Chen-Wei Wei, JinJun Xia, Ivan Pelivanov, Xiaoge Hu, Xiaohu Gao and Matther O'Donnell, University of Washington
A new technique could help metastasis diagnosis by magnetically trapping rare circulating tumor cells in fast blood flow with simultaneous noninvasive photoacoustic detection.
Circulating tumor cells (CTCs) originate in a primary tumor and spread to distant organs through the circulatory system to develop a secondary tumor – i.e., metastasis. The rarity of CTCs – one to 10 cells per milliliter in blood with metastasis1 – makes them very difficult to identify in the blood pool. Although several techniques2-6 have been proposed to isolate and enrich CTCs for further analysis, a noninvasive, real-time approach is still not feasible.
Photoacoustic (PA) imaging, which uses an ultrasound (US) transducer to detect light-induced acoustic signals, is a relatively new imaging modality that maintains the advantages of US imaging, including nonionizing operation, low cost and submilliliter spatial resolution up to several centimeters in depth. However, its unique contrast mechanism – optical absorption – makes it an attractive modality to detect and differentiate specific molecules or diseased cells. A number of investigators have shown that contrast agents with selective absorption at a specific wavelength, such as highly absorptive nanoparticles, can be effectively targeted to biological objects for PA molecular imaging with high sensitivity and specificity.7-9
Unfortunately, identifying CTCs in blood flow using PA techniques remains a big challenge because the weak signals from rare targeted cells can be easily masked by strong background PA signals from blood.
Recently, our group developed a technique called magnetomotive photoacoustic (mmPA) imaging, which introduces magnetic force into conventional PA imaging to suppress background signals.10-12 By biochemically coupling a composite contrast agent with combined strong magnetic and optical absorption properties to diseased cells, these targeted cells can be magnetically enriched and detected by PA imaging. Also, signals from the tagged cells can be modulated by changing the magnetic field spatially or temporally. As a consequence, background signals insensitive to magnetic field can be eliminated with postprocessing of received PA images.
Figure 1 illustrates how mmPA imaging works to detect CTCs. In particular, composite particles combining a gold nanorod (GNR) with Fe3O4-based magnetic nanoparticles (MNPs) and a targeting molecule such as folic acid (FA) target CTCs. By applying a magnet trapping force, these cells accumulate in a certain region within the blood vessel and can be remotely detected using PA imaging (Figure 1a). Background suppression can also be achieved by applying motion filtering to PA images with different magnetic trapping orientations to identify targeted cells sensitively and specifically (Figure 1b).
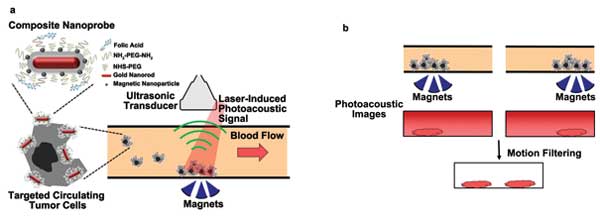
Figure 1. (a) Magnetic trapping and photoacoustic detection of circulating tumor cells targeted with composite GNR-MNP-FA contrast agents in a blood vessel. (b) Motion filtering on photoacoustic images with trapping magnets at different positions to suppress background blood signals. Images courtesy of the authors.
A human cervical cancer cell line, HeLa, was targeted with GNR-MNP-FA particles. These cells served as the imaging target in a flow phantom with a rate of 6 ml/min, mimicking CTCs in a human radial artery. Fusion images combining interleaved ultrasound and PA images of the magnetic trapped cells with a concentration of 5000 cells per milliliter water solution at different time points of magnet action are shown in Figure 2. The ultrasound signals of the tube’s top and bottom wall can be clearly seen in gray-scale display.
The PA signals (shown in the warmer colors) from the cells flowing in the tube are initially under the detection limit at the beginning of trapping, so no PA signals are seen in the trapping region at 5 s after applying the magnetic force. The PA signals increase and expand with time, indicating cell accumulation. At the 310-s time point, the magnet was removed, and accumulated cells were washed out from the trapping region by the flow because they had lost the trapping force. The magnets under the tube also absorbed light and reflected the US transmitting beam, contributing the PA and US signals at a depth of about 20 mm in these images.
