Wei-Chaun Shih, University of Houston
Monolithic hierarchical plasmonic nanostructures improve results from surface-enhanced Raman spectroscopy.
Plasmonic nanostructures of gold and silver have emerged as attractive nanomaterials because of their small size, corrosion resistance, biocompatibility and ability to bind to a wide range of thiolated molecular and biomolecular species through the metal-S bond. These nanostructures absorb and resonantly scatter visible and near-IR light upon excitation of the localized surface plasmon resonance (LSPR), which can be tuned over a wide spectral range by changing intrinsic parameters such as material composition
or particle size and shape (sphere, rod, cube, triangle, cage, core shell, etc.). The spherical gold nanoparticle (AuNP) is known to exhibit an LSPR peak around 530 nm with limited tunability by varying its diameter; AuNPs of other shapes or configurations have been shown to exhibit LSPR redshift to take advantage of the “diagnostic window” for biological and biomedical applications.
Two approaches have been heavily investigated and employed to fabricate plasmonic nanostructures to date: chemical synthesis and lithographic patterning. Chemically synthesized AuNPs are typically prepared in the solution phase with high production throughput; although various shapes such as spheres, rods, core-shell structures and cages have been demonstrated, size and shape control need careful consideration. In contrast, lithographic patterning, borrowed from the semiconductor industry, can define highly regular AuNPs in size and shape but might not have enough production throughput for certain applications.
Thanks to LSPR, the light-scattering signal from AuNPs is intense and provides a significant advantage for biomedical and biological molecular imaging, where the use of dyes, fluorophores or quantum dots is limited by low signal intensities, complex blinking phenomena, photobleaching and toxicity. Besides scattering, the efficient optical absorption enables photothermal disruption and photothermally controlled drug release, leading to highly targeted cancer therapeutics. Immobilized AuNPs have gained intense interest for their potential in biosensing; for example, bio-recognition can be achieved by decorating the gold surface with antibodies, single-stranded oligonucleotides, aptamers and so on.
Besides the plasmonic properties and the thiol chemistry, another key advantage of AuNPs compared with their bulk counterpart is the increased specific surface, which facilitates chemical reaction and thus promotes their effectiveness in various applications. However, existing AuNPs do not exhibit porous features commonly observed in nanomaterials such as mesoporous silica, silicon and carbon.
Large specific surface, plasmonic properties
Nanoporous gold (NPG), in contrast, features nanoscale ligaments and pores throughout its structural monolith.1 NPG films have recently captured intense attention for their large specific surface, on the order of 10 m2/g. An NPG is a product of selective dissolution of a less noble metal from an alloy, also known as “dealloying.” To date, most NPG materials have been fabricated by free corrosion in acids or by electrochemical methods; a gold-silver alloy is the most common starting material. Depending on the atomic fraction of gold and silver in the alloy, the dealloying temperature and time, and postdealloying annealing conditions, various pore sizes – from a few nanometers to a few hundred nanometers – have been reported. NPG has attracted much attention because of its extraordinarily large surface area, a tantalizing feature for applications such as
catalysis, sensing and actuation. In addition, NPG maintains a high degree of structural integrity and uniformity – rare among chemically synthesized nanostructures.
NPG films 500 µm thick have been used as effective catalysts in low-temperature oxidation.2 Taking advantage of a large surface area is not a new idea, but the use of NPG avoids problems associated with techniques that rely on random decoration of ~5-nm AuNPs on supporting substrates – for instance, sintering at elevated temperatures, which degrades performance and the useful lifetime of the catalyst. It also is challenging to build a truly 3-D nanoporous network using self-assembly of tiny AuNPs. But NPG features a continuous porous thin film without the need for a support substrate, thereby increasing long-term stability.
NPG films also have shown great promise as large-surface-area electrodes. An NPG cathode has been demonstrated to withstand 100 charge-discharge cycles without significant performance degradation in Li-air batteries, whereas a porous carbon cathode degrades much faster.3 NPG films have been patterned into ~10 x 10-μm2 electrodes for recording neuronal action potentials. Better signal quality has been obtained because of lower contact impedance from the large surface area.
Recently, researchers have explored the plasmonic aspect of NPG.4 The plasmon resonance peak determined by extinction spectroscopy has been characterized with respect to pore size, pore density and ambient refractive index, showing limited tunability around 600 to 650 nm.5
We are interested in exploiting NPG’s plasmonic properties for biosensing and imaging, and its large surface area for drug-delivery application.
NPG for SERS, fluorescence
NPG has been used for molecular sensing via surface-enhanced Raman spectroscopy (SERS) and metal-enhanced fluorescence (MEF) mechanisms. Since the demonstration of single-molecule detection, SERS has become a heavily researched technique for chemical/molecular sensing. Unlike fluorescence, SERS does not suffer from photobleaching. It also contains rich molecular structural information, and therefore has potential for highly multiplexed sensing and imaging. Most NPG-based SERS sensors are in the thin-film format. A popular fabrication method involves the free corrosion of a ~100-nm thick “white gold leaf” in nitric acid. Although SERS activity has been documented, wide variations in SERS enhancement factors (EFs) have been reported,6-8 possibly reflecting differences in material composition and morphology, fabrication technique, SERS marker and/or excitation wavelength.
