Gabrielle Crétot-Richert and Myriam Francoeur, NüVü Cameras
The prospects of photon-counting imaging are as diverse as they are promising. From space-debris monitoring, to telecommunication satellites and shuttle missions, to the advent of powerful surgical tools, which could help cancer patients through the precise removal of malignant tissues, its applications are numerous and expanding.
Photon counting reveals its strengths in extremely low-light conditions, where other imaging techniques fail to provide valuable data. This is true for all things infinitely distant or infinitely small.
Instead of measuring a source’s incoming light intensity, photon counting detects the presence of sparse photons over a large collection area. Setting a proper detection threshold is key for achieving high sensitivity.
As its name openly suggests, photon counting simply comes down to counting every collected photon from an incoming source of light. A detector’s sensitivity is crucial in this endeavor; however, no perfect sensor exists, and such devices are subject to multiple limitations. Worse, light-emission processes follow a statistical model, which alters the detected signal. Thus, a perfect sensor yields a signal-to-noise ratio (SNR) given by SNR = S/√S, where S stands for the incoming signal, following a Poissonian distribution. The expression √S is commonly referred to as shot noise.
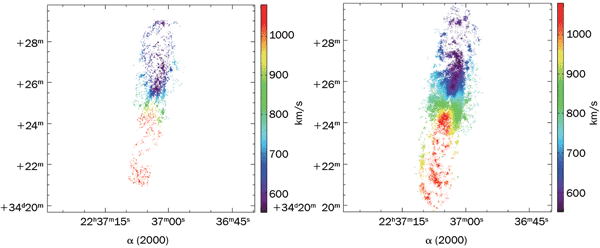
Figure 1. Radial velocity field of the spiral galaxy NGC7331 extracted from Fabry-Pérot imaging spectroscopy data without (a) and with (b) photon-counting imaging mode. Photo courtesy of Olivier Daigle, Observatoire du Mont-Mégantic.
Devices designed for photon counting must take into account three main limitations: photon-to-electron conversion, or quantum efficiency; amplification, or gain; and inherent noise sources from the sensor, which may drown out the actual signal transmission during photon-starving measurements. All limitations lower the SNR below its theoretical value, making single-photon events barely detectable in the best-case scenario. Noise sources must be handled rigorously; some can be altogether suppressed.
Before photon-counting imaging, photomultiplier tubes (PMTs) and photodiodes were used for photon-starving measurements as single-pixel detectors. The former achieve high gain through electron multiplication in a vacuum tube. PMTs are subjected to a low level of noise and exhibit faster response time while operating with large collection areas. However, they operate at high voltages (about 2000 V), raising the risk of short circuits, and their quantum efficiency hardly reaches 30 percent. The latter are small devices with increased quantum efficiency lying between 60 and 80 percent, but possess smaller sensitivity and collection areas.
Combining photon counting with imaging naturally occurred by merging CCD and PMT technologies about 20 years ago with intensified CCD, or ICCD. In such devices, photons are converted to electrons, which will then pass through a photomultiplier tube, hence increasing gain. Electrons are thereafter converted back to photons on a scintillating surface coupled to a CCD via optic fibers. Like PMTs, ICCDs operate at high voltage, and their design is complex and requires fine-tuning. Yet they achieve the greatest image rates in the market of all high-sensitivity sensors because of their gating architecture.
EMCCDs, another CCD-based technology, appeared a few years after ICCDs. These sensors incorporate an electron-multiplying (EM) register for higher gain, yielding negligible readout noise. However, stochastic by nature, the avalanche multiplication process taking place in the EM register introduces what is known as the excess noise factor, which has the same effect on the SNR as halving the quantum efficiency. Photon-counting imaging with EMCCDs comes to the rescue through the binary assessment of each pixel’s photon detection using an electrical threshold. Such treatment eliminates the excess noise factor inherent to the EM process and gets rid of any base-level background (Figure 2). The resulting images are clearer, sharper and better contrasted, thus considerably increasing the reliability and accuracy of any observed phenomena.
