An occasionally overlooked technology leads to major advances in the field
Fiber optics might just be the Rodney Dangerfield of the photonics world: Despite being a workhorse technology that has contributed to countless studies over the years, it doesn’t always get the respect it deserves. But, in fact, it is integral in a broad spectrum of imaging and monitoring techniques.
Here, we look at the importance of fiber optics in the development and application of two techniques that have made – and continue to make – significant contributions in the field of neuroscience.
Untangling BOLD fMRI signal
Blood oxygen level-dependent (BOLD) functional MRI (fMRI) is widely used in neuroscience studies, but it is an indirect and imprecise measure of neural activity; the relationship between the changes in blood flow and oxygenation it measures and the neural activity that spurs those changes is still not entirely clear, so researchers don’t necessarily know how accurately the BOLD signal reflects what is happening at the neural level.

Hoping to better understand the relationship between the blood oxygen level-dependent (BOLD) functional MRI (fMRI) signal and the underlying neural activity, researchers performed multimodal imaging of the rat somatosensory cortex with BOLD fMRI recordings and fiber optic recordings of fluorescent calcium indicator signals. Shown here is the MR scanner used in the study. The optical setup was kept outside the electromagnetically shielded MR scanner area and connected to the probe with an optical fiber more than 7 m in length. Courtesy of Kristina Schulz, Group Helmchen, University of Zurich.
One way to tease out this relationship is to measure the BOLD signal and the underlying neural activity simultaneously, the latter with conventional electrophysiological recordings. “But this technique is really challenging because the magnetic field of the MR scanner heavily interferes with the electrical signal and vice versa,” said Kristina Schulz, a researcher with the Brain Research Institute at the University of Zurich.
In a recent Nature Methods paper, Schulz and colleagues from Fritjof Helmchen’s group and Markus Rudin’s group at the university reported a study in which they combined fMRI recordings of the BOLD signal with fiber optic recordings of fluorescent calcium indicator signals to probe the relationship between the two in a rat somatosensory cortex. Calcium indicators offer a suitable alternative to electrophysiological recordings, they wrote, because they can measure several aspects of cortical dynamics – including neuronal network activations and glial signaling – in vivo, using single-photon excitation delivered through a conventional optical fiber.
Was there anything particularly complicated – anything fancy – about the use of fiber optics in the study? “It was actually pretty straightforward,” Schulz said, “and I guess it could have been done even earlier.” She noted that the novelty of the study, at least in the early stages, lay simply in the idea to combine the two measures.
Using this multimodal approach – with BOLD fMRI and calcium recordings – the investigators found that they could measure signals that have not been accessible with electrophysiological recordings. They could show, for example, how astrocytic activity relates to the BOLD signal.
“By using astrocyte-specific calcium indicator labeling, we discovered that our stimulation paradigm in some cases induced strong astrocytic activation,” Schulz said. “We could further show a relation of these astrocytic calcium waves to prolonged and increased BOLD signals. This relation could only be uncovered by simultaneous measurements.”
The unsung hero of optogenetics
Fiber optic technology is leaving its fingerprint everywhere, it seems. In recent years, it has played an integral role in another cutting-edge neuroscience technique – optogenetics – by enabling delivery of light to deep regions of the brain that might not be accessible otherwise.
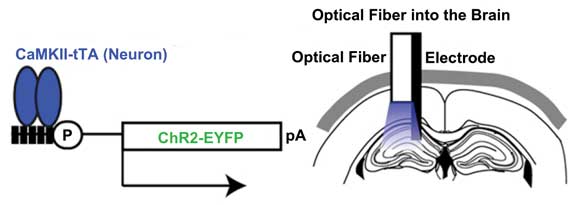
Fiber optics plays an integral role in optogenetics. Here, an example of in vivo manipulation of neurons using fiber optics, as reported by Karl Deisseroth’s group at Stanford University. ChR2(C 128S)-EYFP was expressed in hippocampal CA1 neurons using the aCaMKII-tTa line. Blue light pulses transmitted through an optical fiber inserted into the brain illuminated the dorsal left hippocampus, and neuronal activity was measured. Courtesy of Cell Reports.
Optogenetics seeks to elucidate the role of neural circuits in certain behaviors by silencing or driving particular neural activity patterns. Stanford University’s Karl Deisseroth, a pioneer of the technique, recalls how important fiber optics was to its development. When he and colleagues reported an in vitro, proof-of-principle study in Nature Neuroscience in 2005, he said, other researchers questioned how the method would work in living animals – light would scatter before it reached the targeted tissue, they argued.
