Optical fibers have a broad range of uses in medical and clinical applications and can be adapted for many invasive and noninvasive procedures. However, any optical fiber to be used inside a human body first must be sterilized. This article reports on a study that examined the effects of six common sterilization methods on optical fibers with different coatings.
Andrei A. Stolov, OFS Fitel LLC
The use of optical fiber and fiber optic sensors within the medical world is increasing. These optical fibers and fiber optic-based sensors are ideally suited for a broad range of invasive and noninvasive medical applications, including urology, general surgery, ophthalmology, cardiology, endoscopy, dentistry and medical sensing.
Prior to being used inside the human body, fibers must be sterilized to ensure they are free of microorganisms and perform well during invasive and noninvasive medical procedures. Sterilization is any process that destroys all microbial life, such as fungi, bacteria, and virus or spore forms. The process can be accomplished in a number of different ways and, somewhat arbitrarily, sterilization methodologies can be subdivided into three groups: (a) The use of elevated temperature, (b) chemical treatment and (c) exposure to radiation. In fact, a few tens of sterilization techniques currently are known (some of which are used more often than others), and each method has its own advantages and disadvantages.

Designing and manufacturing the glass raw material, fiber, cable and the assembly of probes — in a controlled low-bioburden room — provides the capability to meet any required specifications. Courtesy of OFS.
In the case of optical fibers, some cannot sustain high temperatures while for others, the use of chemical sterilization may result in volatility or produce toxic and carcinogenic byproducts. Lastly, radiation techniques, such as gamma or electron beam (e-beam), can lead to significant alterations in the materials being treated. High-energy radiation is known to produce ionization and excitation of polymer molecules, which may result in crosslinking and/or chain scission.
So, the question remains that if an optical fiber must be sterilized, will the sterilization process cause any changes to its main properties, (i.e., its ability to transmit light and its mechanical strength)? Furthermore, how does one determine which of the sterilization techniques can or cannot be used? There are many types of optical fibers — each kind may have a different response to sterilization. The most vulnerable part of the fiber is its polymer coating, which typically deteriorates before the silica core and the cladding. Consequently, it is crucial to learn how different sterilization methods may affect fibers with different coatings.
Four types of fibers were selected for the study — all of them were OFS products. The fibers had 200-µm pure silica glass cores and 220-µm fluorine doped silica claddings with a numerical aperture (NA) of 0.22. As is shown in Table 1, different coating and buffer materials were applied to the fibers. Fluoroacrylate is a perfluorinated cladding material with lower refractive index and high hardness. Both fluoroacrylate and silicone function as secondary claddings, each with NA of 0.37. Two fibers were up-buffered with either poly(ethylene-co-tetrafluoroethylene), referred to as ETFE, or with polyether ether ketone (PEEK).
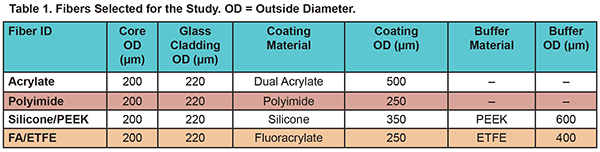
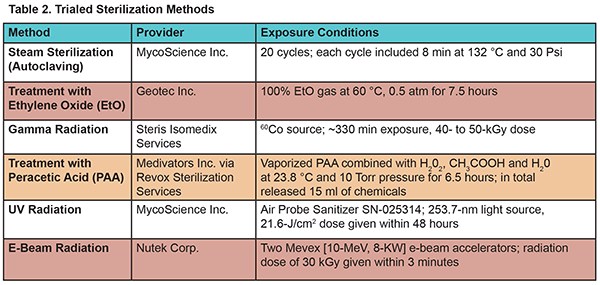
Table 2 lists the six most common sterilization techniques OFS trialed. Per the company’s request, the actual sterilization exposures were performed by different providers, who are experts in the area. We purposely wanted to “overshoot” the standard sterilization conditions and thus we requested that the providers either increase the exposure times or release more of the sterilizing agents onto the fibers. Table 2 also summarizes the actual conditions of the sterilization exposures, full details of which are examined within two published papers.1,2
Ideally, the sterilization of optical fibers should be such that it eliminates all microorganisms but does not affect optical attenuation and mechanical strength. Thus, our study was focused on investigating the mechanical and optical characteristics of the fibers.