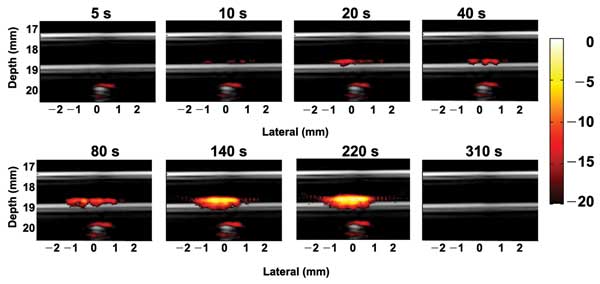
Figure 2. Fusion images (ultrasound plus photoacoustic) of trapping targeted HeLa cells in a tube phantom mimicking a blood vessel at different time points of magnet action. The flow rate is 6 ml/min, and the cell concentration is 5000 cells per millimeter.13
To demonstrate the trapping efficacy and imaging sensitivity of the current system for potential clinical use, the trapping experiments were repeated using a cell sample with a concentration of one cell per milliliter, the lower end of the typical concentration of CTCs in blood. Furthermore, in contrast to the flow setup used in Figure 2, cells are allowed to flow through the trapping region only once – i.e., no recirculation. The purpose was to demonstrate that a large percentage of targeted cells traversing the high magnetic field region can be effectively trapped.
Figure 3 depicts four representative fusion images at different time points of the trapping process. As mentioned previously, no signals can be detected at the beginning. At about 40 min, the signals of the accumulated cells just exceed the background level and can be detected. More and more cells were trapped with time, and several cell clusters can be observed in the trapping region. The trapping efficiency – i.e., the percentage of trapped cells – was estimated as 67 percent, and the minimum detectable cell number was 136.
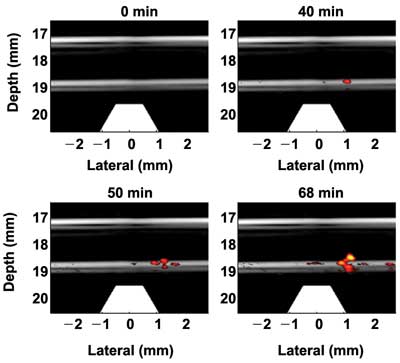
Figure 3. Fusion images (ultrasound plus photoacoustic) of trapping targeted HeLa cells in a solution with a concentration of a single cell per milliliter in a tube phantom.14 The flow rate is 6 ml/min.
These results suggest that the current mmPA setup may be able to provide sensitive metastasis diagnosis within a 40-min exam procedure in vivo and noninvasively. The exam time can be greatly reduced by optimizing the setup to improve the trapping efficiency and detection sensitivity, such as increasing the laser fluence and developing a more robust magnet system.
Trapping cells in a highly absorptive background also was performed to demonstrate background signal suppression using mmPA imaging. Instead of cells in clear water solution, ink was added to mimic strong blood absorption producing strong background signals, which seriously degrade sensitivity and specificity in rare-CTC detection. It is nearly impossible to see the signals in conventional PA imaging from the trapped cells, even with a higher cell concentration (500 cells/ml), shown as Figures 4a and 4b with the magnet at different positions. Nevertheless, changing the magnet field creates corresponding movement of magnetic-particle-targeted cells but not the background signals insensitive to the magnetic field. By applying image subtraction between the two images, the background signals can be eliminated, but the signals from the cells are preserved in the differential image (Figure 4c).
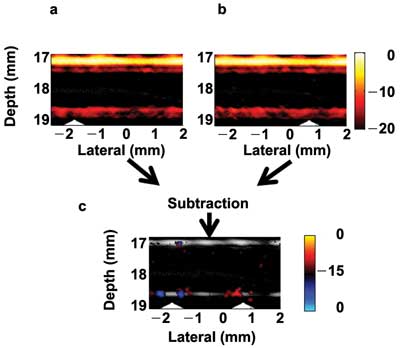
Figure 4. (a, b) Fusion images (ultrasound plus photoacoustic) of targeted cells in ink solution (10 cm–1 at 800 nm) acquired for two different positions of the magnets. (c) Differential image of (a) and (b) showing contrast enhancement of the trapped cells at the magnet positions (triangles) through suppression of the background ink signal.13
Our in vitro results in the phantom study demonstrate that the current mmPA imaging system has great potential to detect a very small number of CTCs with a sensitivity of a single-cell-per-milliliter solution, and to differentiate trapped cells from a large PA background.
