Dr. Ofer Levi, University of Toronto
Agile vertical-cavity surface-emitting light sources deliver fast multimodality brain imaging for disease monitoring and long-term brain studies
Optical imaging techniques play a major role in daily biological research. While current applications focus on in vitro diagnosis and preclinical animal studies, new imaging techniques can also be used in clinical settings to study the brain in action.1,2 High spatial and temporal resolution has made optical imaging a desirable alternative to commonly used high-cost clinical imaging techniques such as functional MRI, single-photon emission computed tomography, or positron emission tomography.
In many cases, animal studies can provide an insight into the disease state that can be applicable to human patients. However, their high sensitivity to movement requires scientists to use anesthesia during the imaging session. The effects of anesthesia on the neural and hemodynamic responses limit the neural signals that can be imaged. As a result, scientists are developing portable, real-time imaging techniques that directly monitor neural activity in awake, moving animals.3
Fluorescence imaging is a popular in vivo imaging technique. It can be applied on different scales, from high-resolution optical microscopy to whole-brain wide-field imaging.4 Optical contrast can be generated by exogenous fluorescence from administered molecular probes, reporter genes and endogenous fluorescence.
Optical signals can reveal underlying physiology, quantify a molecular target, map specific cell populations or structures (such as neurons, glia and blood vessels), and follow their temporal dynamics. Moreover, voltage- or Ca2+-sensitive dyes can be used in functional imaging mode, where brain activity is mapped into a dynamic optical image series.5
Label-free hemodynamic measurements of blood flow or tissue oxygenation can provide complementary valuable information about hemodynamic response and neurovascular coupling, which play a major role in many neurological diseases. This information cannot easily be derived from direct electrophysiology recordings. In particular, intrinsic optical signal imaging (IOSI) and laser speckle contrast imaging (LSCI) techniques are used as complementary methods for quantifying blood oxygenation values and blood flow speeds, respectively.6
In many disease scenarios, such as stroke, epilepsy or traumatic brain injury, the distinction between oxygen metabolism and compensatory blood flow is important in understanding the disease or estimating the size of the affected area, or in evaluating stroke recovery. Simultaneous, multimodal brain imaging allows users to co-register all imaging channels into one camera, thus improving the evaluation of the underlying disease state.
VCSELs for brain imaging
Vertical-cavity surface-emitting lasers (VCSELs) offer great advantages over alternative light sources for brain imaging. In many applications, it is difficult to apply LSCI and IOSI together using the same light source. LSCI requires a highly coherent laser source to produce the speckle needed to discern flow, while IOSI needs a low-coherence, wider-spectrum source to produce the low-noise illumination needed to measure small fluctuations in reflectance. To overcome this challenge, we developed an imaging system that uses coherence control in VCSELs to produce optimized illumination for both IOSI and LSCI from a single laser source.7
When operated near the lasing threshold, high-coherence single-mode operation is optimized for LSCI. Operating in a specialized current sweep mode leads to blending of many transverse laser modes and to reduction of the laser coherence, making it optimized for IOSI. Fluorescence imaging can easily be added to the mix by incorporating an emission filter to the camera imaging path, taking advantage of the narrow linewidth and high brightness of VCSELs as fluorescence excitation sources.
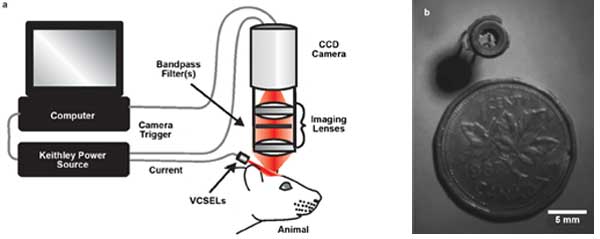
Figure 1. (a) Imaging system. (b) Example of three VCSELs (at 680, 795 and 850 nm) inside a miniature package. Images courtesy of Dr. Ofer Levi, University of Toronto.
