Ekaterina Y. Lukianova-Hleb, Rice University; Xiaoyang Ren, University of Texas; Xiangwei Wu, University of Texas; Joseph A. Zasadzinski, University of Minnesota; Alcom K. Brenner, Baylor College of Medicine and Dmitri O. Lapotko, Ice University
New theranostic platform can find and kill specific cancer cells while sparing normal cells.
Some cancers are nearly impossible to remove without damaging important healthy organs. When such cancers appear to have a high resistance to drugs and radiation, they become practically incurable – and have a high rate of recurrence. The strategic solution would be to use a cell-level tool for detecting, modifying or killing specific cells without influencing surrounding cells and tissues. Unfortunately, modern materials and technologies cannot provide cell-specific, rapid, multifunctional processing and treatment of only the cancer cell.
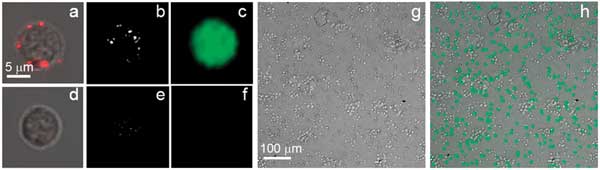
Images of target (CD3-positive) (a-c) and nontarget (CD3-negative) (d-f) cells. (a,d) confocal bright-field and optical scattering images show NP clusters (red) in cells; (b,e) time-resolved optical scattering images show bright PNB in target cell; (c,f) confocal fluorescent and bright-field images obtained after the PNB treatment show GFP fluorescence in target cell after 72 hours. Images of the mixture of cells (CD3-positive/CD3-negative 50:50): (g) bright-field and (h) GFP fluorescence after the PNB treatment (48 hours).
But a new cell-level theranostic platform rapidly detects, modifies or kills target cells while sparing normal cells with plasmonic nanobubbles (PNBs).
Plasmonic nanobubbles
Metal plasmonic nanoparticles (NPs) are the best converters of light into heat through the mechanism of surface plasmon resonance. This unique photothermal property was developed to allow precise manipulation of thermal energy at the nanoscale through engineered plasmon resonances.
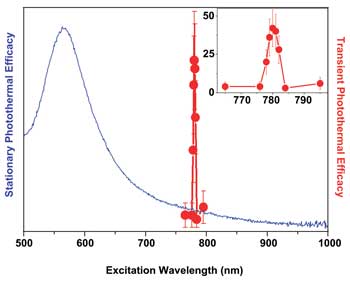
Spectra of the photothermal efficacy of gold solid spheres under stationary (blue) and
nonstationary (red) transient high-energy optical excitation with a 70-ps single laser pulse. Images courtesy of the authors.
Enhancement of the photothermal efficacy and spectral selectivity of such engineered NPs is associated with several principal limitations: Most commonly, stationary optical excitation creates high thermal losses that, in turn, require additional excitation energy, while the pulsed excitation involves high optical intensities that destroy NP structure that provides optical absorbance. Spectral width of absorption spectra of single NPs is tens of nanometers at best, while random clustering of NPs further broadens their spectra to hundreds of nanometers. An ability to deliver high photothermal efficacy with high spectral resolution and minimal thermal losses will therefore significantly improve current applications of plasmonic materials.
Until now, the photothermal and spectral properties of metal NPs have been set during their synthesis and have been assumed to stay constant during their excitation. This is a stationary paradigm, but an alternative approach can be based on the nonstationary excitation of NPs: the plasmonic nanobubble method.
The PNB is not a particle but, instead, a transient nanosecond event, a vapor nanobubble. It emerges in liquid overheated by a short-laser-pulse gold nanoparticle, expands and then collapses in nanoseconds. As a photothermal phenomenon, it can precisely deliver the localized mechanical impact and, at the same time, prevent bulk heating because it insulates all heat produced by an NP inside the vapor so the outside temperature does not rise.
