The inherent three-dimensional spatial resolution and live sample compatibility of multiphoton microscopy are a perfect match for high-resolution studies in neuroscience using the latest optogenetic tools.
MARCO ARRIGONI and DARRYL MCCOY COHERENT INC.
More than half of the thousands of deployed multiphoton microscopes are used for neuroscience — in large part because of major funding initiatives across three continents. Some of these initiatives are specifically targeted toward in vivo studies, such as the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) initiative in the U.S. The most advanced of these studies combine multiphoton techniques with optogenetics to enable “all-optical” functional studies of neural networks. Two-photon microscopy is the only method of performing this stimulation and interrogation of three-dimensional live samples with single neuron resolution. An overarching trend in these experiments is to increase the number of neurons that are simultaneously investigated, which is driving exciting developments in both lasers and microscopes.
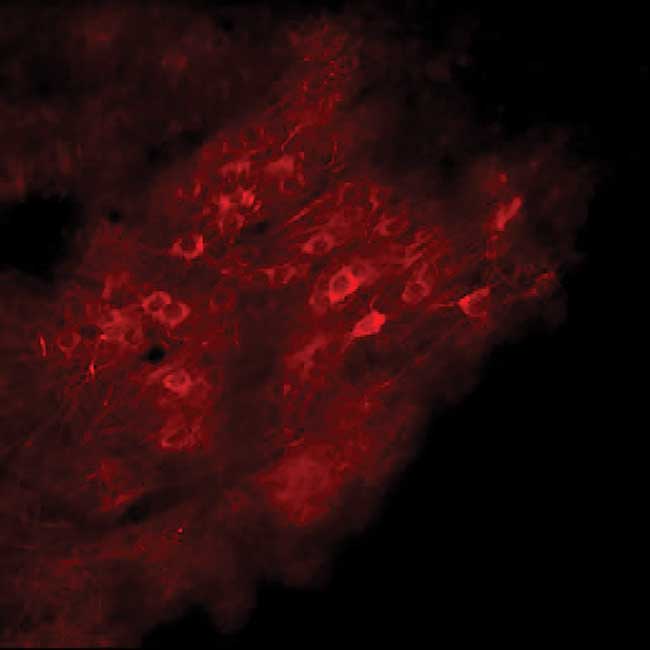
Figure 1. In vivo calcium imaging via two-photon
excitation of RCaMP2 using a next-generation ytterbium fiber laser
(Coherent Fidelity 2). Courtesy of Florent Haiss, University of Aachen,
Germany.
Evolution of optical microscopy
The optical microscope has a long history in neuroscience, originally for fixed and then for in vivo tissue samples. This includes techniques to image neurons and subcellular structures, based on absorption, reflection, polarization and fluorescence. In the last 20 years, fluorescence microscopy has been revolutionized by two developments. One was the availability of genetically encoded probes based on fluorescence proteins, such as eGFP, YFP and the mFruit series. Replacing conventional chemical dyes in many applications, these proteins enabled the establishment of colonies of model animals where only predesignated groups of cells respond with fluorescence to an excitation light stimulus. The other factor was the development of other genetically encoded indicators — whose fluorescence or other characteristics change dramatically due to local chemistry, such as calcium ion concentration, pH and membrane potential. Among these, the so-called genetically encoded calcium indicators (GECIs) have a prominent position.
In neuroscience, these developments enabled live tissue imaging in model animal brains, such as murine cortex, showing both structural and dynamic metabolic changes and activity. While many fluorescence microscopy studies use filtered lamps, the use of laser excitation here is often a critical advantage because of the lasers’ unique spatial and spectral brightness. More importantly, laser excitation enables fluorescence images with 3D resolution, which is hard to achieve with nonlaser illumination in real time. At first, this 3D imaging was in the form of confocal microscopy, but since the landmark study published by Denk, Strickler and Webb in 1990, multiphoton excitation methods are widely used because of several advantages.
In vivo biological samples, such as neural networks, are 3D systems. For many years, out-of-focus fluorescence background was a barrier to 3D imaging at high spatial resolution. With wide-field illumination, fluorescence is excited throughout the sample, wherever the fluorophore is present. With laser beam illumination, fluorescence can be limited in the XY plane but is still excited everywhere along the beam as it passes through the sample. Confocal laser scanning microscopy successfully addressed this challenge by focusing the laser to a small spot using a high NA objective. In its simplest implementation, fluorescence from this spot is imaged onto a photodetector through a pinhole that rejects the out-of-focus fluorescence. An XYZ image can then be built up point by point by scanning the sample and/or the optics.