LED sources are a possible illumination solution, but VCSELs offer further advantages over LEDs and over edge-emitting laser diodes, including low cost, very high efficiency, high brightness and rapid response in scenarios with rapid on/off cycles. Due to their small gain volume, VCSELs can reach thermal stability within microseconds following a current change, allowing stable tracking of tissue dynamics.8
System optimization, current performance
My colleagues and I – in collaboration with our industrial partners QImaging of Surrey, British Columbia, and Teledyne Dalsa of Waterloo, Ontario – have developed a prototype VCSEL-based brain imaging system. Our tabletop CCD-based imaging system includes a 14-bit electron multiplying CCD camera from QImaging that operates at speeds greater than 60 fps at 512 × 512 image size. We also integrated 680-, 795- and 850-nm VCSEL sources from Vixar of Plymouth, Minn., that enable fast switching between high- and reduced-coherence illumination. The system uses intrinsic optical signal imaging and LSCI techniques to measure tissue oxygenation and blood flow simultaneously.9

Figure 2. Multimodal imaging during a seizure in a rat’s brain: This image series shows relative changes in deoxy-Hb (HbR), indicated in reflection changes in 680 nm (top row); fluorescence changes from an OGB1-AM calcium indicator (middle row); and relative changes in blood flow (bottom row). The seizure onset occurred after the first three images were recorded (interval between images = 7 s).
The high sensitivity and speed enabled by this multimodal system were demonstrated in tracking an ischemic stroke. Figure 1a shows the schematics of the imaging system. The miniature illumination module (Figure 1b) includes the three VCSELs in one small package.
In another study, the high dynamic range and fast camera response were used in collaboration with Dr. Peter Carlen’s lab at the Toronto Western Hospital to track optical and electrical changes simultaneously during a seizure.
Figure 2 shows our initial studies, where blood flow (measured by LSCI), deoxyhemoglobin (deoxy-Hb) concentration changes (measured by IOSI) and neural activity (measured by Ca2+ fluorescence) were tracked simultaneously. The onset of the seizure was established through electrophysiology recordings made in parallel to the optical imaging. The first three images in the time series were recorded before the seizure onset.
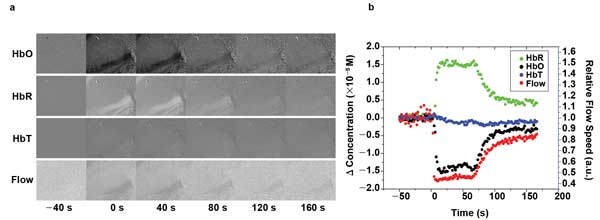
Figure 3. Multimodal imaging with a 10-bit CMOS camera during an induced ischemic stroke. (a) The image series show relative changes in oxy-Hb (HbO, first row), relative changes in deoxy-Hb (HbR, second row), relative changes in total-Hb (HbT, third row), and changes in the relative blood flow velocity (Flow, fourth row). The illumination was provided by alternating between 680-, 795- and 850-nm VCSELs. The region of interest is 1.5 × 1.5 mm. (b) Time course of the observed signals from (a) spatially averaged in a vein, inside the region of interest.
The observed increase in neural activity after the onset of the seizure (as shown by the fluorescence Ca2+ indicator in the last two images of Figure 2) was accompanied by an increase in the cerebral blood flow and an increase in deoxy-Hb concentration (shown by a decrease of reflection at 680 nm) in the tissue. This suggests that large metabolic changes occur during seizures and play a major role in the dynamics of the seizure, in agreement with previous observations. Our multimodal imaging approach will support correlating the spatiotemporal metabolic changes to the spatiotemporal changes in neural activity.
One of our objectives is to enable multimodal optical imaging studies in freely behaving, moving animals. We have also developed a low-cost, portable CMOS-based imaging system using Teledyne Dalsa’s Genie HM1400-XDR 10-bit camera, which operates at speeds greater than 100 fps at 512 × 512 image size. The dynamic range and image quality of the miniature imaging system proved sufficient to track the dynamics of an ischemic stroke as shown in Figure 3.

