ASHBURN, Va., Oct. 26, 2015 — With resolution seven times greater than conventional light-sheet microscopes, an advanced device can capture cell-level 3D images across entire small organisms.
Developed at the Howard Hughes Medical Institute's Janelia Research Campus, the microscope also captures images quickly enough to watch the movement of developing embyronic cells or the flashes of neuronal circuits.
Called IsoView, it's the first light microscope capable of imaging large, nontransparent specimens at subsecond temporal resolution and subcellular spatial resolution in all dimensions, according to group leader Philipp Keller. In one test, the device imaged fruit fly larvae at a spatial resolution of 1.1 to 2.5 μm and temporal resolution of 2 Hz for several hours.
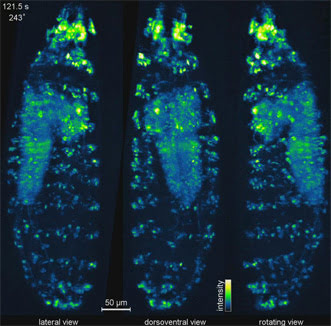
A 3D IsoView image of an entire fruit fly larva expressing a fluorescent indicator for neural activity through the nervous system, including the brain, ventral nerve cord and peripheral nervous system. Courtesy of the Keller Lab, HHMI/Janelia Research Campus.
Rather than collecting a single image of a sample with a single objective, the microscope simultaneously collects light and creates images from multiple angles. From each side, an objective produces a thin beam of light that sweeps the sample from top to bottom. The beams from each of the four objectives are staggered so that they do not interfere or intersect with one another, and a rolling shutter in each scientific CMOS keeps pace with the beam.
Each image still suffers from poor resolution along one axis, but the most useful data from each image can be combined computationally to generate a final image with good resolution in all dimensions. The team developed new software capable of processing 10 terabytes of data from one hour of imaging in about two days.
To maintain the imaging speed, coverage and resolution the team wanted, the microscope's four objectives needed to fit closely together with just enough room to move during imaging. They also had to be big enough to image large fields of view with good spatial resolution.
No commercially available objectives met these needs, so the researchers designed the parts they needed and waited about nine months for them to be custom built.
The researchers have used the IsoView to visualize cell-by-cell activity throughout the nervous system of an entire living fruit fly larva, an organism that has more than 10,000 neurons and is about 50 times larger than the roundworm C. elegans, which until now was the only animal whose complete nervous system had been imaged at the single-cell level.
The IsoView can produce images as the larvae moves freely in a loose gel, which, according to Keller, "opens up the possibility of functional imaging in an entire, behaving animal. It is even possible to perform high-speed functional imaging over developmental time scales, as we demonstrated in imaging a fruit fly embryo developing into a larva."
The scientists also performed high-resolution functional imaging of activity in the entire brain of a larval zebrafish, demonstrating that neurons in the deepest, least accessible regions of the brain could be seen clearly, separate from their neighbors.
Finally, they used the IsoView to track cells in a developing fruit fly embryo. A multicolor imaging mode enhanced the information presented in the resulting videos by detecting different fluorescent labels on cells' membranes and nuclei.
The research was published in Nature Methods (doi: 10.1038/nmeth.3632).