Super-corrected optics, increased sensitivity and faster throughput mean that confocal systems are revealing more biology in its natural context than ever before.
Like most things, confocal microscopes have improved with age. The technique dates back to 1955, when American cognitive scientist Professor Marvin Minsky proposed that the ideal microscope would eliminate all light rays not initially aimed at the focal point.
Gathering the optical tools that were available to him at the time, Minsky fashioned the first confocal microscope using an arc lamp to illuminate the specimen and a low noise photomultiplier to measure the output. Instead of scanning mirrors, the specimen was sequentially moved to image point after point. Finally, the image was reconstructed on the screen of a military surplus radar scope. (The image remained visible for about 10 seconds, which was also how long it took to make each scan.)
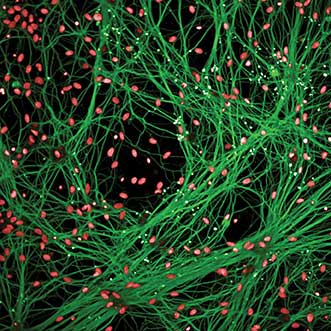
Primary dorsal root ganglion neurons co-cultured with primary rat astrocytes were stained with Tuj1/488 (axons, green) and Draq5 (nuclei, red) and imaged on the Opera Phenix High Content Screening system in confocal mode using the 20× water objective. Courtesy of York Rudhard, Evotec AG.
Fast-forward to today and advances in every single component are allowing researchers to observe processes with more accuracy and with less damage to cells. From fixed-cell to live-cell imaging to applications in optogenetics — a research field that is currently exploding — confocal microscopy is the essential imaging workhorse in today’s core imaging facilities.
Arc lamps have been replaced by lasers, high-efficiency scanning mirror units and high-throughput fiber optics allow for faster image acquisition, and there are better thin-film dielectric coatings and detectors with reduced noise characteristics. But can confocal microscopes compete with multiphoton and other highly advanced microscopy systems available today?
Fighting back with competitive innovations are the leading imaging specialists who understand that even mature technologies can offer exciting new possibilities.
“Manufacturers have developed super-corrected optics that allow co-localization studies with much more accuracy than before,” said Kevin Jia, senior marketing manager of Life Science at Olympus Scientific Solutions Americas in Waltham, Mass. “Also, throughput on our systems is much higher. Spectral imaging is vastly improved and live-cell imaging is possible with very low laser power.”
Working distance, wavelength ranges and other aspects of the optics now are tailored for specific applications to help confocal move from a fixed-cell technique to a highly sensitive, live-cell technique.
“These are incremental advances, but important incremental advances like these can make a huge difference in the progress of science,” Jia said.
One of the biggest recent innovations is the Zeiss Airyscan system, which uses an array of 32 GaAsP photomultiplier tubes (PMTs) to scan the airy disc pattern from a light source. The result is a “superresolution” image that beats the Abbe diffraction limit by a factor of about 1.7 to achieve 140 nm in XY and 400 nm in Z.
“This gives a significant improvement in image quality, and is especially useful in looking at structures on the limit of traditional confocal imaging,” said Dale Moulding, facility manager at the Institute of Child Health at UCL, UK. “Things like small organelles and mitotic structures such as kinetochores. I believe one real benefit will be in co-localization analysis, as the improvement in Z resolution is a big step forward.”
Zeiss also claimed that the system helps in live imaging due to the inherently lower noise from the 32 sensors providing instant averaging.
“Images are certainly less noisy using the Airyscan detector [as opposed to the] GaAsP PMT, so acquisition can be done about four times faster and give the same quality image,” Moulding said.
Recent improvements in PMT sensitivity have increased photon detection efficiency from about 20 percent (in standard alkaline PMTs) to 45 percent (in GaAsP-PMT and GaAsP-HyD). This allows faster imaging, higher sensitivity and signal-to-noise ratios, which means lower laser power can be used and much less damage is caused to the specimens during imaging. But many feel there is still room for improvement.
If you can’t beat them, join them
With superresolution technologies such as photo activated localization microscopy (PALM), stochastic optical reconstruction microscopy (STORM) and stimulated emission depletion microscopy (STED) gaining traction in many emerging applications, confocal microscope manufacturers are putting more emphasis on multifunctional designs.
“One emerging application is combining confocal and superresolution, which several companies now do,” said James Lopez, laser scanning confocal and multi-photon specialist at Olympus Scientific Solutions Americas. “Some users want confocal systems to be multifunctional. They want a high-end research system that can do localization, superresolution and confocal on a single microscope.”
The Olympus OSR superresolution system provides resolution down to 120 nm with a couple of clicks. While it may not provide the localization you get with PALM, STORM or STED, it resembles structured illumination in terms of performance.
“We see confocal being combined with other imaging techniques,” Jia said. “For instance, we are looking at much higher-resolution and higher-contrast imaging through new superresolution technologies that are incorporated with spinning-disk or other confocal systems to create solutions providing fast, high resolution, high-contrast data.”
One example is the Opera Phenix High Content Screening System from Perkin-Elmer, which is a microlens-enhanced Nipkow spinning disk confocal system that allows the capturing of confocal images from samples in multi-well plates or on slides at a very high throughput.
“Compared with conventional confocal laser scanning microscopes (CLSM), the Opera Phenix system allows unsupervised image acquisition, which can produce up to 400,000 images per day,” said Karin Boettcher, associate product manager for High Content Screening and Applications at PerkinElmer, Hamburg, Germany. “This far higher throughput, compared to conventional CLSM, allows screening of whole small molecule or siRNA libraries for a desired phenotypic effect.”