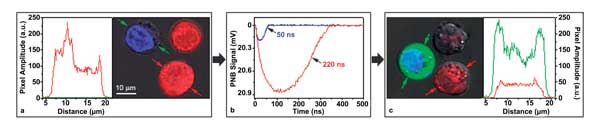 (a) J32 cells with intracellular clusters of gold spheres (NSP-OKT3, blue DAPI marker) and shells (NS-OKT3, Calcein Red marker); (b) optical scattering PNB-specific time responses of individual cells to a single laser pulse show simultaneous generation of small PNBs in blue and large PNBs in red cells in presence of extracellular molecular cargo FITC-Dextran (the lifetimes of PNBs are shown); (c) postlaser treatment blue cells show the injected FITC-Dextran (green fluorescence), and red cells show leaked-out Calcein Red dye and distorted membranes resulting from their destruction. The fluorescence intensity profiles of individual cells in (a) and (c) are indicated by small color-matched arrows.
(a) J32 cells with intracellular clusters of gold spheres (NSP-OKT3, blue DAPI marker) and shells (NS-OKT3, Calcein Red marker); (b) optical scattering PNB-specific time responses of individual cells to a single laser pulse show simultaneous generation of small PNBs in blue and large PNBs in red cells in presence of extracellular molecular cargo FITC-Dextran (the lifetimes of PNBs are shown); (c) postlaser treatment blue cells show the injected FITC-Dextran (green fluorescence), and red cells show leaked-out Calcein Red dye and distorted membranes resulting from their destruction. The fluorescence intensity profiles of individual cells in (a) and (c) are indicated by small color-matched arrows.
PNBs have the benefit of nonstationary optical excitation of gold nanoparticles with a very short picosecond optical pulse. For example, for solid gold spheres (known also as gold colloids), we achieved with this method an approximately hundredfold transient amplification of the photothermal efficacy and the unprecedented narrowing of the photothermal spectra to 2 to 3 nm for solid gold nanospheres under off-resonant nonstationary optical excitation at 780 nm. This amplification and spectral narrowing were achieved with a 70-ps laser pulse for a wide range of fluences starting from 10 mJ/-2; and a range of gold nanoparticle properties, such as size, aggregation state and environment, including living matter.
The transient nature and high spectral selectivity of the observed effect can be associated not with the nanoparticle itself, but with the nonstationary formation of a transient nanostructure with new optical properties. Thus, nonstationary optical excitation of metal nanoparticles can significantly improve their photothermal efficacy and spectral selectivity, and this can be reliably reproduced using the methodology described above. In particular, it allows the successful use of solid nanospheres, which are cheap, easily available, biologically safe and stable NPs for near-infrared applications.
Gene and cell therapy
Most of the cell and gene therapies that have shown promise against human diseases such as cancer require ex vivo processing of human cell grafts to eliminate unwanted cells from a heterogeneous suspension and to genetically modify one or more cell subsets to increase their therapeutic efficacy. Ideally, both elimination and transfection should be highly efficient, selective, fast and safe for cells.
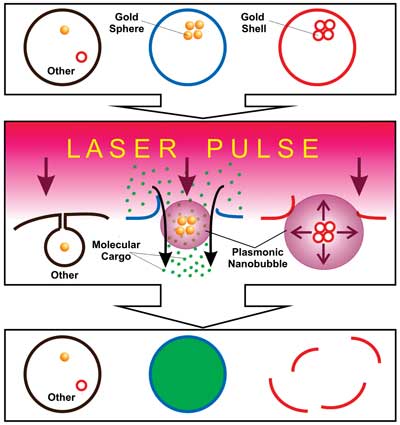
Multifunctional cell-specific processing of heterogeneous cell system is illustrated, with plasmonic nanobubbles that are selectively generated around clusters of gold spheres in spheres-targeted cells (blue) and around clusters of gold shells in shells-targeted cells (red) with a single laser pulse. This results in simultaneous delivery of molecular cargo into blue cells resulting from injection of the molecules (green dots) with small plasmonic nanobubbles (PNBs) and mechanical destruction of red cells with large PNBs without damage to other cells, all realized in a single pulse treatment.
Existing methods, however, lack such characteristics, especially multifunctionality and selectivity when applied to a heterogeneous cell system. As a result, current cell processing often is slow, expensive and labor-intensive and is compromised with high cell losses and poor selectivity, limiting the efficacy and availability of cell therapies.