The fiber strength was measured using a two-point bend testing approach at a standard strain rate of 4 percent per minute. Since it is known that the apparent fiber strength can be affected by the atmospheric humidity, we ensured that the Telcordia GR-20 condition was met for the measurements: Relative humidity = 50 ± 5 percent, and room temperature = 23 ± 2 °C. Figure 1 summarizes the obtained data.
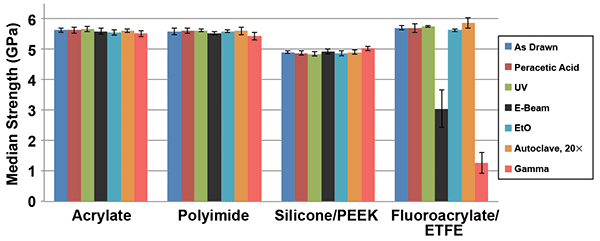
Figure 1. Effects of sterilization conditions on median strength of optical fibers. The error bars correspond to standard deviations.
In Figure 1, we see that the strength of the “as drawn” fibers with dual acrylate, polyimide and fluoroacrylate coatings is approximately 5.6 gigapascals, while the fiber with the silicone coating is inherently weaker. Four of the six trialed sterilization methods do not affect the fiber strength of all four fibers. Those methods are steam sterilization, UV radiation, and treatments with peracetic acid (PAA) and ethylene oxide (EtO). Gamma and e-beam radiation did not cause degradation to three out of four fibers — those with acrylate, polyimide and silicone/ETFE coatings remained unaffected. However, the fiber with the fluoroacrylate coating and ETFE buffer became noticeably weaker after such treatments. The gamma-treated fiber became so weak that we were not able to handle it without it breaking.
Why did this happen with the fluoroacrylate-coated fiber but not with the other ones? It is believed that the strength degradation occurs whenever the coating and buffer materials contain significant amounts of fluorine. Fluorine-containing polymers are known to be much more sensitive to radiation than fluorine-free polymers. Most likely, interactions with e-beam and gamma radiation generate hydrofluoric acid (HF) as one of the reaction’s products. This acid easily can diffuse through the coating and deteriorate the glass surface, which results in significant strength degradation. Clearly, since the observed strength degradation is related to the fluorine content in the coating/buffer layers, similar behavior is expected for other types of commercially available fluorinated polymer-coated fibers. Therefore, it should be concluded that both the gamma and e-beam radiation are unfavorable for optical fibers with perfluorinated coatings and/or buffers.

Among the trialed sterilization methods, autoclaving proved to be the fastest and the easiest approach. It is worth noting that a single autoclaving cycle provides a sufficient sterilization effect. If the fiber is to be used more than once, though, it will need to be sterilized for each occasion. For this reason, it is important to know whether the fiber can withstand not just a single autoclaving cycle but can endure multiple autoclaving sterilizations.
Within an autoclave, the fibers are exposed to a relatively high temperature and highly concentrated water vapor. This combination may cause cracking of the polymer coatings and/or deterioration of the glass cladding surfaces. Both of these factors may result in strength degradation of the fiber, which is typically cumulative. Furthermore, if stress is applied to silica-based optical fibers, their respective strengths become time-dependent due to crack growth that is enhanced by moisture. The degradation of fiber over time is known as “fatigue,” which is characterized by the stress corrosion parameter (nd). Higher values of nd correspond to lower rates of crack growth (i.e., higher mechanical reliability of the optical fiber).3 Per industry standards, the fiber is considered reliable if nsub>d exceeds 18. The nsub>d values can be determined via two-point bend testing of fibers at different strain rates.
It was found that even after 20 autoclaving cycles, the nsub>d values remained greater than 18 for all four fiber types being analyzed. This means that for reasonably short times, these optical fibers are usable in harsh conditions, although not necessarily within medical applications. Still, it should be said that some minor features of degradation were observed: For acrylate-coated fiber, the nsub>d value decreased from 30.7 ± 1.1 to 25.1 ± 1.1 after autoclaving. This was the only statistically significant change.