We plan to continue in vitro studies, including tests of targeting efficiency of contrast agents to CTCs in real blood, specific tests using control cell lines and toxicity tests. We also will move toward preclinical studies in small animals with a well-developed metastasis model. Based on these studies, mmPA imaging has the potential to become a valuable clinical tool for ultrasensitive detection of CTCs in the human vasculature.
Meet the authors
Dr. Chen-wei Wei is a postdoctoral researcher in bioengineering at the University of Washington (UW) in Seattle; email: [email protected]. Dr. Jinjun Xia and Dr. Ivan Pelivanov work on ultrasound and photoacoustic imaging at UW. Dr. Xiaoge Hu was a postdoctoral researcher in Dr. Xiaohu Gao’s lab at UW from 2008 to 2012. Dr. Gao is a faculty member in the Department of Bioengineering and the Center for Nanotechnology at UW. Dr. Matthew O’Donnell is a professor of bioengineering at UW.
Acknowledgments
This work was supported in part by NIH RO1EB016034, RO1CA170734, R01CA131797, R01CA140295, T32CA138312, NSF 0645080, the Life Sciences Discovery Fund 3292512, and the Department of Bioengineering at the University of Washington.
References
1. M.C. Miller et al (2010). Significance of circulating tumor cells detected by the cell search system in patients with metastatic breast, colorectal and prostate cancer. J Oncol, Vol. 2010, Article ID 617421.
2. M. Fleisher (2008). A new opportunity for therapeutic management of cancer patients. Clin Lab News, Vol. 34, No. 11.
3. S. Nagrath et al (2007). Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nat, Vol. 450, pp. 1235-1239.
4. S. Zhenga et al (2007). Membrane microfilter device for selective capture, electrolysis and genomic analysis of human circulating tumor cells. J Chromatog A, Vol. 1162, Issue 2, pp. 154-161.
5. D.R. Shaffer et al (2007). Circulating tumor cell analysis in patients with progressive castration-resistant prostate cancer. Clin Canc Res, Vol. 13, Issue 7, pp. 2023-2029.
6. P. Paterlini-Brechot and N.L. Benali (2007). Circulating tumor cells (CTC) detection: clinical impact and future directions. Canc Lett, Vol. 253, Issue 2, pp. 180-204.
7. A. Zerda et al (2010). Ultrahigh sensitivity carbon nanotube agents for photoacoustic molecular imaging in living mice. Nano Lett, Vol. 10, Issue 6, pp. 2168-2172.
8. S.Y. Emelianov et al (2009). Photoacoustics for molecular imaging and therapy. Phys Today, Vol. 62, Issue 8, pp. 34-39.
9. P.C. Li et al (2008). In vivo photoacoustic molecular imaging with simultaneous multiple selective targeting using antibody-conjugated gold nanorods. Opt Exp, Vol. 16, Issue 23, pp. 18605-18615.
10. Y. Jin et al (2010). Multifunctional nanoparticles as coupled contrast agents. Nat Comm, Vol. 1, Article No. 41, doi:10.1038/ncomms1042.
11. J. Xia et al (2012). Suppression of background signal in magnetomotive photoacoustic imaging of magnetic microspheres mimicking targeted cells. J Biomed Opt, Vol. 17, Issue 6, 061224.
12. C.W. Wei et al (2012). Trapping and dynamic manipulation of polystyrene beads mimicking circulating tumor cells using targeted magnetic/photoacoustic contrast agents. J Biomed Opt, Vol. 17, Issue 10, 101517.
13. C.W. Wei et al (February 2013). Magnetic trapping with simultaneous photoacoustic detection of molecularly targeted rare circulating tumor cells. Proc. SPIE, Vol. 8581, Article No. 85814W, San Francisco.
14. X. Hu et al (2012). Trapping and photoacoustic detection of CTCs at the single cell per milliliter level with magneto-optical coupled nanoparticles. Small, doi: 10.1002/smll.201202085.