
Breakthrough for Real-Time THz Imaging
When paired with fast detectors, terahertz quantum cascade lasers are ideally suited for industrial nondestructive testing and real-time biomedical imaging.
PIERRE GELLIE, LYTID SAS
Terahertz (THz) technologies have become a major field of applied research, driven by the tremendous potential of applications such as nondestructive testing (NDT), biomedical imaging, security screening and telecommunications. Imaging using THz radiation has garnered increasing attention, as it promises penetrating, contactless and submillimeter diffraction-limited imaging that is suitable for dry, nonpolar, nonmetallic materials such as plastics and ceramics. The main challenge in achieving the full potential of THz imaging: the limitations in the performance of current THz sources and detectors.
THz quantum cascade lasers (QCLs) are electrically pumped unipolar semiconductor lasers. Their operation mechanism differs fundamentally from standard semiconductor diodes as they exploit intersubband transitions in cascaded quantum well structures to generate terahertz radiation between 2 to 5 THz or 150 to 60 μm in wavelength. Unlike standard diode lasers where the emission frequency is determined by the energy bandgap difference between the materials used, QCL emission frequency is determined by the engineered band structure — the size and width of the quantum wells — and is independent of the materials used. The cascaded structure, hence the name QCL, allows for quantum efficiencies greater than unity. Each electron that goes through the structure will emit N THz photon, where N is the number of periods in the laser. Typically, the value of N is around 100. The Ga/As Al/Ga/As material system used for THz QCLs offers excellent growth control as well as mature processing technology.
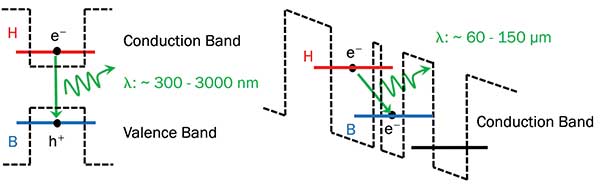
Standard semiconductor laser diode using interband transitions to generate radiation between 300 and 1000 nm (right). This range is limited by the energy bandgap difference of the materials used. Schematic (far right) of a single QCL period exploiting an intersubband transition between two adjacent coupled quantum wells to generate directly THz radiation between 60 and 150 μm (2 to 5 THz). Courtesy of Lytid SAS.
Turning state-of-the-art quantum devices into user-friendly tools
Several challenges had to be tackled before turning this quantum technology into a tool suitable for science and industry. Among them was developing compact laser drivers designed for the specific needs of THz QCLs; controlling the beam profile, especially for imaging applications; and achieving a cryogenic operating temperature.
Notably, QCLs can operate in continuous wave (CW) mode as well as quasi-CW (QCW) mode. With limited available cooling power, QCW with a high duty cycle generally provides higher average output powers. QCW operation also provides direct electrical modulation that allows THz QCL to be used in combination with any THz detector on the market without the need for a mechanical chopper. To drive these QCL structures in quasi-CW mode, short high-current electrical pulses are required.
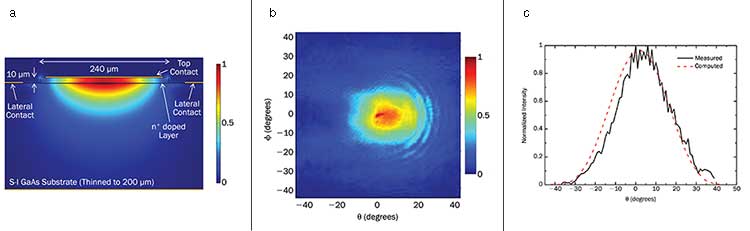
Computed intensity mode profile of THz QCL (a) in a surface plasmon waveguide. The optical mode is confined between a top metallic layer and a bottom highly doped contact layer. The optical mode can leak into the underlying substrate because the doped layer thickness is smaller than the skin depth. Typical measured far-field beam profiles of THz QCLs (b) provided by using surface plasmon waveguide design. The beam profile (c) offered by these waveguides is quasi-Gaussian. Courtesy of Lytid SAS.
Commercial systems exist that, in microseconds, generate up to 2-A short pulses needed to drive THz QCLs. These tend to be very bulky, housed on 19-in. rack systems, and are therefore not compatible with integration into a compact system. The goal is to design such a laser driver that would fit into the palm of a hand.
Another crucial consideration is optimizing the beam profile for imaging applications. By using custom-designed waveguides adapted to the longer THz wavelengths it’s possible to obtain a quasi-Gaussian beam profile. These waveguides are based on surface plasmon confinement of the optical THz mode between metallic layers and highly doped semiconductor contact layers. As the optical THz mode is free to leak into the substrate, the resulting output beam profile is excellent and is particularly adapted to imaging applications.
As of now, THz QCLs still require cryogenic cooling to operate. Although the maximum operating temperature is around 200 K, excellent performance can be obtained at temperatures of several tens of K, accessible to closed-cycle, cryogen-free cooling engines. At these temperatures, typical output powers are in the milliwatt range, which is about three orders of magnitude higher than other available commercial THz sources, at this frequency. This is based on either pulsed THz generation or nonlinear up-conversion (CW using two distributed feedback lasers). Another important aspect is the emission frequency. As the water vapor absorption of THz radiation is very strong — especially at higher frequencies — emission in an atmospheric transmission window is highly desirable for real-life applications.