The effects of sterilization on optical fiber attenuation are displayed in Figure 2. As in Figure 1, there are four sterilization methods (steam sterilization, UV radiation, treatment with PAA and treatment with EtO) that do not affect
optical loss. In contrast, gamma and e-beam radiation create a problem — this time for all four fibers. The attenuation induced by these types of radiation is wavelength dependent and increases at shorter wavelengths.1,2
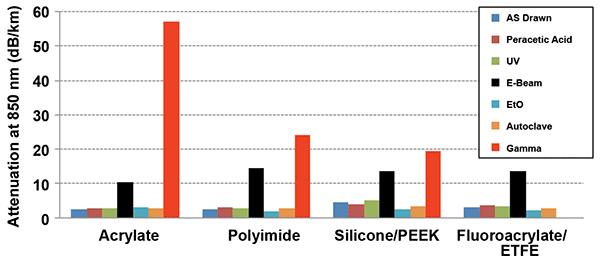
Figure 2. Attenuation at 850 nm (optical time-domain reflectometer data) for “as drawn” and sterilized optical fibers.
Interactions of silica glass with ionizing radiation are well-studied.4,5 Gamma and e-beam radiation are able to travel some distance (typically a few inches) in most materials. The radiation gets into the fiber core, where it then may induce different types of defects, such as the nonbridging oxygen hole center (^Si-O*), the E’ center (^Si*), the peroxy radical (POR: ^Si-O-O*) and the trapped electrons, where the notation ‘‘^’’ represents three bonds with other oxygen in the glass network and ‘‘*’’ denotes an unpaired electron.5 All those defects result in the development of absorption bands in the wavelength range below 800 nm. The lower-frequency wings of those bands spread throughout the whole visible and near-infrared spectral regions and are responsible for the changes indicated in Figure 2. Thus, it follows that gamma and e-beam radiation are not the best methods of sterilization — even for fibers with nonfluorinated coatings.
So, which sterilization methods for optical fibers are good and which are not so good? Treatment with EtO, PAA and exposure to UV light did not affect the main fiber properties and should be considered as the least-harmful techniques. Steam sterilization is another good approach; we found that up to 20 autoclaving cycles produced only a minor influence on reliability — and this occurred in only one of the four studied fibers.
In contrast, e-beam and gamma radiation induce additional optical loss, negate any possible benefits obtained through their use, and largely should not be considered as advisable methods for sterilizing optical fibers. Certain fibers (i.e., those without fluorinated coatings) still may be considered sterilizable with e-beam or gamma radiation if the induced losses fall within the allowed attenuation budget of the equipment. Perfluorinated materials, however, definitely are not compatible with sterilization via e-beam and gamma radiation.
Based on our observations, can we say which coating type is optimal for use in medical applications? It looks like all trialed coating types perform pretty well, at least with four out of six applied sterilization methods. Since sterilization does not cause a problem, optical equipment designers can utilize fibers with any of these coatings and base their respective selections on only the requirements of the specific medical application.
Meet the author
Andrei A. Stolov is a member of the technical staff at OFS Fitel LLC in Avon, Conn.; email: [email protected].
References
1. Stolov, A.A. et al. (2013). Effects of sterilization methods on key properties of specialty optical fibers used in medical devices. Proc SPIE, Vol. 8576, p. 857606.
2. Stolov, A.A. et al. (2014). Effects of sterilization on optical and mechanical reliability of specialty optical fibers and terminations. Proc SPIE, Vol. 8938, p. 893806.
3. Kurkjian, C.R. and Matthewson, M.J. (2007). Mechanical strength and reliability of glass fibers. Specialty Optical Fiber Handbook. Elsevier, Amsterdam, pp. 735-781.
4. Girard, S. et al. (2008). Radiation effects on silica-based preforms and optical fibers—I: Experimental study with canonical samples. IEEE Trans Nucl Sci, Vol. 55, No. 6, pp. 3473-3482.
5. El-Kheshen, A.A. (2012). Glass as radiation sensor. Curr Top Ioniz Radiat Res. InTech, Rijeka, pp. 579-602.