The technology could advance a range of neuroscience applications.
As a postdoc in the early 2000s, Dr. Ruth Empson worked with one of the first multiphoton microscopy systems in Britain. The technology was robust, she said, but handling it could sometimes prove a bit of a challenge – especially for a biologist like herself, who didn’t have the technical background of the physicists who had developed it and who, at the time, made up much of its user base.
Today, Empson has again turned to multiphoton microscopy. Now an associate professor in the department of physiology at the University of Otago in Dunedin, New Zealand, she played a key role in bringing the very first multiphoton microscopy system – an A1R system from Nikon – to the relatively remote country. And she’s found that working with the technology is a very different experience than when she was a postdoc.

Researchers at the University of Otago in Dunedin, New Zealand, recently brought the first multiphoton microscopy system to the country. Courtesy of Ruth Empson, University of Otago.
“The technology has moved on tremendously since then. It’s more turnkey and better, hugely better,” she said, drawing out the first syllable in “hugely” to emphasize just how much more impressive it is. She and colleagues are using the system to explore neural connectivity, with an emphasis on reconnectivity after stroke. “The stuff we were doing 15 years ago was nowhere near as good as what we’re doing routinely now.”
Advances in the technology and the introduction of more turnkey systems are pushing multiphoton microscopy ever further into the mainstream of biomedical research – especially in the neurosciences. Thanks to advances in speed and penetration depth, the approach is offering significant new insights into how the
brain processes information. Some researchers are beginning to look at how it can contribute to the management and treatment of neurological disorders and neurodegenerative diseases.
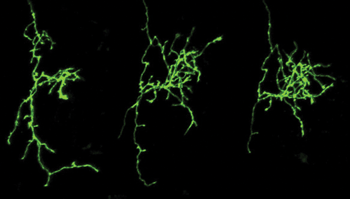
Using multiphoton microscopy, researchers have shown how nerve cells in the brain can use sensory stimuli to rewire and refine themselves by forming new connections with neighboring cells. Shown here is a time-lapse image (Day 1, Day 2, Day 3) of normal growth and refinement of a GFP-expressing retinal ganglion cell axon terminal in the optic tectum of a living Xenopus tadpole. Courtesy of Aydin Tavakoli/Ruthazer Lab, McGill University.
In the research realm, multiphoton microscopy has been helping to shed light on neural circuitry and how the brain is wired together. In a recent study performed at McGill University in Montreal, for example, the technique played an integral role in showing that nerve cells in the brain can use sensory stimuli from the environment to rewire and refine themselves by forming new connections with neighboring cells. The study was reported in the May 2014 issue of Science.
The study’s senior author, Dr. Edward Ruthazer, an associate professor of neurology and neurosurgery at the Montreal Neurological Institute at McGill, said that ever since he began studying neuroscience, he’s been fascinated by the idea that sensory experience and neural activity can alter the structure and connectivity of brain cells.
This idea can be traced to classic experiments by Nobel laureates Dr. David Hubel and Dr. Torsten Wiesel studying the development of the visual system, “but over the intervening decades, for me the holy grail has been to be able to actually watch such circuit rewiring occur in real time,” Ruthazer said. “It is only in the last decade that technology has evolved to the point that this is now possible.”
The Science study confirmed for the first time in real time that synchronous firing of cells in the brain leads to strong, stable neural connections, while asynchronous firing actively weakens connections (A surprising finding was that it also results in increased formation of nerve cell branches seeking connections with other cells).
The researchers achieved this by imaging nerve cell branch formation in the transparent Xenopus tadpole while stimulating the specimen’s eyes.
However, the single most important breakthrough in the study came from the “good old-fashioned approach of simply being observant and thoughtful,” Ruthazer said – the student carrying out the experiments had the insight that a mistargeted ipsilateral axon he had noted in a small fraction of the animals could be used to explore the role of firing in axonal development.
State-of-the-art technologies used in the study included an Olympus FV300 confocal microscope (which Ruthazer and colleagues custom-converted into a two-photon system), as well as a Thorlabs commercial resonant scanner system. The systems shared a Spectra-Physics Mai Tai BB laser.

