Fluorescence Lifetime Imaging (FLIM) combined with Optical Projection Tomography (OPT) has been demonstrated as useful for imaging and monitoring apoptosis in whole live organisms in 3D. Termed FLIM OPT, the method may provide a means to visualize cell signaling processes and even responses to potential drug therapies in live organisms over space and time.
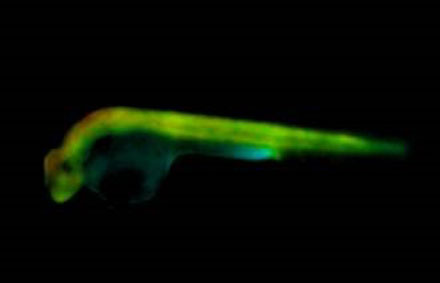
Fluorescence lifetime imaging (FLIM) combined with op-tical projection tomography (OPT) has the potential to map Förster resonant energy transfer (FRET) readouts in space and time in intact transparent or near transparent live organisms such as zebrafish larvae, thereby providing a means to visualize cell signaling processes in their physiological context. Courtesy of Imperial College London.
Researchers at the Imperial College London and University College London used FLIM OPT to read out biological function in live transgenic zebrafish larvae. Apoptosis in the zebrafish was mapped in 3-D by imaging the activity of a genetically expressed Förster Resonant Energy Transfer (FRET) biosensor for Caspase 3, an enzyme that is essential during development and homeostatic tissue turnover as well as during disease. As soon as the researchers activated the Caspase 3 enzyme, it started cleaving the biosensor. The fluorophores were separated and the energy transfer between them decreased. FRET reads out the colocalization of two or more fluorophores via the strength of the resonant energy transfer that occurred between them.
In order to demonstrate in vivo FLIM readouts of the Caspase 3 FRET biosensor throughout whole intact zebrafish embryos, it was necessary for the researchers to address three main challenges: (1) the generation and validation of appropriate transgenic zebrafish expressing the Caspase 3 biosensor; (2) the optimization of OPT imaging instrumentation and protocols for live zebrafish embryos, and (3) the analysis of large, complex FLIM OPT data sets to realize the quantitative FRET readouts. The researchers generated a novel transgenic zebrafish line expressing a Caspase 3 FRET biosensor under the control of a ubiquitous promoter in a nonpigmented zebrafish.
OPT was used because it offered several benefits, including the ability to image with transmitted light and with fluorescence and to utilize an array of fluorescent markers and biosensors. Because OPT is a wide-field illumination and detection technique, the incident light intensities associated with it are lower than those associated with laser scanning microscopy, potentially reducing photobleaching and phototoxicity. OPT is easily scaled over a range of sample sizes and is able to image entire mesoscopic samples in a few minutes, which is important for live animal imaging when the sample is anesthetized.

Only 150 seconds were required to acquire the 3-D FLIM dataset, compared to 300 seconds required to image a single optical section using laser scanning confocal microscopy with time-correlated single photon counting. This imposed less stress on the organism, allowing the fish to survive longer. The low light dose associated with wide-field imaging may enable extended time-lapse studies to be undertaken with the potential to recover the zebrafish after imaging for further longitudinal studies, thus enabling apoptosis in the same zebrafish to be monitored over a period of time.
Imaging functional cell behavior in the context of whole live organisms may be of significant use in the development of potential therapies and may contribute to a deeper understanding of cellular signaling responses. Zebrafish are ideal organisms for such whole-body imaging techniques; however, 3-D imaging technology that is suitable for zebrafish is still under development. FLIM OPT could provide a way to visualize whole body responses to potential therapies. The researchers believe that this approach could be useful in drug discovery, as zebrafish are an ideal model for drug screening.
For the study of systemic effects of disease, the development of potential therapies, and a deeper understanding of cellular signaling responses, microscopy of cells in culture may not be sufficient. The ability to image functional cell behavior in the context of whole live organisms may further the study of systemic effects of disease and potential therapies as well as the understanding of localization and propagation of signaling responses in cells.
The research was published in Journal of Biophotonics (doi: 10.1002/jbio.201500258)