Yong Huang and Jin U. Kang, Johns Hopkins University; and Adjani Peralta, Princeton University
Researchers share their demonstrations of real-time monitoring of 6.1-µm quantum cascade laser-based ablation and coagulation.
Thanks to advances in laser therapy, lasers are now irreplaceable tools in modern medicine.1 Many laser systems are becoming standard instruments for surgical procedures; examples include the UV ArF excimer laser for ablation of corneal stroma during laser refractive surgery, the green argon laser for photocoagulation for retinal detachment therapy and diabetic retinopathy,2 and the Nd:YAG laser in breast tumor cutting.3
Lasers have enormous potential as high-precision surgical instruments because laser radiation can be focused into a small submicron spot selectively and strongly absorbed by tissues. When a train of pulses of laser light is focused on biological tissue, the radiation will be absorbed by the target tissue, which results in a significant local temperature increase. Coagulation of soft tissue will occur when the temperature rises above 60 ºC, and ablation will occur above ~130 ºC.
Laser power and exposure time are important factors for proper laser therapy. If sufficient energy is delivered to a target tissue site within a short period of time – less than the thermal relaxation time – the desired therapeutic results can be achieved with minimal collateral damage. Of course, precise removal of tissue with minimal thermal damage to the surrounding regions requires high absorption of laser radiation. Most soft tissues have high water content (up to 65 percent) and protein (up to 35 percent). Therefore, the water and protein absorption coefficients are two important factors that determine which wavelength to use for a specific task.
The high IR absorption coefficients of biotissues in the infrared range makes the IR laser a good candidate for high-precision biotissue ablation. In addition, in the mid-infrared range, wavelengths of 6.1 and 6.45 µm fall within the absorption peaks of amide protein groups, amide-I and amide-II, respectively.4 The absorption peak at 6.1 µm also coincides with the deformation mode of water. These properties can produce improved therapeutic results compared to purely water absorption-based therapy. However, until recently there had not been a practical IR source in the 6-µm range.
Quantum cascade lasers (QCLs) are compact and highly efficient mid-IR semiconductor lasers.5 Recent years have seen significant improvements in their power and stability.6 They are also low-cost and tunable, making them a great alternative to tunable free-electron lasers and optical parametric oscillators.
To precisely guide laser therapy, careful monitoring of the tissue is desired. Therefore, various image-based monitoring systems including magnetic resonance imaging, computed tomography and ultrasound have been demonstrated. However, limitations such as low image resolution, slow imaging speed and high complexity prevent effective real-time therapy guidance.7 Alternatively, optical coherence tomography providing 3-D imaging of the tissue in real time can potentially act as a real-time monitoring system that helps avoid unnecessary damage to surrounding tissues.
OCT is an established noninvasive optical imaging technology that can provide high-speed, high-resolution, 3-D images of biological samples. Since the early 1990s, it has been widely used for diagnosis, therapy monitoring and ranging.8 In this study, we performed QCL-based laser thermal therapy with real-time feedback from an OCT system to monitor the laser treatment. In particular, we performed tissue ablation and coagulation using a 6.12-µm QCL with real-time OCT monitoring.
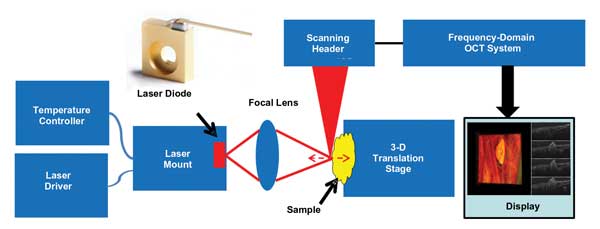
Figure 1. This study was a demonstration of real-time OCT monitoring of 6.1-µm QCL-based lipid phantom tissue ablation, chicken egg yolk coagulation, and tissue and blood vessel coagulation in a chicken embryo. Here is the system configuration used for this work. Images courtesy of Yong Huang, Jin U. Kang and Adjani Peralta.
Methods
The study was performed using a C-mount QCL from AdTech Optics. We used one CaF2 mid-IR lens (LB5774 from Thorlabs) with a 25.4-mm focal length to direct the light onto the target sample. When operated at a pulse mode of T = 15 ºC, the laser wavelength is centered at 6.12 µm with a bandwidth of 0.2 µm. The laser has a threshold current of 0.4 A and a peak output power of 791.7 mW at 2.1 A/15.3 V. To drive the laser, we used a 4303-QCW laser source from Arroyo Instruments. It can provide a maximum current of 2 A with a pulse length limit of 0.1 ms. We also used a 242 thermoelectrically cooled (TEC) C-mount and, to cool the laser, a 5305 TEC controller from Arroyo Instruments. The laser mount was kept at 18 ºC, which was above the local dew point.
