Three imaging companies discuss the factors driving the market for adaptive optics for biological applications, and offer a look at where the field is going.
Adaptive optics started off looking out to the sky, helping astronomers correct for distortions caused by the Earth’s atmosphere – but the technology has since found additional applications of a more inward nature: in biology, where it helps researchers correct for the distortions they encounter as they send light into tissue.
To take the pulse of the bio market for adaptive optics, BioPhotonics spoke with representatives of three companies in the field: Michael Feinberg, director of sales and marketing at Boston Micromachines Corp. (BMC) in Boston; James Joubert, applications scientist at Photometrics in Tucson, Ariz.; and Christian Theriault, president and CEO of Tag Optics Inc. in Princeton, N.J.

The Multi-DM is a versatile, robust deformable mirror system for advanced wavefront control, available in both continuous and segmented surfaces for adaptive optics or spatial light modulator applications. Courtesy of Boston Micromachines Corp.
Boston Micromachines produces high-resolution MEMS deformable mirrors for biological applications including confocal, two-photon and standard optical microscopy. Photometrics designs and manufactures scientific cameras for bioimaging, offering high capture rates and sensitivity; these cameras are suitable for both wavefront sensing and image capture. Tag Optics offers an adaptive lens that changes focal length in submicroseconds; it allows the researcher to create 3-D Z-stacked images instantly without any moving parts.
Q: What do you see as the “next big thing” in adaptive optics for bio, in general? Are you seeing any new and exciting advances coming out of R&D and/or university labs?
Theriault: Adaptive optics (AO) technologies, which enable the extraction of depth information (such as 3-D volumes), are starting to come of age and provide benefits across many aspects of research. In biological imaging, devices like ours can be added to imaging modalities such as optical coherence tomography (OCT) to provide greater control over optical biopsies. For researchers using multiphoton microscopy for studies of neuronal pathways, these devices allow real-time studies to be undertaken. Unlocking crucial in situ information from a multitude of depths is where adaptive optics technologies provide a key capability that may help enable future researchers and clinicians to see phenomena they could never observe before.
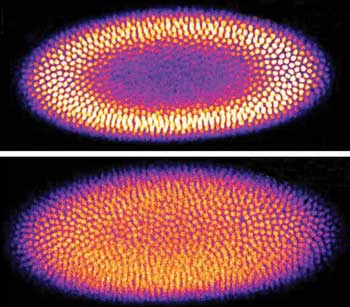
Multiphoton images of a developing Drosophila embryo captured with and without the Tag Lens. Imaging without the Tag Lens provides an optical slice of the sample; the Tag Lens enables the acquisition of a more global view of the sample by increasing the depth of field of the optical system. Courtesy of Tag Optics.
Another branch of adaptive optics that is attracting increasing attention revolves around new autofocus-related technologies. These have significant advantages over their mechanical counterparts, as they have no moving parts and add much-needed speed to the process. These enabling AO technologies are of particular importance in high-throughput, time-sensitive automated systems such as those used in pathology.
Feinberg: For the next big thing in adaptive optics for bio applications, we look to two fronts: multiphoton microscopy and retinal imaging.
For two-photon applications, there has been exciting work on the R&D front in a handful of universities around the world as well as at BMC in what has been termed superpenetration multiphoton microscopy. This technique extends the depth-of-imaging capability twofold and, due to higher-speed correction capability, will enable in vivo imaging. This is enabled by the use of a high-actuator-count wavefront corrector which can operate at kilohertz speeds. This research is going on at such locations as Janelia Farm at the Howard Hughes Medical Institute, Institut Langevin at the CNRS in France and at Boston University, with whom we are collaborating.
On the retinal imaging front, many would argue that the next big thing has already arrived in vision science, as scanning laser ophthalmoscopes (SLO) and OCT systems have been outfitted with adaptive optics for over a decade. However, this capability has been confined strictly to research applications. Recently, the technology has been adopted by a handful of clinical researchers who are attempting to prove its applicability in diagnosing conditions and evaluating the efficacy of drug therapies. This has been carried out by many researchers in the field, two of which are located at UCSF and the Joslin Diabetes Center here in Boston.
Joubert: I see wavefront sensing and correction being applied to a variety of dynamic microscopic techniques deeper into tissues, particularly in neuroimaging, through a combination of new bioimaging tools and approaches. An example is [the work of Dr. Joel Kubby at the University of California, Santa Cruz], which combines fluorescent protein guide stars with a highly sensitive EM [electron multiplying] CCD-based wavefront sensor and a laser-scanning confocal microscope to image brain tissue. The combined setup improved contrast and created a 3× improvement in signal-to-noise ratio compared to standard confocal imaging.