Figure 4. (a,b) A head-mounted multimodal imaging system is demonstrated on a freely moving rat. (c) Relative-blood-flow velocity map recorded from the sensory cortex of the rat brain.
Combining the miniature camera and optics with a fast VCSEL-based illumination source has resulted in a compact imaging system that can be mounted on the head of a moving rat, as shown in Figure 4. Figure 4c shows an example of a relative-blood-flow velocity map, acquired from a moving rat. Our current prototype physical dimensions – 15 g in weight, 40 mm in height – are suitable for conducting imaging studies on rats. Further weight and size optimization will enable adaptation to imaging (smaller) moving mice, as demonstrated recently in portable fluorescence imaging.10 Our multimodal studies focus on evaluating epilepsy and stroke animal disease models in awake, moving rats, providing a path to a portable, multimodal, anesthesia-free and minimally invasive investigation of many brain diseases.
Outlook
Multimodal brain imaging opens many opportunities to study diseases such as epilepsy, stroke and traumatic brain injury. With their agile operation and fast response times, VCSEL-based light sources can enable such important research. Using fast, label-free recording of metabolism (blood flow, oxygenation levels) simultaneously with optional fluorescence imaging of embedded fluorescent markers, researchers can accurately study the progression of disease and the recovery process in real time.
We foresee great advantages in combining real-time optical imaging with electrical or optical stimulation (through optogenetic tools, for example) and with drug delivery. This will enable physicians to observe seizures while concurrently administering therapies by applying a stimulus to inhibit the developing seizure,11 and to evaluate in real time the dose and effects of delivered medications, thus accelerating the recovery process in the case of traumatic brain injury and stroke.
Meet the author
Dr. Ofer Levi is an assistant professor in the Institute of Biomaterials and Biomedical Engineering and the Department of Electrical and Computer Engineering at the University of Toronto, Toronto, Canada; email: [email protected].
Acknowledgments
The author would like to acknowledge the support of the Natural Sciences and Engineering Research Council of Canada/Canadian Institutes of Health Research, the contributions of Suzie Dufour, Yaaseen Atchia, Dene Ringuette, Iliya Sigal and Raanan Gad, and the collaboration efforts with QImaging, Teledyne Dalsa, Vixar, and Dr. Peter Carlen and Dr. Bojana Stefanovic.
References
1. P. Osten and T.W. Margrie (2013). Mapping brain circuitry with a light microscope. Nat Methods, Vol. 10, pp. 515-523.
2. E.M. Hillman (2007). Optical brain imaging in vivo: techniques and applications from animal to man. J Biomed Opt, Vol. 12,
p. 051402.
3. V. Marx (2012). Rendering the brain-behavior link visible. Nat Methods, Vol. 9, pp. 953-958.
4. H.-U. Dodt et al (2007). Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nat Methods, Vol. 4, pp. 331-336.
5. R.D. Frostig, ed. (2009). In Vivo Optical Imaging of Brain Function. CRC Press, Boca Raton, Fla.
6. P.B. Jones et al (2008). Simultaneous multispectral reflectance imaging and laser speckle flowmetry of cerebral blood flow and oxygen metabolism in focal cerebral ischemia. J Biomed Opt, Vol. 13, p. 044007.
7. E.A. Munro et al (2011). Multi-modality optical neural imaging using coherence control of VCSELs. Opt Express, Vol. 19, pp. 10747-10761.
8. N. Parashurama et al (2012). Continuous sensing of tumor-targeted molecular probes with a vertical cavity surface emitting laser-based biosensor. J Biomed Opt, Vol. 17, p. 117004.
9. H. Levy et al (2012). Rapid monitoring of cerebral ischemia dynamics using laser-based optical imaging of blood oxygenation and flow. Biomed Opt Express, Vol. 3, pp. 777-791.
10. K.K. Ghosh et al (2011). Miniaturized integration of a fluorescence microscope. Nat Methods, Vol. 8, pp. 871-878.
11. J. Tonnesen et al (2009). Optogenetic control of epileptiform activity. PNAS, Vol. 106, pp. 12162-12167.