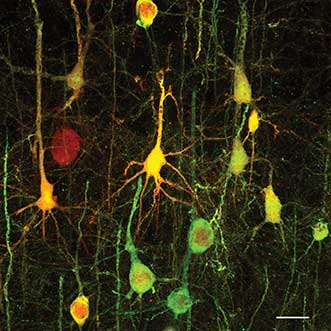
Confocal image of neurons from mouse motor cortex. Image acquired with an Olympus FluoView FV1200 microscope, using a 60× oil immersion 1.35NA objective. Scale bar represents 20 µm. Courtesy of the Byungkook Lim Laboratory, UC San Diego.
Spinning disk microscopy in general offers advantages for live cell microscopy, as it reduces photobleaching and phototoxicity compared to conventional point or line scanning microscopes.
In a bid to increase the speed of high content screening systems, simultaneous multicolor imaging is commonly used. The drawback to this approach, however, is spectral crosstalk, which can limit assay sensitivity.
To overcome this, PerkinElmer developed an innovative optical concept called Synchrony Optics. “Synchrony Optics separate fluorescence excitation and emission in space and time, which allows simultaneous imaging of four channels with minimal crosstalk,” Boettcher said.
There is another enemy that can blight image quality. As well as spectral crosstalk between fluorophores, spinning disk methods can be compromised by a phenomenon known as “pinhole crosstalk.” This interference comes from out-of-focus light and scattered emission bleeding through neighboring pinholes on a spinning disk, leading to increased background haze when thicker samples are analyzed.
“The confocal scanning unit of the Opera Phenix High Content Screening System provides an increased distance between pinholes to reduce the pinhole crosstalk,” Boettcher said. “This leads to improved 3D image quality and results in clearer images when samples such as 3D microtissues are analyzed.”
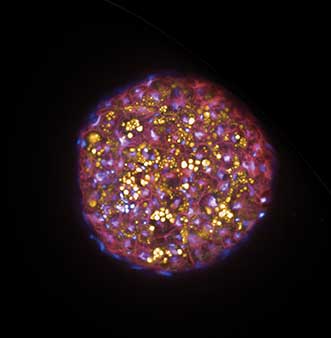
Maximum intensity projection image of a primary 3D Insight human liver microtissue from InSphero stained with Hoechst (nuclei, blue), Nile Red (lipids, orange) and CellMask DeepRed (membranes, red), imaged on the Opera Phenix High Content Screening System in confocal mode as stack (50 µm) with the 20× Water objective. Courtesy of PerkinElmer.
Ultimately, the future challenge for confocal imaging will be to deliver even higher sensitivity while becoming easier to use and more flexible. At Olympus, Jia explains that the aim is to create an ultra-high-performance system with the ease of use of a smartphone.
“Confocal will remain one of the key 3D optical imaging methodologies for life science researchers for a long time to come,” Lopez said. “We are approaching the top potential efficiency for each of the contributing parts of the system making these systems perform better than ever, but new and unanticipated advances in confocal technology also will arise.”
[email protected]
Confocal microscopy: a quick comparison
Choosing the right microscope for the job largely depends on the application, the type, size and thickness of the sample, as well as the information that the user is trying to extract. Here is an overview of how common microscopy techniques stack up:
Confocal vs. multiphoton microscopy
• If resolution is what’s required, then confocal wins hands down. Multiphoton microscopy inherently gives optical sectioning due to the excitation by two simultaneous photons being restricted to a very shallow depth of field. However, the longer wavelengths of multiphoton excitation result in worse resolution than confocal.
• Generally, resolution scales proportionally to excitation wavelength (Abbe diffraction limit), so the >800 nm resolution of a multiphoton is about half as good as from a single photon system.
• For 3D imaging, confocal performance may be limited in depth compared with multiphoton, but it is much easier to generate a beautiful stack for research and publishing, and for many experiments confocal imaging is more than sufficient.
• When it comes to spectral separation, confocal offers less crosstalk so four-color imaging can be routinely performed with high axial resolution. If this were attempted using multiphoton microscopy, it not only would be expensive, but two different lasers might be required or a single infrared laser might need to be retuned.
• A confocal system can cost one-third to one-half of the cost of a fully outfitted multiphoton system.
• On the other hand, if deep penetration into tissue of intact small animals is required, then multiphoton is preferred (light sheet microscopy is limited in depth unless the specimen is small or cleared). The main reason for this is absorption of excitation energy, as well as scattering of excitation and emission light by the specimen. Ways to overcome this limitation are, for example, the immersion of tissues into optical clearing solutions like Scale (Hama et al., 2011) to reduce the light scattering in tissue and make tissue more transparent. However, this technique works only in fixed samples and is currently not applicable to live cell samples.
Confocal vs. widefield fluorescence microscopy
• Traditional widefield fluorescence imaging includes out-of-focus light from above and below the imaging plane — however, this can be removed post acquisition by deconvolution.
• Laser scanning microscopy (LSM) is slow compared with widefield, as images are scanned one pixel at a time. However, spinning disc confocals are just as fast as widefield.
• Spinning disc systems are limited to a set resolution depending on the camera used as well as the optics, so they may not always achieve the best possible resolution.
• Since PMT sensitivity is still a long way short of other sensors, the excitation input of LSM confocal needs to be high. This can lead to sample bleaching problems. This isn’t such an issue with spinning disc confocal.