The sample under test was put on a 3-D translation stage for accurate positioning. The OCT monitoring system was an in-house-built spectral-domain OCT system with lateral resolution of 15 µm; its effective axial resolution is 3.6 µm in air and 2.8 µm in water. The OCT system operates at 70,000 A-scans per second and can be switched between B-mode and C-mode; each B-frame consists of 1000 A-scans, and 256 B-frames form a volume. Volume acquisition time is 3.7 s. Customized software using a graphics processing unit accelerated real-time data processing and visualization.9 This immediate 3-D image feedback of the targeted area enables responsive evaluation of the therapy outcome and laser dosimetry.
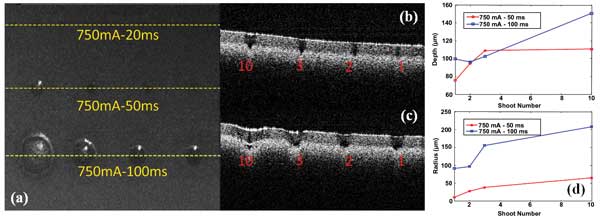
Figure 2. Lipid phantom ablation (a) is shown in an en face project image of the ablated area (H x W: 2 x 2.67 mm); a cross-sectional image of line 750 mA/50 ms (b); a cross-sectional image of line 750 mA/100 ms (c); and the depth and radius measurements of ablated craters under different laser conditions (d).
Results and discussion
We conducted a series of basic laser thermal therapy based on various phantoms to evaluate the performance of 6.1-µm QCL-based laser therapy. Driving current, pulse length and number of pulses applied to the target tissue are parameters that can be tuned to achieve precise tissue ablation.
We tested the lipid phantom ablation under different pulse lengths and pulse numbers. The laser driving current was set at 750 mA, which corresponds to a 163-mW peak optical power. The pulse length was set at three different values: 20, 50 and 100 ms. The number of pulses applied was set at one, two, three and 10.
Figure 2a shows en face projection of an OCT-acquired volume image data set, which is similar to a microscope image after a flat lipid phantom has been exposed to various numbers of laser pulses. From the en face image, we can clearly see craters of different radii induced by laser pulses. However, for a pulse length of 20 ms, we didn’t observe any craters, perhaps because the energy density is below the threshold. Figures 2b and 2c show the cross-sectional depth profile of the dashed lines on Figure 2a, corresponding to the pulse lengths of 50 and 100 ms, respectively. The number of pulses applied are marked in red. From Figures 2a, 2b and 2c we can quantify the depth and radius of the craters, shown in Figure 2d. The trend of increased depth and radius with increased pulse length and number is apparent. Meanwhile, there is a saturation effect with the number of pulses
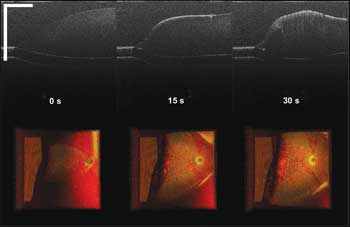
Figure 3. Chicken egg yolk coagulation via laser radiation (scale bar: 500 µm; color image size H x W: 1 x 2 mm), seen at different time points within 30 s.
Localized tissue coagulation/ablation forms the basis of tumor treatment, providing a minimally invasive treatment technique: Due to the associated coagulation of blood vessels, hemorrhaging can be prevented. This is of particular importance because tumors are usually highly vascularized.1 In neurosurgery, it is also important to prevent injury to adjacent healthy tissue and sensitive structures. As a proof-of-concept demonstration, we performed OCT-monitored, QCL-based chicken egg yolk coagulation, soft-tissue and blood vessel coagulation in a chicken embryo model. The laser was driven at a 1.3-A current, which corresponds to a 440-mW peak optical power with a 30-ms pulse length and 20-Hz repetition rate.
Figure 3 shows screen-captured images of the chicken egg yolk under laser radiation at different time points within 30 s. In the top row of Figure 3 are the B-mode cross-sectional images of the central line of the top-view 3-D volume, rendering images seen in the bottom row. In the B-mode images, we can identify the gradual formation of the whitening part from the yolk surface to the inside. Whitening indicates yolk coagulation, as the coagulated tissue gets dense, thereby increasing the scattering of signal light. In the top-view 3-D volume rendering images, we can clearly see the increased volume of coagulated tissue with continuous laser radiation.
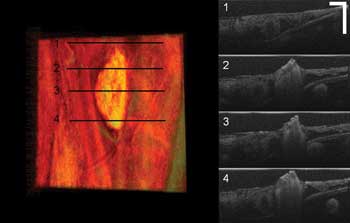
Figure 4. Soft-tissue coagulation in a chicken embryo surrounded by healthy tissue (scale bar: 250 µm; color image size H x W: 1 x 2 mm). On the left is the top-view volume rendering image; at right are four B-mode cross-sectional images corresponding to the line positions marked at left.
We further evaluated soft-tissue and blood vessel coagulation on the chicken embryo model.
