Dr. Gerhard Holst, PCO AG
Biologists use high-speed imaging to study how microorganisms move.
Some like it hot – for example, micro-organisms that live in the deep sea on “black smokers,” where heat rises from the interior of the earth. These archaea, or “ur-bacteria,” form a separate branch in the phylogenetic tree of life and are veritable survival artists: They become active only at temperatures of 75 °C or higher, and thrive at temperatures between 80 and 100 °C. To find the temperate zone between the 400 °C water coming from inside the earth and the cold sea water with a temperature of just 3 °C, they roam about, clinging to stone that offers a suitable habitat in terms of temperature and food supply. Biologists have long wondered how exactly it is that archaea propel themselves.
The microorganisms move rapidly through hot water and cling to stone using tiny appendages on the cell surface that function as “oars” and “sticky feet.” And a high-speed, highly sensitive camera is helping biologists at the University of Regensburg in Germany examine exactly how these cell appendages function.
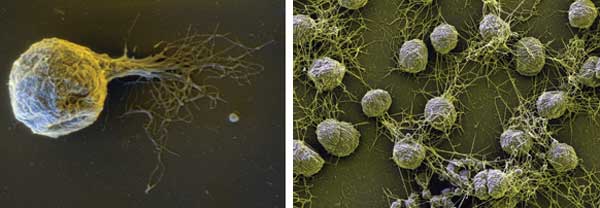
Methanocaldococcus villosus moves with the help of more than 50 flagelli. Images courtesy of Gerhard Wanner, Ludwig Maximilian University of Munich.
It is known from electron microscope images that the organisms on the cell surface have flagella, which are used for motility and presumably also for adhesion. Professor Dr. Reinhard Wirth of the university’s Institute for Microbiology wants to know more about this and has been examining these amazing microorganisms for many years. To understand how they move, his team designed a thermal microscope. The mechanical stage can be heated to 95 °C, a temperature conducive to the well-being of the archaea. The objectives also are heated because, otherwise, it would not be possible to attain the necessary temperature.
A fast camera for fast ‘ur-bacteria’
An important element in the experiment is the camera. The cell appendages used by the archaea for motility are just 15 nm long, which means that they cannot be directly observed under a conventional microscope. Therefore, they are colored with a fluorescent dye and analyzed with a fluorescence microscope. As it turns out, however, of the 46 archaea species examined, only one – Methanothermobacter thermautotrophicus – can be dyed, and even this one emits only weak fluorescence. The camera therefore must be very sensitive, but also must have a high scanning frequency to follow the fast-swimming microorganisms.
The researchers selected PCO AG’s pco.1600 for the task, based on its image quality as well as on the company’s geographic proximity to the University of Regensburg (less than half an hour). Wirth’s team has been using the camera since 2007, and that proximity has proved useful, especially during an incident that occurred when a student accidentally fouled the lens with metal filings. The camera was taken to PCO, where the filings were removed in half an hour at no charge. The biologists had borrowed the camera originally, tested it extensively, then paid for it with funds from the German Research Institute.
The camera supplements the thermal microscope in observing the archaea, which, despite their small size, are extremely fast. Pyrococcus furiosus – the “racing fireball” – can travel 200 times the length of its body in just 1 second, and some archaea can even reach speeds of up to 600 body lengths per second. In comparison: The fastest a human being can run is five body lengths per second. The most popular “normal” microorganisms in biology labs are halobacteria and the intestinal bacterium E. coli, which can move at 10 and 40 µm/s, respectively. Only a camera with a frame rate of 100 images per second is fast enough to continuously film microorganisms at such speeds. Based on the magnification scale of the microscope, it is possible to calculate the distances traveled by the archaea from one frame to the next.
Microorganisms with variable speed
The microbiologists discovered two “swimming styles” with their apparatus. First, the microorganisms jet through the water at high speed, traveling long distances in the search for a suitable living environment in their biotope. It is assumed that they use the temperature gradient for orientation. Once they have found a suitable location, they “shift gears,” swimming at one-tenth their original speed in zigzag patterns, apparently searching for a place to cling to the wall of the test tube. The team found that the archaea use the same organelles for both types of motion as well as for adhesion.
The researchers’ work is purely basic, Wirth said. Nevertheless, it provides potentially economically valuable findings, such as the adhesive on the ends of the organelles used by the archaea to hold onto surfaces. The adhesive remains sticky even in boiling water – a property with potential for many technical applications; e.g., gluing silicon in microchips that become hot during operation.
Avoidance of oxygen
In future experiments, the investigators want to find out to which sensory stimuli the archaea actually respond. Presumably the answer will be temperature, but it also could be nutrients or toxic substances; e.g., the archaea do not breathe oxygen – it is actually toxic for them – and initial findings show that the organisms swim much faster when forced to avoid oxygen.
To research this further, the team is building a new test apparatus that, in addition to the heating capability, also contains zones with different oxygen content and that generates different fluid flows simultaneously within the glass container. High water pressure such as that in the deep sea is not necessary because the bacteria also thrive at the normal pressure in the lab. For the next phase of research, the team plans to lease another high-speed, high-sensitivity camera, Wirth said.
Future experiments also will examine the cell appendages that do not serve the purpose of motility. They are hollow and could possibly be used to transport substances, but this is only a theory at present. One thing is certain: The complexity of the experiments will require an even faster camera with even higher sensitivity.
Meet the author
Dr. Gerhard Holst is head of the research department at PCO AG in Kelheim, Germany; email: [email protected].