We have developed a universal technology for multifunctional simultaneous guided transfection of target cells and elimination of subsets of unwanted cells in heterogeneous grafts that has single-cell-type selectivity, high efficacy and processing rate, and low toxicity. We have shown that the ability of each NP type to generate PNBs of different sizes – under identical optical excitation, coupled with the cell-specific targeting and clustering of NP conjugates – allows simultaneous delivery of molecular cargo into gold sphere-targeted T-cells, and destruction of gold shell-targeted unwanted cells in a single bulk treatment of the mixed cell suspension with high efficacy, speed and selectivity and with low toxicity. This technology will create a universal platform for cell and gene therapy and for stem cell transplantation.
The long-term objective of this research is to improve the outcome of diseases whose treatment requires ex vivo cell processing. The ability to simultaneously genetically modify target cells and eliminate other specific cells from a highly heterogeneous cell system (graft), with single-cell selectivity and without compromising other important cellular components, will enhance the feasibility and effectiveness of cell engineering in general and gene therapies in particular. The technology may subsequently be applied to process any liquid tissues
to improve the outcome of other cell-based interventions in cancer and other disorders.
Drug delivery
Some aggressive cancers are still difficult to treat because of 1) incomplete removal of cancer cells by surgery, especially when complicated by micrometastasis and colocalization of cancer cells with functionally or cosmetically important structures; 2) multidrug resistance of cancer cells; and 3) acute and long-term toxicities of radio- and chemotherapies. Therefore, new treatment strategies are needed to provide cell-level selectivity of cancer diagnosis and treatment, and high efficacy against drug-resistant cells.
The specificity and functionality of drug delivery to cancer cells has been improved by more than one order of magnitude through the mechanism of plasmonic nanobubbles. The PNB method effectively discriminates between cancer and normal cells under the identical treatment of both with NPs and optical radiation. By combining the threshold nature of PNBs and the enhanced accumulation and clustering of NPs in cancer cells, we have shown that PNBs, unlike NPs, can be minimized or avoided in normal cells despite the uncontrollable nonspecific uptake of NPs by such cells.
The temporally and spatially controlled initiation and collapse of PNBs create local optical and mechanical effects that can enable imaging intracellular molecular targeting, localized drug or gene delivery, and selective elimination of cells for therapeutics, theranostics and microsurgery. The optical and acoustical properties of PNBs provide a mechanism for real-time guidance of their therapeutic action. PNBs also offer improved safety as a result of their transient, on-demand nature; PNBs do not exist until activated with an optical pulse; then they disappear within nanoseconds.
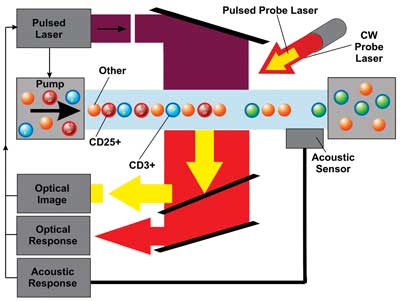
Functional diagram of the flow system with the pulsed broad excitation laser (purple), flow
cuvette with the pump and reservoir, and three PNB detection paths: Optical time-response is detected with continuous red laser, optical scattering image is detected with yellow pulsed laser, and acoustic response is detected with acoustic sensor; all signal detectors and the flow pump are synchronized with the pulsed excitation laser.
We compared three modes of the cell-level delivery of therapeutic effect with gold NPs, hyperthermia (thermal), plasmonic nanobubbles (mechanical) and a combination of drugs with plasmonic nanobubbles (chemotherapeutic). Of these, the combination of plasmonic nanobubbles with standard anti-cancer drugs demonstrated the best effect as a proof of principle for a novel nanotherapeutic mechanism for selective, efficient, safe and guided treatment of drug-resistant superficial cancer.
1. Selective intracellular delivery of standard extracellular drugs via laser-induced PNBs overcomes drug and thermal resistance of cancer cells.
2. High therapeutic selectivity is achieved through high cellular specificity of PNBs under excitation with broad laser beams in single-pulse mode at a physiologically safe level of laser radiation.
3. Drug doses required for total destruction of cancer cells are reduced by an order of magnitude, while nonspecific toxicity among normal cells is reduced sevenfold, and treatment time is reduced from days to minutes.