The chicken embryo’s chorioallantoic membrane is a well-established model for studying microvasculature and has been used extensively to investigate the effects of vasoactive drugs, optical and thermal processes in blood vessels, and retina simulation.10
Figure 4 shows a coagulated soft-tissue lesion surrounded by healthy tissue. The left color image is the top-view volume rendering image; at right are four B-mode cross-sectional images corresponding to the line positions marked on the left. From the OCT top-view volume rendering image, we can clearly see the morphology and size of the lesion. By combining it with the B-mode cross-sectional images, we can determine how deep the coagulation goes. Real-time imaging feedback can help a surgeon determine whether further laser radiation is necessary, which helps prevent unnecessary damage to healthy tissues.
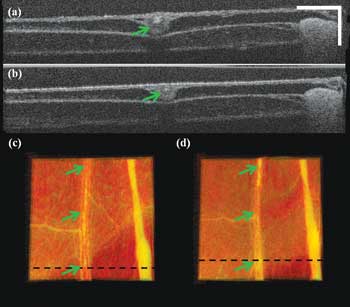
Figure 5. The blood vessel coagulation cross-sectional image (a) corresponds to the dashed line in (c); the cross-sectional image (b) corresponds to dashed line in (d). The top-view volume rendering image of the area of interest before laser radiation is seen in (c), and the top-view volume rendering image of the area of interest after laser radiation for 1 min is seen in (d). Scale bar: 250 µm; color image size H x W: 1 x 2 mm.
Figure 5 shows the blood vessel coagulation test with the QCL. Figures 5a and 5b are the cross-sectional images at the positions marked by black lines in Figures 5c and 5d, respectively. Figures 5c and 5d are the top-view volume rendering images of the acquired 3-D data. Figure 5c is before laser radiation, and Figure 5d is after one minute of laser radiation. Comparing Figures 5c and 5d shows that the central blood vessel marked by the arrows gets thinned, and the membrane surface gets clean as some tiny soft-tissue structure was also coagulated. This also can be confirmed by comparing Figures 5a and 5b. We can see that the cross-sectional vessel size has decreased and that the top membrane surface becomes thinner and more uniform from (a) to (b).
In this study, we have demonstrated real-time OCT monitoring of 6.1-µm QCL-based lipid phantom tissue ablation, chicken egg yolk coagulation, and tissue and blood vessel coagulation in chicken embryo. In the near future, we plan to perform endoscopic laser ablation using in vivo animal models.
Meet the authors
Yong Huang is a PhD candidate in the department of electrical and computer engineering at Johns Hopkins University in Baltimore; email: [email protected]. Jin U. Kang is a professor and chairman in the same department; email: [email protected]. Since participating in this work, Adjani Peralta has graduated from the department of ecology and evolutionary biology at Princeton University in New Jersey and is pursuing a master’s degree in environmental sciences and policy at Central European University in Budapest, Hungary; email: [email protected].
Acknowledgment
This project is funded by NSF ERC (MIRTHE). Yong Huang is supported partially by the China Scholarship Council. The authors thank AdTech Optics and Dr. Janyu Fan for providing the laser and for the fruitful discussions.
References
1. M.H. Niemz (2007). Laser-Tissue Interactions: Fundamentals and Applications. 3rd Ed. Springer-Verlag, Berlin, Heidelberg.
2. S.S. Hayreh et al (1990). Argon laser panretinal photocoagulation in ischemic central retinal vein occlusion. Graefes Arch Clin Exp Ophthalmol, Vol. 228, pp. 281-296.
3. S.V. Esser et al (2009). Ultrasound-guided laser-induced thermal therapy for small palpable invasive breast carcinomas: A feasibility study. Ann Surg Oncol, Vol. 16, pp. 2259-2263.
4. V.A. Serebryakov et al (2010). Medical applications of mid-IR lasers. Problems and prospects. J Opt Technol, Vol. 77, pp. 6-17.
5. J. Faist et al (1994). Quantum cascade laser. Science, Vol. 264, pp. 553-556.
6. Y. Yao et al (2012). Mid-infrared quantum cascade lasers. Nat Photonics, Vol. 6, pp. 432-439.
7. S.A. Boppart et al (1999). High-resolution optical coherence tomography-guided laser ablation of surgical tissue. J Surg Res, Vol. 82, pp. 275-284.
8. W. Drexler and J.G. Fujimoto (2008). Optical Coherence Tomography: Technology and Applications. Springer-Verlag, Berlin, Heidelberg.
9. Y. Huang et al (2012). Real-time 3D and 4D Fourier domain Doppler optical coherence tomography based on dual graphics processing units. Biomed Opt Express, Vol. 3, pp. 2162-2174.
10. T. Leng et al (2004). The chick chorioallantoic membrane as a model tissue for surgical retinal research and simulation. Retina, Vol. 24, pp. 427-434.