Clémentine Bouyé, Tematys, and Carlos Lee, EPIC
Physicians, companies come together to address clinical translation issues.
It’s a long-standing truth: For the biophotonics community to meet the needs and constraints of the medical field and to translate technological advances for clinical use, it is necessary to improve communication and collaboration between technology providers and clinicians. After all, photonic technologies developed for clinical use must take into account the requirements of working in a medical area where time is short, space is precious, hygiene must be high, and resources can be few. Also, multidisciplinary developments can lead to better integration of a new technology into the surgery work flow.
To that end, EPIC (the European Photonics Industry Consortium) recently hosted a workshop at the Erasmus Medical Center in Rotterdam, Netherlands, on intraoperative assessment of tumor-resection margins. At the event, 70 participants – surgeons, pathologists, researchers, and small and large companies that specialize in photonic technologies for guided surgery – discussed the difficulties that surgeons and pathologists face in the delineation of margins during tumor resection, with the goal of determining how optical image-guided surgery can help assess tumor margins intraoperatively to reduce the number of local recurrences.
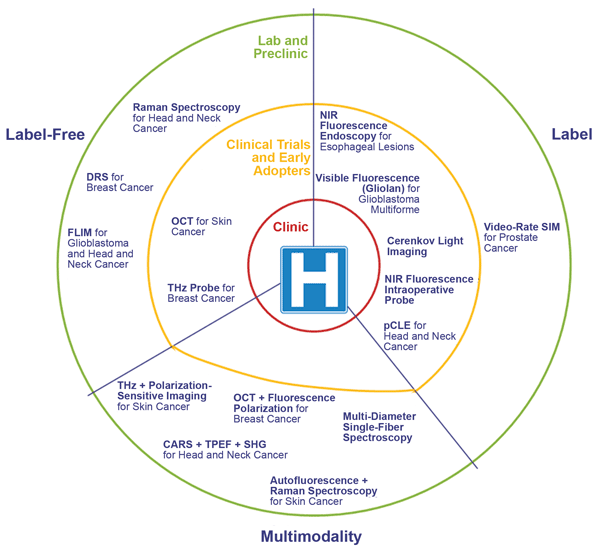
A pictorial representation of the maturity of the biophotonic technologies, presented in July at the EPIC workshop on intraoperative assessment of tumor-resection margins. ‘Label’ includes any technique that uses an external agent to allow and/or enhance the image. DRS = diffuse reflectance spectroscopy; FLIM = fluorescence lifetime imaging microscopy; THz = terahertz imaging; CARS = coherent anti-Stokes Raman spectroscopy; TPEF = two-photon excited fluorescence imaging; SHG = second-harmonic generation; pCLE = probe-based confocal laser endomicroscopy; SIM = structured illumination microscopy. Images courtesy of EPIC/Tematys.
To kick off the workshop, doctors presented the current challenge for tumor resection: making sure that all tumor tissue has been removed while the highest possible quantity of healthy tissues (nerves, blood vessels, etc.) is conserved. As tumors have blurred edges, a surgeon cuts around the tumor with a “safety margin” of a few millimeters (the so-called “clean margin”) to avoid local recurrences and metastasis.
Currently, margins are assessed by pathologists after resection (intraoperatively or not). In the case of inadequate margins (cut through tumor or too close to the tumor), the pathologists guide the surgeon to obtain a clean margin. Head and neck surgery, for example, sees inadequate margins 85 percent of the time. In breast cancer surgery, they are reported in 20 to 70 percent of cases, resulting in additional surgery. On the pathology side, an important issue is that it is not possible to assess the whole margin, as it is costly and time-consuming.
In terms of performance, high specificity and sensitivity are required, as well as sufficient penetration depth for tumor depth assessment. A solution for a precise evaluation of wide surfaces would be to scan a wide field first, to detect suspicious regions and then to microscopically analyze the identified area. Also, scanning and imaging will generate a huge amount of data; solutions to store and share all that data need to be investigated.

Surgeons and pathologists discussed their issues and needs with technology developers at EPIC’s workshop on intraoperative assessment of tumor-resection margins.
Surgeons and pathologists both underline the need for a tool that assesses all tumor margins objectively and in vivo to guide the surgery. Objectivity is important: Today’s evaluation of margins is highly subjective, based on each pathologist’s experience. A tool for objective assessment of margins intraoperatively (ideally, in vivo) would avoid wasting time and money, and improve patient wellness.
For example, a tool allowing an overlay of visible images and tumor-identifying images would be of great help in prostate resection surgery, said a urologist from Erasmus MC. In another example, an Erasmus MC pathologist explained how the department improved inadequate-margin rates by enhancing communication between surgeons and pathologists through regular meetings, systematic examination of resected specimens, and discussions on diagnoses and treatment options.
After the clinicians had spoken, companies and research groups presented a wide variety of photonic technologies for assessing tumor margins; these solutions ranged from label-free technologies such as terahertz imaging to fluorescence imaging and even multimodal techniques combining autofluorescence and Raman spectroscopy, for example. The variety arises because the general tumor-margin problem is the same across specialties, but each has its own issues. For example, in head and neck surgery, the structure is very complex, so getting the optical tool to the right place is tricky. In lumpectomies, the aim is to precisely delineate nonpalpable tumors while staying as noninvasive as possible for aesthetic reasons. Flexible endoscopes can be a solution.
In general, label-based technologies are currently closer to the clinic. Some – such as fluorescence-guided resection of glioblastoma multiforme, a rare and complicated brain tumor, with Gliolan – are already occasionally used by clinicians. However, as the workshop participants pointed out, huge challenges remain, including the lack of approved labels, as well as the long development time and cost required to get approved labels. Proposed solutions to limit the investment risk included a fluorescent marker based on an already approved drug, or Cerenkov light imaging that uses PET tracers as probes for tumor margin identification.
But label-free technologies are getting closer to the clinic, thanks to efforts to improve their performance. In the case of optical coherence tomography for the diagnosis of skin cancer, 3-µm resolution is already possible. Multimodal imaging methods might be less mature technologies, but they show great promise for a better assessment of tumor margins. One example is a combination of three imaging modalities: coherent anti-Stokes Raman scattering, two-photon excitation microscopy and second-harmonic generation. Each modality allows identification of the different elements present in a tissue (protein, lipids, keratin and so on) and thus enables precise localization of the tumor borders.
Looking ahead
The key theme of the workshop was that a multidisciplinary approach involving collaboration between clinicians and technology developers is necessary for the development and the implementation of new medical devices.
For European Union companies and clinicians, the Horizon 2020 funding program could help foster that kind of cooperation between technology and medicine. The program funds R&D collaborative projects that answer key societal challenges – such as cancer surgery – through a multidisciplinary consortium. Another EU initiative to promote collaboration between technology developers and users is the Responsible Research initiative. From this, the Van ’t Hoff Program for medical photonics was created, involving health foundations, hospitals, industries and research institutes.
At the workshop, one of the technology-side participants asked about technology adoption by clinicians: “Would you accept changing or adapting the way you work to integrate technologies?” The clinicians answered, without hesitation, “Yes, if it has a real added value.”
Meet the authors
Clémentine Bouyé is the biophotonics technology and market analyst at Tematys; email: [email protected]. Carlos Lee is the director general of EPIC, the European Photonics Industry Consortium; email: [email protected].