Electron multiplying CCD cameras can be combined with fluorescent protein guide stars and a laser-scanning confocal microscope to image brain tissue. Courtesy of Photometrics.
Kubby made further advances in a wide-field microscope by using a high-speed, high-sensitivity EMCCD camera for image acquisition and applying interferometric focusing, instead of traditional geometric focusing, of excitation light onto the sample’s guide stars. This increased the potential imaging depth into tissues by reducing light loss due to scattering and doubling the signal-to-noise ratio.
Q: How would you say the market has been in the past few years for adaptive optics (for bio)? Where do you think the market is going?
Joubert: Adaptive optics techniques for bioimaging have been used in retinal imaging for some time and are now being applied to an expanding list of microscopy methods, most often to those also intended for imaging thicker tissue samples. We see confocal and multiphoton microscopies benefiting from adaptive optics as well as techniques more recently gaining interest, such as light-sheet microscopy, stimulated emission depletion (STED) superresolution microscopy, structured illumination microscopy (SIM), second-harmonic generation and optical coherence tomography.
As more research is aimed at imaging in more native environments, more microscopy methods are being used on tissues, organs and whole animals. As a result, these techniques are adapting to the challenges of imaging thick samples. The development of clearing agents, such as [the] recently published CLARITY, is allowing even deeper tissue imaging by reducing the scatter and absorption caused mostly by the lipid membranes of the nucleus and organelles, but the differences in refractive index and sample homogeneity through such depths still require the correction capabilities of adaptive optics. Even 3-D localization superresolution techniques such as PALM and STORM will incorporate and take advantage of adaptive optics.
Feinberg: The market has been steady over the last few years for bio applications, as the need has been able to sustain quite a few players in the market. I personally think that the market will eventually shift over to a more OEM instrument-type market, away from the laboratory-built atmosphere that has persisted for more than a decade.
Theriault: The market is definitely on the rise. As with any novel technology, I think a lot of customers are waiting to see hard data indicating the benefits of AO to their specific applications. Those kinds of studies are beginning to be published, so I fully expect that the market will see even stronger growth in the coming years, as early AO technology adopters continue publishing their results.
Q: What are the biggest challenges to new advances in adaptive optics for bio?
Feinberg: The biggest challenge to new advances in AO is clinical relevance of the technique. While researchers can sustain an industry, a mass-market application is needed to enable companies to push the technology to the next level. These advances include higher-resolution correction and higher-speed operation.
Joubert: Adaptive optics of the future will require rapid, on-the-fly analysis of changes in the imaging environment and making necessary corrective adjustments. For biological imaging of rapidly changing events within cells or across tissues, these measurements, analyses and adjustments will need to be made more rapidly, pushing the limits of current hardware sensitivity and speed as well as software processing speed and efficiency. The development of new approaches to measuring distortion will also help overcome these challenges.
Theriault: It’s interesting, but I think the biggest challenge is not a technology one. For users, I think the biggest challenge is education. Many researchers and clinicians still are only becoming aware of the potential benefits of AO for their work. There are many new applications yet to be explored.
Only recently are we seeing the kind of user/corporate collaboration that helps advance the more rapid general acceptance of new scientific technologies. Because optics can have very long life cycles of a decade or more, change in this field can sometimes be slow. Now, as more research papers are being published, we are seeing vastly more demand. Out of that demand and the specific needs of the next generation of users will come the next advances in AO.
Q: And which application areas would you say are thriving – and why?
Theriault: Optical biopsy and other applications necessitating advanced depth control and 3-D data acquisition have taken off in recent years. In fact, both OCT and multiphoton imaging have seen phenomenal growth and are the subject of much research themselves. AO systems are thriving in both of these areas.
In addition, although still in its infancy, focusing technologies based on adaptive optics have also taken off in the past few years, as users have come to appreciate their advantages over mechanical stages. While mechanical stages are slow and require much closed-loop feedback control, autofocus/AO devices with no moving parts create fast and accurate focusing, helping users acquire usable 3-D data from living specimens.
Joubert: Applications which involve more homogeneous samples that are easier to correct over a wide area or which are more established, such as retinal imaging, can most easily take advantage of adaptive optics. However, any imaging application that involved multidimensional imaging, particularly of thicker samples, can and will take advantage of adaptive optics. However, the higher the speed, the deeper the tissue and the more inhomogeneous the sample, the more difficult it is to apply adaptive optics correction.
Feinberg: I think that the definition of “thriving” is relative. With this in mind, the application that seems to be the steadiest and most dependable for future use is retinal imaging. A decade ago, research using SLO and OCT systems commented in great detail on the type of AO system being used for each research endeavor. Now, it isn’t much more than a footnote.
This is what the industry has been striving for: that the AO is just another feature of the system and that, in the end, it just gives a better image that allows researchers to make more discoveries in their field.