Laser-powered nanoblade technology typically used to transfer materials between cells has now been used to slice through a cell’s membrane to enable the insertion of mitochondria. Other bioengineering applications of the nanoblade, which can deliver cargo as small as a few microns in size, are also being explored.
University of California, Los Angeles (UCLA) bioengineers Mike Teitell and Pei-Yu “Eric” Chiou conceived the nanoblade several years ago to transfer a nucleus from one cell to another. However, they soon delved into the intersection of stem cell biology and energy metabolism, where the technology could be used to manipulate a cell's mitochondria.
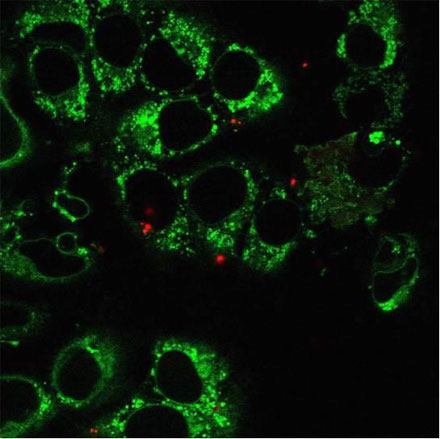
This image shows human osteosarcoma cells and mitochondria (green), with additional mitochondria (red) from human embryonic kidney cells transferred via the nanoblade. Very few red-labeled mitochondria are actually delivered by the nanoblade into each cell, compared to the pre-existing green mitochondria. Over a few weeks, the mitochondrial DNA in the red-labeled mitochondria will massively amplify. Courtesy of Ting-Hsiang Wu.
The nanoblade apparatus consists of a microscope, laser, and titanium-coated micropipette to act as the "blade," operated using a joystick controller. When a laser pulse strikes the titanium, the metal heats up, vaporizing the surrounding water layers in the culture media and forming a bubble next to a cell. Within a microsecond, the bubble expands, generating a local force that punctures the cell membrane and creates a passageway several microns long that the cargo — in this case, mitochondria — can be pushed through. The cell then rapidly repairs the membrane defect.
The UCLA team used the nanoblade to insert tagged mitochondria from human breast cancer cells and embryonic kidney cells into cells without mitochondrial DNA. When they sequenced the nuclear and mitochondrial DNA afterward, they saw that the mitochondria had been successfully transferred and replicated by 2 percent of the cells, with a range of functionality. Other methods of mitochondrial transfer have only achieved success rates of 0.0001 to 0.5 percent, according to the researchers.
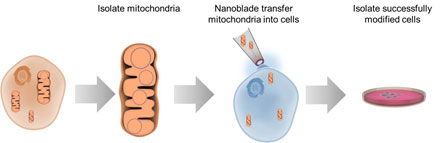
This diagram illustrates the process of transferring mitochondria between cells using the nanoblade technology. Courtesy of Alexander N. Patananan.
Studying the effects of mutations in the mitochondrial genome, which can cause debilitating or fatal diseases in humans, has been bottlenecked, according to Teitell. "So we are working on a two-step process: edit the mitochondrial genome outside of a cell, and then take those manipulated mitochondria and put them back into the cell. We're still working on the first step, but we've solved that second one quite well," he said.
The team has already scaled up the nanoblade apparatus into an automated, high-throughput version. Their goal is to enable other researchers to work with the insertion technology for such cargo as antibodies, pathogens or synthetic materials.
The research was published in Cell Metabolism (doi: 10.1016/j.cmet.2016.04.007). It was supported in party by the Air Force Office of Scientific Research, the National Institutes of Health, the National Science Foundation, California Institute for Regenerative Medicine, a Prostate Cancer Foundation Challenge Award, a Broad Stem Cell Research Center Training Grant and Innovator Award, an American Cancer Society Research Scholar Award, a Melanoma Research Alliance Established Investigator Award, the National Center for Advancing Translational Sciences, and NanoCav LLC.