Recognizing that the intrinsic LSPR peaks of NPG films exhibit limited tunability around 600 to 650 nm, my group at the University of Houston has developed an approach to shift these peaks by shape engineering, or patterning, NPG films into discrete submicron or nanoscale disks. Noting that LSPR peak shift in solid (i.e., nonporous) Au disks of similar size has been extensively investigated, the team developed a hybrid fabrication process that involves both “top down” and “bottom up” approaches to produce submicron NPG disks (NPGDs), as shown in Figure 1a.

Figure 1. (a) A top view of nanoporous gold disks (NPGDs) ringed by gold; (b) an unpatterned NPG film. The author’s group has developed a hybrid fabrication process involving “top down” and “bottom up” approaches to produce submicron NPGDs.
To fabricate an NPGD, an alloy film is deposited on a silicon substrate by physical vapor deposition; e.g., sputtering. A lithography step using polystyrene beads as masks is used to define the lateral geometry of individual units, or disks, and is followed by free corrosion in nitric acid. This process enables fabrication of monolithic NPGD arrays with high uniformity, excellent shape control and decent yield. Note that the patterning step does not alter the ultrafine NPG structures because it has been carried out before dealloying. In other words, the ultrafine nanoporous network inside the disk is similar to that obtained in unpatterned NPG thin films (Figure 1b) fabricated by the same dealloying procedure. The cracks caused by shrinkage during Ag dissolution are quite similar as well.8
We have investigated using NPGDs as SERS substrates, with 785 nm as the laser-excitation wavelength and benzenethiol (BT) molecules as the SERS marker. We chose BT molecules and this wavelength because the absence of a BT absorption peak near 785 nm minimizes the ambiguity presented by resonant Raman scattering, while the ability of BT to form self-assembled monolayers (SAMs) enables the number of molecules on individual NPGDs to be quantified. Additionally, the SERS activity at 785-nm laser excitation has critical significance for deep-tissue penetration in any potential biological and biomedical applications.
Benzenethiol SAMs were deposited on unpatterned NPG, NPGDs and Klarite (SERS enhancement factor at least 106 for benzenethiol, according to Renishaw) substrates. SERS measurements were carried out with 785-nm excitation using a home-built line-scan Raman microscopy system,9 enabling SERS mapping over 133 x 133-µm2 regions with ~1-µm2; resolution (full width half maximum) at a spectral resolution ~8 cm–1.
Figure 2 shows the SERS spectra in CCD count rate (CR), normalized to the laser power and the area of the laser spot, for BT SAMs on the NPGD, NPG and Klarite substrates, as well as the normal Raman spectrum obtained from neat benzenethiol solution. (The curves are offset for clarity.) The CR for NPGD substrates is ~500 times larger than for unpatterned NPG and ~400 times larger than for Klarite. Because the nanoporous structure and thickness are identical for the NPGD and unpatterned NPG substrates, including the density and average size of the cracks, the patterning process has produced a 500-fold increase in enhancement factor. The interpretation of the increased Raman scattering of the NPGD versus Klarite substrate requires an estimate of the total number of BT molecules attached to a disk, and the results suggest our NPGD has an absolute SERS EF >108; this has been confirmed by comparing it with non-SERS measurements from bulk BT solutions. A variety of NPGD diameters can be fabricated using polystyrene beads of different sizes. Currently, we have made an NPGD with a diameter as small as 100 nm.
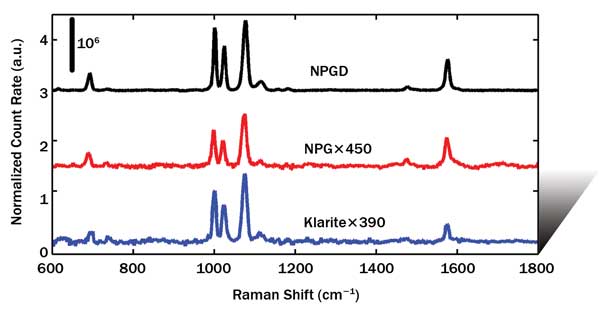
Figure 2. Normalized count rate (CR) of BT self-assembled monolayers from an NPGD, unpatterned NPG and Klarite vs. Raman shift (cm–1). The curves are offset for clarity.
The number of adsorbed BT molecules in and on an NPGD is the product of the area of the disk, the roughness R of NPG (the ratio of the chemically active surface area to the geometrical surface area), and the BT surface density. Reference 6 describes a procedure for estimating the roughness of an NPG film from scanning electron microscope images of the surface, assuming uniform nanostructure through the film. An analysis of Figure 1b using ImageJ shows a roughness of ~22. Assuming a packing density of 6.8 x 1018 molecules/m2, approximately 8.4 x 106 molecules (~14 attomoles) are adsorbed on a single disk.
