especially if anastomoses are involved, and other treatments are more invasive. The current best option for treating TTTS is fetoscopic laser photocoagulation, in which a beam from an Nd:YAG or diode laser is sent through a fiber optic cable (via an endoscopic tube) to the sites on the placenta that have anastomoses. The surgeon uses the beam to ablate select tissue, and to photocoagulate the vessels left behind. This procedure leaves two functioning, separate blood vessels and increases the survival rate for both twins to 40 to 60 percent. The rate for one twin’s survival climbs to 70 to 90 percent.