MONTREAL, Jan. 27, 2016 — A nanotherapy that converts near-infrared light to UV light may mitigate side effects caused by drug regimens that can damage healthy cells.
Researchers from McGill University and the University of Quebec demonstrated the technique using nanoparticles that converted near-infrared (NIR) light into UV light, coating the particles in a UV-sensitive hydrogel shell infused with a fluorescent protein as a stand-in for drug molecules.
When exposed to NIR light, the nanoparticles instantaneously converted it to UV, which induced the shell to release the protein payload. The researchers said their on-demand delivery system could not only supply drug molecules, but also agents for imaging and diagnostics.
An existing approach to localized drug delivery uses drug-carrying materials that, when exposed to UV light, release their therapeutic cargo. However, UV light cannot penetrate body tissues and is carcinogenic with repeated exposure.
NIR light, however, can penetrate 1 to 2 cm, but photosensitive drug-carriers don't react to it.
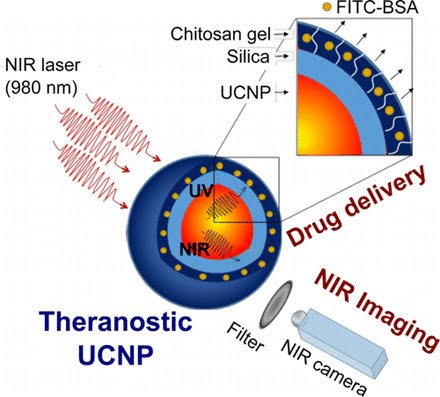
Theranostic lanthanide-doped upconverting nanoparticles. Courtesy of the Journal of the American Chemical Society.
To address these limitations, researchers identified lanthanide-doped upconverting nanoparticles (UCNPs) as excellent nanotransducers for converting longer wavelength near-infrared (NIR) light to shorter wavelengths spanning the UV to the visible regions of the spectrum. That multiphoton absorption process is known as upconversion.
The team developed NIR to UV–VIS–NIR UCNPs consisting of LiYF4:Yb3+/Tm3+@SiO2 individually coated with a 10 ± 2-nm layer of chitosan (CH) hydrogel crosslinked with a photocleavable cross-linker (PhL).
They then encapsulated fluorescent-bovine serum albumin (FITC-BSA) inside the gel. Under 980-nm excitation, the upconverted UV emission cleaved the PhL crosslinks and instantaneously liberated the FITC-BSA under 2-cm-thick tissue. The release was immediately arrested when the excitation source was switched off. The upconverted NIR light allowed for the tracking of particles under the tissue.
Nucleus pulposus cells cultured with the UCNPs were viable both in the presence and in the absence of laser irradiation. Controlled drug delivery of large biomolecules and deep tissue imaging could make the system a theranostic — or individualized — platform for tissue engineering, biomapping and cellular imaging applications, the researchers said.
The work was published in the Journal of the American Chemical Society (doi: 10.1021/jacs.5b12357).