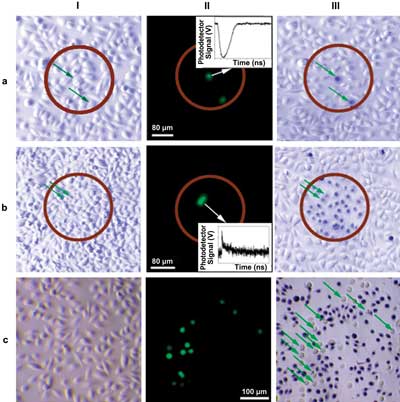
Bright-field (I, III) and fluorescence (II) microscopy images of a co-culture of normal (NOM9) and squamous cell carcinoma (HN31, green or shown with green arrows) cells. Images I and II were taken before treatment, and image III was taken after treatment and after staining the cells with Trypan Blue (blue: dead cells; white: live cells). (a) PNB treatment with a single laser pulse (70 ps, 820 nm, 40 mJ/cm-2), the time-response (insert) obtained from one of the cancer cells shows a PNB; (b) Nano-hyperthermia treatment (NP concentration was increased fivefold, laser treatment: 40 Hz, 820 nm, 24 J/cm-2) the time-response (insert) obtained from one cancer cell shows a heating-cooling signal; (c) Cisplatin (5 µg/ml) treatment for 72 h.
Perspectives
Our long-term goal is to develop a universal platform that radically and selectively improves the treatment of cancer cells but spares normal cells and organs. We address several critical needs of current laser surgery and chemo-radiotherapeutics: 1) the resistance of several cancers to current therapeutics; 2) the severe nonspecific toxicities and functional impairments resulting from high doses of drugs and x-rays; 3) the cosmetic and functional morbidity after surgical resection of some tumors; 4) residual disease and local recurrences; and 5) lack of oncological specificity, efficacy and speed among new technologies and materials.
Cancer cell theranostics (united diagnosis and treatment) with PNBs offers an opportunity that is unique and distinct from current surgical and therapeutic agents due to the following: 1) Cell-level treatment is achieved through an on-demand intracellular mechanical, nonthermal process that 2) is efficiently localized only in cancer cells under identical simultaneous exposure of tumors and normal tissues to laser pulse and gold nanoparticles, 3) uses safe doses of gold nanoparticles and a single near-infrared laser pulse, 4) can be activated in superficial and deep tissue with standard clinical laser surgical tools and 5) combines detection of cancer cells with their immediate destruction in a single rapid procedure with real-time guidance.
Meet the authors
E.Y. Lukianova-Hleb is a research scientist in the Department of Biochemistry and Cell Biology at Rice University in Houston; email: [email protected]. X. Ren is a senior research assistant in the Department of Cancer Prevention at the University of Texas MD Anderson Cancer Center in Houston; email: [email protected]. X. Wu is associate professor in the Department of Cancer Prevention at the University of Texas MD Anderson Cancer Center in Houston; email: [email protected]. J.A. Zasadzinski is a professor in chemical engineering and materials science at the University of Minnesota in Minneapolis; email: [email protected]. M.K. Brenner is a professor in the Departments of Pediatrics and of Medicine, Center for Cell and Gene Therapy at Baylor College of Medicine, Texas Children’s Hospital, The Methodist Hospital in Houston; email: [email protected]. D.O. Lapotko is faculty fellow in the Department of Biochemistry and Cell Biology at Rice University in Houston as well as faculty fellow in the Department of Physics and Astronomy at Rice; email: [email protected].
References
1. E. Lukianova-Hleb et al (2012). Cell-specific transmembrane injection of molecular cargo with gold nanoparticle-generated transient plasmonic nanobubbles. Biomat. Vol. 33, pp. 5441-5450.
2. E. Lukianova-Hleb et al (2012). Plasmonic nanobubbles enhance efficacy and selectivity of chemotherapy against drug-resistant cancer cells. Adv Mater. Vol. 24, pp. 3831-3837.
3. E. Lukianova-Hleb et al (2012). Cell-specific multifunctional processing of heterogeneous cell systems in a single laser pulse treatment. ACS Nano. Vol. 6, pp. 10973-10981.
4. E.Lukianova-Hleb et al (2012). Transient enhancement and spectral narrowing of the photothermal effect of plasmonic nanoparticles under pulsed excitation. Adv Mater (doi: 10.1002/adma.201204083).
For more information on the work done at Rice University’s Nanobubble Lab, visit http://lapotko.rice.edu.