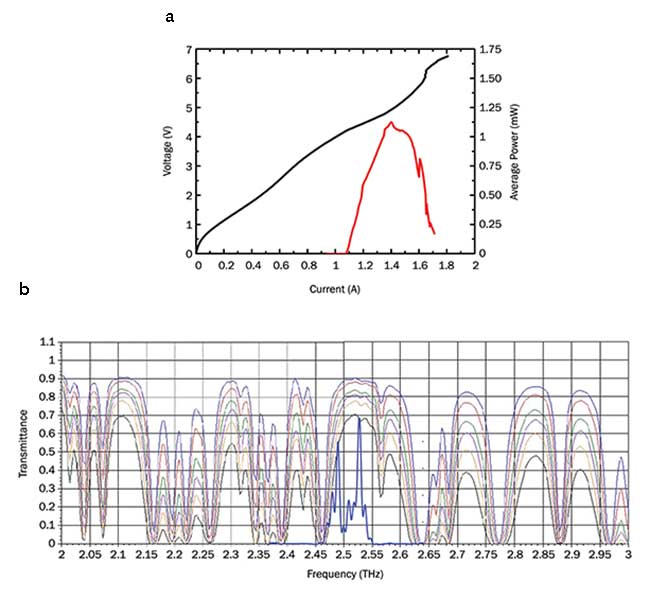
Light-current-voltage curve (a, top) of a typical commercial THz QCL provided by Lytid. Emission frequency of THz QCL systems (b, bottom) should ideally be inside an atmospheric transmission window to avoid strong water vapor absorption. Lytid’s QCL system emits in a 2.5-THz window. Courtesy of Lytid SAS.
These recent technical developments in THz QCL technology have led to the development of compact, powerful, fully integrated and user-friendly commercial THz sources. These versatile and portable systems can be easily integrated into real-world medical and industrial applications.
New possibilities in THz imaging
With the high output power available from THz QCL and their high emission frequency, new fields of THz imaging are opening up1. Higher power means faster image acquisition even with slow thermal detectors, such as pyroelectric, golay cell or bolometer detectors. It also means higher signal-to-noise ratios (SNR). With a standard room temperature operated commercial pyroelectric THz detector, SNR higher than 60 dB using lock-in amplification is possible. At frequencies higher than 2 THz, this is a few orders of magnitude better than available THz pulsed spectroscopic or CW systems. Higher frequency also allows higher imaging resolution. At 2.5 THz for instance, the diffraction limited resolution is around 250 μm and the beam waist is typically around a few hundred microns. In industrial NDT applications this allows the detection of smaller defects and the small beam size improves the measurements on highly curved surfaces compared to using a lower frequency.
In addition to speeding up classic raster scanning systems for image acquisition, the high power output of THz QCLs also enables the use of THz cameras based on microbolometer technology. These types of cameras are based on different technologies from CEA-Leti (France), INO (Canada) and NEC (Japan). Although microbolometer THz cameras offer noise equivalent powers (NEP) in the order of tens of pW, the high number of pixels (>60,000) require very high THz powers to illuminate the sensor. These cameras are also most sensitive at higher frequencies due to their large pixel size (several tens of μm).

The TeraCascade 1000 series from Lytid provides mW-level of THz power in a compact, fully integrated and turnkey system. This product won a 2016 Prism Award for Photonics Innovation in the Scientific Lasers category. Courtesy of Lytid SAS.
Real-time THz imaging could enable
inline THz NDT applications for the industry where both x-ray tomography (offline only) and ultrasonic system (needs physical contact) fail to provide satisfactory solutions. Compact, fully integrated THz QCL systems can be mounted on a robotic arm to provide automatic quality control, and the reliability of the coolers and QCL chips is robust enough for industrial applications. In the field of biomedical imaging, THz imaging is a strong contender for rapid, contactless detection of cancer and skin burn assessment. Real-time THz imaging could be a revolutionary tool in the biomedical field used extensively by health-care professionals.
Next-generation commercial THz QCL systems have brought this promising new technology out of the laboratories and into the hands of researchers eager to explore new THz applications, or industrials who want to implement THz solutions into their processes.
Reference
1. P. Dean et al. (2014). Terahertz imaging using quantum cascade lasers — a review of systems and applications. J Phys D Appl Phys, Vol. 47, No. 37.
Meet the author
Pierre Gellie co-founded Lytid SAS, a company that develops terahertz sources based on quantum cascade laser technology, in 2015. He holds a Ph.D. in physics from Paris Diderot University and has 8+ years of expertise in terahertz technologies; email: [email protected].
Cancer and Burn Diagnoses to Benefit from THz Imaging
THz radiation’s deep penetration combined with its strong water absorption has attracted a lot of attention in the biomedical field. That’s in large part due to the fact that water concentrations reveal a lot about the health of human tissue, with water content in cancerous cells higher than in healthy cells.
Monitoring this biological marker through imaging could help with the assessment of excision margins when tumors are surgically removed and provide information more quickly than today’s histopathology techniques do. Another crucial aspect is the reduced risk of additional surgery if the margins are badly delimited. Similarly, differentiating between second- and third-degree skin burns is very challenging as they are morphologically indistinguishable. This issue is of particular importance as second-degree skin burns will heal — but third-degree will not.
Water content variation and skin rehydration speed is the key to understanding and assessing whether skin will heal or not. Research has shown that THz radiation could be used to that end, but some challenges have to be addressed first. Most of the advanced studies were conducted on in-vivo tissue samples and faced the difficulty of focal plane variations due to the subject’s micro-movements originating from breathing and heart pulse. In these studies, images were acquired using slow raster scanning systems, which take several minutes to complete and are therefore affected by patient movements.
The focal plane changes reduced the quality of the results and render interpretation difficult or nonconclusive in some cases. Real-time imaging systems using high-power sources such as THz QCL and THz cameras could be the key to solving this issue and might lead to the development of new tools for biomedical THz imaging applications.
/Buyers_Guide/Lytid_SAS/c25100