“That was a good criticism,” Deisseroth continued, “and it was a couple of years before we figured it out.”
The researchers tried a variety of approaches. They tried surface illumination; they tried opposing LEDs; they considered strategies to make cells generate their own light. Other groups also sought to tackle the problem. But the basic question of how to deliver light to deep regions of the brain remained unanswered.
Finally, the Stanford group coupled laser diodes to optical fibers and, in 2007, reported the first fiber optics-based neurointerface. The fibers were off the shelf, Deisseroth said, nothing fancy. The method worked, though, and today it is a fundamental part of optogenetics studies.
And there is no shortage of these. In 2010, Science hailed optogenetics as one of the breakthroughs of the decade, noting that, within a few short years, “thousands of scientists in hundreds of labs” were using the technique to explore how the brain works. The questions it can help to answer are myriad, ranging from how cocaine use affects the brain’s reward pathway to a universe of questions in the area of psychiatry. And fiber optics is at the center of it all.
Ed Boyden, a researcher in the MIT Media Lab and another pioneer in the field – he was first author on the 2005 Nature Neuroscience paper and is recognized with Deisseroth as a co-inventor of optogenetics – emphasizes the importance of fiber optics to the success of the technique.
“For the in vivo, live-brain case,” he said, “it would have been much, much harder without fiber optics or waveguides. (In vitro, of course, you can just use a microscope to illuminate the cells through the objective lens.)
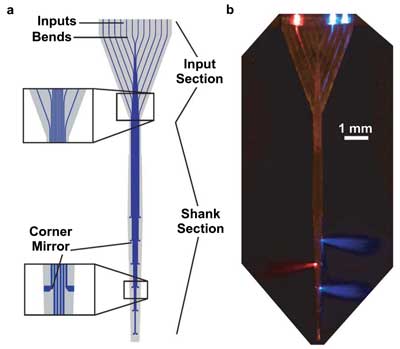
Researchers have demonstrated a 360-µm-wide array of 12 multimode waveguides for delivery of light to distributed targets in the brain. A schematic of the multiwaveguide probe is shown on the left. On the right, a photomicrograph of the probe immersed in a scattering medium, with light coupled into three of the 12 waveguides. Courtesy of Optics Letters; see Zorzos et al. Opt Lett. 2010 Dec 15; 35(24)4133-5.
“Basically, you have two choices for illuminating a deep target: implant a light source in the brain or use a waveguide or fiber to serve as a conduit to get light into the brain. Implanting a light source into the brain is tricky because it can rapidly result in prohibitive heat.” This essentially leaves use of a waveguide or fiber as the only viable option.
As successful as fiber optic technology has proved already, however, scientists are looking at how they can use it to advance their research still further.
At Stanford, Deisseroth’s group is looking at the possibility of having extremely dense arrays of independently addressable sources and using the fiber bundle as a collimator coupled to image information on either end.
Also, Boyden and colleagues have demonstrated a multiwaveguide probe that offers optical control over sets of targets in the brain by independently delivering light to targets along the probe axis. The probe consists of many waveguides running in parallel to one another and thus enables systematic manipulation of neural activity in different parts of brain circuits, with minimal damage to the brain.
The researchers are now applying the probe to map such circuits; for example, using it to stimulate a number of individual sites in the brain in rapid succession to see which most strongly connect to a downstream target. This could help to determine whether the functional connections between different parts of the brain change over time.
Also, Boyden said, by stimulating multiple sites in temporal registration to one another, they can “play back” activity to the brain. “One of my hopes is that we can use this technology to test detailed theories of the neural codes utilized by the brain. Say that we could stimulate a set of neurons with a realistic pattern of activity: We could try to figure out whether that realistic pattern of activity actually drives a particular perception, action or behavior.”
New probes, new data
MIT’s Ed Boyden and colleagues are working to expand their multiwaveguide approach into three dimensions, designing probes that enable light delivery to thousands of individual sites in the brain in full 3-D patterns.

Researchers have developed and are beginning to perform implants of 3-D multiwaveguide probes. Courtesy of Ed Boyden.
“In this way, we can try to simulate complex patterns of stimulating the brain, important for testing theories of how the brain encodes for information,” Boyden said.