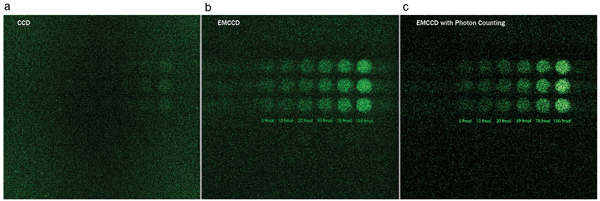
Figure 2. Microplate imaging with varying ATP (adenosine triphosphate) concentrations from 5 to 156 femtomoles. (a) CCD: 30-s total exposure time, single-frame acquisition. (b) EMCCD acquisition: gain of 5000, 5-s total exposure time, five frames stacking. (c) EMCCD with photon counting: gain of 5000, 5-s total exposure time, five frames stacking. Photo courtesy of Réjean Lebel, Centre hospitalier universitaire de Sherbrooke.
Technical challenges
Several factors must be considered to achieve the clearest images in photon-counting imaging. Basically, it is all about increasing the SNR closer to its theoretical value. On the one hand, quantum efficiency must be very high, over 90 percent, and the quality of the optical components directing light onto the detector must be excellent. On the other hand, electron amplification boosts the signal’s amplitude, rendering the readout noise negligible. Unfortunately, it also amplifies noise-induced electrons just like any other transiting photoelectrons.
As with CCDs, an EMCCD’s SNR is sensitive to three main sources of noise: dark current, charge-transfer efficiency and clock-induced charges. Atoms in the silicon lattice are excited by heat and generate dark current, which may generate false signals. After detection, photoelectrons will travel through the chip’s registries, where the signal may undergo losses, depending on the efficiency of the charge transfer. Finally, clocks driving the sensor readout also inject new electrons in a stochastic manner, creating clock-induced charges that are the dominant noise source for short exposures. Quality and design of the camera’s electronics are key in decreasing these noise sources.
Groundbreaking applications
Faint flux imaging via photon counting is achieving innovative application goals – and, with just the tip of the iceberg in sight, the possibilities seem endless. Each research avenue is uncovering new potential that can be beneficial for astronomy, and biomedical and now medical purposes.
In biomedical research, the study of molecular complexes is of paramount importance to understand the body’s physiological functions and its pathological behavior. Isolating single molecules and examining them in vivo helps to identify and characterize the proteins making up a complex, and their interactions. Dr. Paul Maddox, formerly of the Institute for Research in Immunology and Cancer in Montreal, developed a confocal superresolution microscope that combines powerful Nikon optics and an EMCCD camera suited for extremely low-light applications. Performed through total internal reflection fluorescence (TIRF) microscopy, single-molecule imaging uses a laser to extract information from the molecular complex. In photon-counting mode, Maddox detected single fluorescently labeled biomolecules and determined their conformational states, mixture compositions and interactions.1 The added sensitivity of the camera allowed for another major improvement in this application: While traditional TIRF microscopy calls for high-intensity lasers to trigger molecular fluorescence, EMCCD photon counting is so sensitive that researchers can choose less powerful light sources for their studies (Figure 3). This improvement increases not only the accuracy of TIRF microscopy, but also the viability of observed cells under the microscope. Cell biology and pharmacology will greatly benefit from such advancements.

Figure 3. Single biomolecules imaged through TIRF microscopy in (a) conventional acquisition mode with a laser power of 100 percent, and (b) photon-counting mode with a laser power of 20 percent. Photo courtesy of Dr. Paul S. Maddox, University of Montreal’s Institute for Research in Immunology and Cancer.
Furthermore, fluorescence is becoming a powerful tool in surgery, especially oncology. For treatment, tumor resection is inevitable, and its success strongly increases a patient’s odds of survival. But some cancerous tissues are harder to operate on than others – brain tumors, for example, where complete yet selective eradication of tumors is challenging at best. Although the bulk of a tumor is generally detectable by sight, invasive cancer cells can penetrate tissues without altering the latter’s external appearance. Fluorescence biomarkers can selectively bind to malignant cells, and a high-sensitivity
detector reveals them in rich detail. To this end, Dr. Frédéric Leblond of the École Poly-technique de Montréal, in collaboration with McGill University Health Center, is performing clinical trials on brain tumor resections using a Zeiss surgical microscope coupled with a high-capacity EMCCD camera.2 The results are promising: The system, operating in photon-counting mode, can detect the faintest amount of fluorescence emitted in real time. Thus, fluorescence microscopy enhances the precision and efficiency of tumor removal, and the chances of recurrence are greatly diminished, hence helping to save lives.