Advantages of multiphoton stimulation and imaging
With multiphoton excitation, an ultrafast, or femtosecond, laser is tightly focused within the sample. In the focal plane, and only in the focal plane, the peak intensity is high enough to drive nonlinear processes. The simplest example is two-photon absorption (i.e., excitation) in a fluorophore, where a near-IR laser wavelength is used to excite a label, fluorescent protein or indicator that has a strong single-photon absorption at about half the near-IR laser wavelength. Other nonlinear techniques used for 3D microscope imaging include three-photon excitation of fluorescence, coherent anti-Stokes Raman scattering (CARS) and second harmonic generation (SHG). With all of these techniques, the light signal — fluorescent or otherwise — is only generated at the beam waist, enabling simple and efficient imaging optics to collect and detect this light.
Multiphoton excitation microscopy delivers three key benefits that can be critical advantages in neuroscience. Inherent 3D resolution means cells, or groups of cells, can be imaged at typically micron resolution in the Z-axis and as small as a few hundred nanometers in the XY plane. Also, in the case of imaging of dynamic events, a fourth dimension of time can be studied.
A second advantage is the improved ability to study live samples because of greatly reduced photobleaching and photothermal degradation. A typical example in neuroscience is to image the outer cortex of an anesthetized mouse via glass cover that replaces part of the cranium. People are often surprised to learn that damage at the focal plane is actually similar for single- and multiphoton excitation. However, single photon excitation damages the samples over a much larger volume — above and below the focal plane.
A third advantage for neuroscience is the ability to image at greater sample depths. Light scatter is often the limiting factor for microscopy imaging of thicker samples. As a researcher “looks” through more tissue, more light is scattered, resulting in a signal-to-noise decrease in the images. Much of this scatter is of the Mie type, which has a highly nonlinear dependence on wavelength. Consequently, the use of a 2× or even 3× longer wavelength results in a 16× or 81× reduction in scatter with a concomitant increase in potential imaging depth.
Optogenetics and all-optical physiology studies
The most recent and exciting advance in photonic experiments in neuroscience is the development of optogenetics — pioneered by Karl Deisseroth’s group at Stanford University and Ed Boyden’s group at the Massachusetts Institute of Technology. Optogenetics involves the use of opsins, proteins that can be expressed in targeted cell membranes and that act as ion gates or pumps that are switched on or off in response to illumination at an appropriate wavelength. In genetically modified animals or tissues, light can be used to stimulate changes in cell membrane potentials. In the case of neurons, light can be used to mimic an action potential spike and thereby stimulate signal transmission.
Optogenetics is already used in a broad range of experiments. In microscopy, optogenetics has enabled all-optical physiology experiments where optical microscopy is used to image the morphology of a group of live neurons, such as those in an animal cortex, and to stimulate selected neuron(s). It is also used to observe resultant activity in connected neurons via the use of calcium ion indicators for example. In addition, the use of ultrafast laser light for all of these processes allows these experiments to be conducted at single cell resolution (Figure 1).
The demands for faster and deeper imaging
Much of the capabilities in animal and human brains is a result of the way that neurons are massively interconnected. Consequently, there is a drive in multiphoton microscopy applications in optogenetics to study increasingly larger groups of neurons, and to image deeper into live brain tissue or over wider areas. Today’s cutting-edge experiments involve simultaneous stimulation/interrogation of a few tens of neurons, but some researchers would like to push this number to as many as 1,000 neurons. This is challenging both from spatial and temporal points of view. Specifically, it has long been known that neural signaling is characterized by both the interconnection pathways between neurons and by the relative timing of the signaling between them.
Real-time optical physiology experiments need imaging frame rates of tens of Hz — at least over some parts of the imaging field. When experiments involve an increase of two orders of magnitude in the number of neurons, an important consideration is how to image them in their location that can be a column of cortex measuring, for example, 250 µm in each dimension and at a depth up to 1 mm. It is also important to minimize any crosstalk between the stimulation and interrogation processes in these experiments. Laser manufacturers and microscope suppliers are both responding to this situation with exciting new developments.
Advances in lasers
All-optical functional studies with large neuron populations create four clear requirements in ultrafast lasers.
• Availability of two or more separated wavelengths to avoid crosstalk between stimulation and activity imaging. Initial morphology images can be performed with one of these same wavelengths.
• These wavelengths need to be as long as possible — as supported by the spectral characteristics of the available opsins and probes — in order to reach as deep as possible into the live tissue.
• Higher power is needed for higher speed. No matter how increased imaging speed is achieved, the result is dilution of the signal at each sample location. Higher power is required to maintain acceptable signal to noise.
• Laser ease of use and reliability are other paramount requirements, so that biologists can focus all their energies on these difficult experiments rather than the operation or optimization of their lasers.