The researchers plan to use the money that accompanied the prize to invest in a new two-photon microscope. This will help them gain new insights into how grid cells interact, said Dr. Edvard Moser. Image courtesy of Geir Mogen/ NTNU.
The work reported in the Science paper could have important implications, even beyond the better understanding of brain circuitry that it’s already provided. The wiring and rewiring in the animal models that the investigators are using follow rules “remarkably similar” to those in the human brain, exhibiting the same kinds of experience-dependent plasticity.
“Our work, by figuring out in detail what those rules are, offers hope in situations where miswiring of neural circuits needs to be corrected,” Ruthazer said. They now recognize that it should be possible to use certain patterns of neural firing to destabilize and remodel incorrect connections, and to stabilize and consolidate appropriate ones. The potential applications of this new understanding include systematic, noninvasive treatment for autism and other neurodevelopmental disorders.
Into the hands of the masses
While multiphoton microscopy has tremendous potential for a broad range of applications, relatively few investigators have access to it. So what will it take to get multiphoton microscopy into the hands of the masses?
One of the obstacles to widespread adoption doesn’t involve collecting images, Ruthazer said, but rather analyzing them. He and colleagues collect dozens of 3-D structural images over a span of hours and
then have to painstakingly reconstruct them. Automating this process would save a considerable amount of time. “Unfortunately, most of the software currently on the market does not appear to do an adequate job compared with manual tracing,” he said.
And then, of course, the big one: cost. Turnkey multiphoton microscopy systems are pricey, sometimes costing upwards of a million dollars. That the purchase of a new system can still warrant a press release – as with the University of Otago – underscores the fact that this is by no means an everyday event. It takes more than just pocket change to buy one of these.

University of Otago investigators will use the system for a wide range of applications – for example, to explore neural connectivity after a stroke. Courtesy of Ruth Empson, University of Otago.
We are often reminded of how much a big-ticket item a system can be. Researchers Dr. Edvard Moser and Dr. May-Britt Moser, co-directors of the Kavli Institute for Systems Neuroscience at the Norwegian University of Science and Technology, were recently awarded the Körber European Science Prize. They plan to use the NOK 6.1 million (about US $1 million) they received with it to invest in a new two-photon microscope.
The prize recognizes the pair’s 2005 discovery of specialized neurons called grid cells – which, they demonstrated, facilitate precise spatial orientation in rodents. They have already made considerable progress in developing understanding of what these cells are and how they work, and others have explored the grid cell network in humans. But incorporating a two-photon microscopy system into their tool kit will help to advance the understanding of spatial orientation even further.

Dr. Edvard Moser and Dr. May-Britt Moser of the Kavli Institute for Systems Neuroscience at the Norwegian University of Science and Technology were awarded the 2014 Körber European Science Prize in recognition of their 2005 discovery of specialized neurons called grid cells, which enable spatial orientation in rodents. Image courtesy of Geir Mogen/NTNU.
“It will allow us to visualize the simultaneous activity of large populations of grid cells,” Edvard Moser said in a statement, “and thereby help us understand how groups of grid cells interact.”
Multiphoton microscopy systems can still be prohibitively expensive for many researchers, but in some ways, this is beginning to change. Prices have been falling in recent years, even as the technology has been improving by proverbial leaps and bounds. Just ask Empson, the University of Otago researcher, who has benefited considerably from this. “It probably took two and a half years to get the money together for our system,” she said. “But in that time, we actually got a better machine than we thought we could have at the start.”
She pointed specifically to the GaAsP detectors they now have.
These very sensitive detectors were critical for some of the things they wanted to do but originally were out of their price range. By the time they had raised the money they needed for their system, however, the detector had become affordable. Even more than before, the technology was within their reach.