Astronomy applications also benefit from these leaps in sensitivity; one even relates to our modern lifestyle. Since the famous Sputnik 1 launch in 1957, several thousand satellites and other man-made objects have been put into orbit, revolving around the Earth in a matter of hours. Today’s way of life highly depends on these satellites for uninterrupted telecommunications services and high-precision tracking networks. But satellites have expiration dates: They do not possess limitless energy supplies and shut down after a few years of operation. Furthermore, these devices are subject to extremely harsh environments and sustain irreparable damage due to cosmic rays or colliding objects, sometimes resulting in malfunctions. As they deteriorate, orbiting satellite remains create “space debris” in a cascading effect that may jeopardize other space infrastructures.

Figure 4. Imaging system integrating high-sensitivity cameras with neurosurgical microscopes to superimpose the image of the tissue with its molecular spectroscopic signature. Photo courtesy of McGill University Health Center.
The satellites and debris are small, faint objects that travel through space at velocities ranging between 3 and 9 km/s. Many countries have put effort into developing space surveillance projects and even satellite servicing that would remove debris or reduce the current amount in orbit. EMCCD cameras are adapted to this task, as they possess all the required features for the proper detection of decommissioned satellites from Earth, or space debris itself from above. Operated in photon-counting imaging mode, EMCCDs can differentiate between the faintest events, thanks to their extremely low noise. Better still, they can achieve hundreds of frames per second, making real-time tracking of a target possible. The camera’s shorter-exposure capability combined with photon counting allows us to distinguish moving objects from the background by virtually eliminating its signal. Regular cameras would instead collect extra photons from the background, thereby producing images of lower contrast. EMCCD technology will undoubtedly represent an important enhancement for ground satellite-monitoring projects3 as well as space-based instrumentation, pushing further the frontiers of our observable universe.
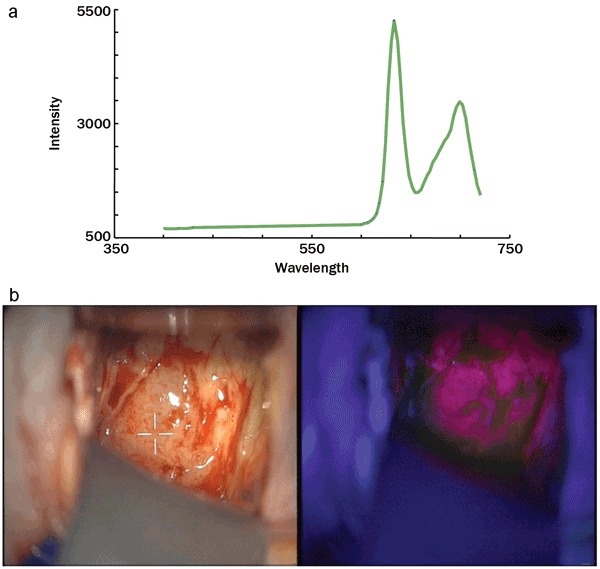
Figure 5. (a) Spectroscopic signature of the fluorescent marker. (b)
Brain tissue images with the neurosurgical microscope, and (c) the
added marker spectrum. Photo courtes of Yoann Gosselin, École
Polytechnique de Montréal.
At the crossroads between science and engineering, photon-counting imaging is still an evolving technique that lets us foresee an exciting future. Alongside its increasing progress, new needs will arise, and the technique will only grow in efficiency as it matures. New lines of EMCCD cameras optimized for photon-counting imaging are now being developed, namely the 4K × 4K detectors. In addition to high sensitivity and frame rates, these cameras will offer larger fields of view suited for integral field spectroscopy or differential photometry in the field of astronomy, as well as larger-body imaging of complete human organs for a more comprehensive assessment of pathologies.
Meet the authors
Gabrielle Crétot-Richert is a biomedical scientist at Nüvü Cameras; email: [email protected]. Myriam Francoeur is an application scientist at Nüvü Cameras; email: [email protected].
References
1. A. Padeganeh et al (January 2012). Improved single-molecule imaging based on photon counting with an EMCCD camera. Biophys J, Vol. 102, Issue 3, p. 480a.
2. Y. Gosselin et al (February 2014). Through-microscope spectroscopic excitation and emission for fluorescence molecular imaging as a tool to guide neurosurgical interventions. Proc SPIE, Vol. 8928-A, 8928-9.
3. O. Djazovski et al (October 2013). Electron-multiplying CCDs for future space instruments. Proc SPIE, Vol. 8915, p. 89150Q.