For optogenetic experiments with just a few neurons, researchers use a titanium:sapphire (Ti:S)-based laser for one wavelength. When needed, a second wavelength is provided by an independently tunable optical parametric oscillator (OPO) pumped in parallel by the Ti:S laser. This combination delivers long wavelengths, flexibility and the ease of use/reliability researchers need. However, larger neuron population studies require more than 1 watt at wavelengths longer than 1 micron.
In response, laser manufacturers have developed closed-box ultrafast lasers based on ytterbium-doped gain materials that are the first commercial alternative to Ti:S in over 20 years. This includes streamlined fixed wavelength lasers (Figures 1 and 2), such as the Coherent Fidelity, which produces up to 18 W around 1040 to 1070 nm. For experiments requiring two wavelengths from a single source, laser manufacturers also developed lasers such as the Chameleon Discovery with 1.5 W of widely tunable (680 nm to 1300 nm) output together with 1.5 W of fixed wavelength output at 1040 nm.
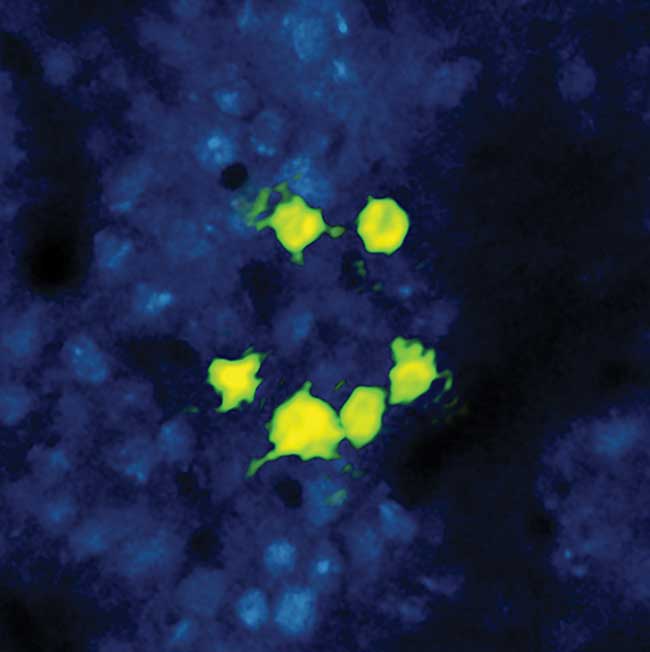
Figure 2. Some researchers are using fixed wavelength ytterbium fiber lasers to complement their existing tunable Titanium:sapphire lasers. This bright ‘smiley’ image shows neuronal cells in a live mouse subject. The cell fluorescence is due to the calcium ion activity via the expression of CCaMP6 excited by two-photon absorption using the output of a tunable Titanium:sapphire laser (Coherent Chameleon Ultra II). The ‘smiley face’ pattern has been photoactivated via expression of a long wavelength photoactivator (C1V1) via two-photon absorption using the multiwatt output of an ytterbium fiber laser (Coherent Fidelity) at 1055 nm. Courtesy of Lloyd Russell, Hausser group, University College London.
This new generation of ytterbium-based ultrafast lasers offers the requisite power and wavelength options to perform multineuron studies based on established two-wavelength approaches using longer wavelength opsins like C1V1 and a somewhat shorter wavelength calcium indicator such as Oregon Green. They are equally well-suited to alternative wavelength pairings based on red-shifted calcium indicators of the GCaMP family currently under development.
Just as important, ytterbium laser technology offers a reasonably straightforward path to even high powers to meet the inevitable demand as researchers soon move into the hundreds of neurons. The availability of dual wavelength pulses that are phase-correlated also makes these new lasers perfect sources for other nonlinear imaging modalities used in neuroscience and elsewhere, such as CARS and SRS.
Microscope advancements
Developments in microscopes are also enabling faster imaging. These developments fall into two categories: faster scanning modalities and multipoint imaging schemes to simultaneously create multiple beam waists within the sample. In the former group, the use of two or four acousto-optic modulators enables faster and addressable scanning, as in random access multiphoton (RAMP) microscopy pioneered by Saggau and co-workers. In the latter group, a spatial light modulator (SLM) enables 3D-patterned illumination that can be used to image or activate preselected zones of the sample (digital holography or generalized phase contrast).
The frontier of knowledge
Multiphoton microscopy enables unique experiments in neuroscience, particularly when combined with optogenetics. As scientists are pushing the frontier of knowledge in these studies, they are being supported by targeted developments in the two key technologies that are critical to this work: ultrafast lasers and fast scanning optical microscopes.
Meet the authors
Marco Arrigoni is the scientific market segment director of marketing for Coherent Inc. in Santa Clara, Calif.; e-mail: [email protected]. Darryl McCoy is director of product marketing for Coherent Inc. and is based in Glasgow, Scotland; e-mail: [email protected].