To demonstrate the NPGD’s potential in fast detection of trace molecular amounts, we have performed a series of experiments by varying the CCD integration time. In Figure 3a, we show the results with integration time from 1 s to 25 ms. The signal-to-noise ratio (SNR, in dB) versus integration time is plotted in the inset, suggesting that a single NPGD coated with BT SAM can be detected with an SNR of ~13 dB in 25 ms (the shortest integration time allowable in our system). The laser power density was 170 µW/µm2. All previous results were obtained using a CCD cooled to –70 °C.
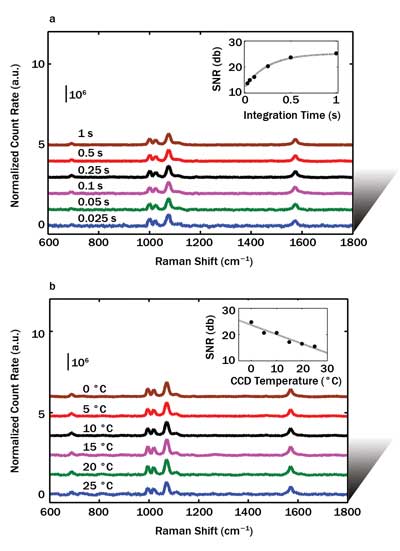
Figure 3. The author’s group demonstrated the potential of NPGDs in fast detection of trace molecular amounts with tests that involved varying the CCD integration time. Shown is the normalized count rate (CR) and signal-to-noise ratio (SNR) from a single NPGD vs. CCD integration times (a) and CCD temperatures (b).
Next, we explored the detection capability of a single NPGD with various CCD temperatures; the results are shown in Figure 3b. The SNR versus CCD temperature is plotted in the inset, suggesting NPGD can be detected with an SNR of ~14 dB at a CCD temperature as high as 25 °C. The laser power density was 1.7 mW/µm2, and the integration time was 1 s.
The NPGD’s excellent SERS performance derives from its high-density internal “hot spots” around the gaps and junctions of the nanoporous network, as suggested by our recent finite difference time domain simulation results. The electric field near these hot spots can be enhanced to >40 times. NPGD represents a new type of plasmonic entity with “three dimensional” plasmonic enhancement. NPGD has a distinct structural hierarchy – the outer disk shape (~100 to 500 nm) and ultrafine internal pores (~5 to 15 nm), which is rare in existing AuNPs created either by solution synthesis or lithographic patterning. One potential use of NPGDs is for biochip applications; e.g., label-free detection of biomolecules. Our early results suggest that a single hybridization event can be detected on an NPGD.
Figure 4. Hybrid nanofabrication produces monolithic hierarchical nanoporous gold disks with stronger plasmonic enhancement for surface-enhanced Raman spectroscopy.
Meet the author
Wei-Chuan Shih is an assistant professor in the Department of Electrical & Computer and Biomedical Engineering at the University of Houston in Texas; email: [email protected].
References
1. J. Erlebacher et al (2001). Evolution of nanoporosity in dealloying. Nat, Vol. 410, Issue 6827, pp. 450-453.
2. A. Wittstock et al (2010). Nanoporous gold catalysts for selective gas-phase oxidative coupling of methanol at low temperature. Sci, Vol. 327, Issue 5963, pp. 319-322.
3. Z.Q. Peng et al (2012). A reversible and higher-rate Li-O–2 Battery. Sci, Vol. 337, Issue 6094, pp. 563-566.
4. J. Biener et al (2008). Nanoporous plasmonic metamaterials. Adv Mat, Vol. 20, Issue 6, pp. 1211-1217.
5. X.Y. Lang et al (2011). Localized surface plasmon resonance of nanoporous gold. Appl Phys Lett, Vol. 98, Issue 9, p. 093701.
6. S.O. Kucheyev et al (2006). Surface-enhanced Raman scattering on nanoporous Au. Appl Phys Lett, Vol. 89, p. 053102.
7. L. Zhang et al (2011). Wrinkled nanoporous gold films with ultrahigh SERS enhancement. ACS Nano, Vol. 5, issue 6, pp. 4407-4413.
8. J. Qi et al (2013). Surface-enhanced Raman spectroscopy with monolithic nanoporous gold disk substrates. Nanoscale, Vol 5, pp. 4105-4109.
9. J. Qi et al (2012). High-throughput Raman and surface-enhanced Raman microscopy. Proc SPIE 8219, Biomedical Vibrational Spectroscopy V: Advances in Research and Industry, p. 821903.
10. W.-C. Shih (2013). Nanoporous gold nanoparticles as high-payload molecular cargos, photothermal/photodynamic therapeutic agents, and ultrahigh surface-to-volume plasmonic sensors, US Patent Application 13,796,201.