The use of quantum dots in combination with LEDs shows huge potential benefits for the lighting industry. The versatility and tunability of quantum dots can offer solutions when optimizing luminaire design for peak performance.
JAMES HARRIS AND DAVID WEBB, NANOCO TECHNOLOGIES LTD.
The invention of blue LEDs and the subsequent rapid rate of development of new phosphor and down-converting technologies have enabled the phenomenal growth of all LEDs in general lighting applications. White LEDs initially employed the use of a blue LED combined with a single phosphor with broad yellow emission. However, the light quality provided by this relatively simple solution was less than satisfactory, particularly in the red part of the visible spectrum. But more recently, new phosphors and phosphor combinations (green-yellow plus red) have been developed to provide a higher quality of light. While these advancements afford a significant improvement over previous offerings, phosphor technologies have yet to deliver desirable white light with rich color rendering ability along with the energy efficiency typically attributed to LEDs.
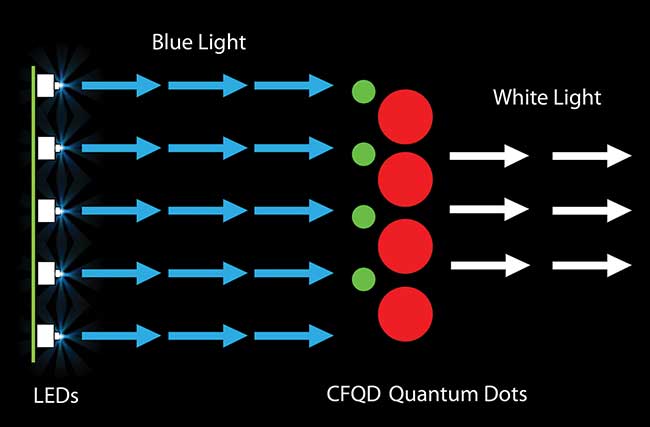
Blue LEDs excite red and green cadmium-free quantum dots, resulting in high-quality white light. Image property of Nanoco Technology.
Key lighting indicators
Generally speaking, acceptable white light corresponds to light that is close in color to that of a blackbody radiator and is commonly described by correlated color temperature (CCT), color rendering index (CRI) and luminous efficacy of source (LES). These are detailed below.
CCT is often used to describe the color of white light and refers to the temperature of the blackbody radiator that is closest in color to that of the light source being described. Good color rendering refers to the ability of the light to illuminate objects as they appear (to the observer) similar in color, just as they would when illuminated by a blackbody light source of the same CCT.
CRI measures a light source’s ability to reveal the intrinsic colors of the objects it illuminates. Ranging from −100 to 100, a perfect score of 100 is achieved when the illuminated color matches the blackbody radiator at the same correlated color temperature.
The figure of merit most often referenced when discussing electrical efficiency is the LES, which is a measure of how many lumens of light per electrical watt of energy a light source can supply.
In addition to these common indicators, the efficacy of a light source can be described by its luminous efficacy of radiation (LER). More simply, this is a measure of the optical power spectrum of the light source that overlaps with the photopic eye sensitivity curve (Figure 1). The photopic eye sensitivity curve describes how bright a particular wavelength of light appears to the human eye, with a peak at 555 nm, tailing off into the red and blue regions. Thus any light emitted outside of the region where the eye is sensitive leads to inefficiencies.
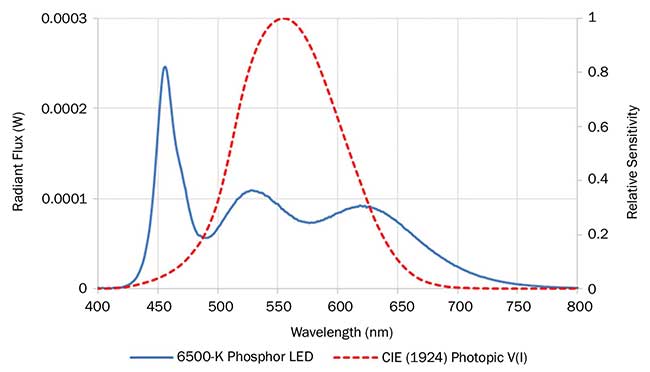
Figure 1. Spectral power distribution of a commercially available high-quality 6500-K white LED (blue). Photopic eye sensitivity curve (red). Image property of Nanoco Technology.
Searching for optimum CRI and efficiency
Good color rendering ability and high efficiency are unfortunately mutually exclusive, and for a given color temperature there is an optimum spectrum that can provide the best color rendering with minimum compromise to efficiency. Figure 1 shows an example spectrum of a high-quality white LED producing superior quality 6500-K light via the use of a green-yellow plus red phosphor combination. The spectrum features a narrow blue emission combined with a broad green-yellow-red emission from the phosphor blend.
To achieve the optimum spectrum, manufacturers of phosphor materials have sought to develop phosphors with narrow emission spectra that are widely tunable over a broad range of wavelengths. However, one of the challenges phosphor developers face is narrowing the spectral emission width of red phosphors so that more of the intensity emitted falls within the sensitive wavelengths of the human response curve. With broad emission red phosphors, much of the emitted red light falls outside of the region of human spectral response and therefore is effectively wasted, resulting in devices with less than optimized performance. In close review of Figure 1, the dashed line of the photopic eye sensitivity curve in relation to the LED spectrum demonstrates how some of the red light emitted from the 6500-K LED is wasted.
Quantum dots to the rescue
A strong and promising solution to the aforementioned challenges is the utilization of quantum dot (QD) technology, which has the natural ability to absorb some of the LED’s light, convert it and then re-emit it with the desired color characteristics. Quantum dots are nano-sized semiconductor crystals, whereby the size of the crystal is such that the material displays quantum confinement of electron and hole wave functions, giving rise to discrete energy levels similar to those in atoms or molecules. This quantum confinement effect results in a strong relationship between the size of the crystal and the electronic bandgap, and correspondingly the emission wavelength of the material. This strong relationship between size and bandgap presents a number of advantages: The emission of an ensemble of quantum dots can be easily tuned via the average crystal size, and the bandwidth of the emission from the ensemble can be tuned because it is a reflection of the size dispersion of the crystals. Typically the diameters of quantum dots are in the range of 1 to 20 nm and can be envisioned to have an onion-like core-shell structure (Figure 2), where the innermost part is the quantum dot core that is coated in a shell of a wider bandgap semiconductor whose role is to isolate the electron and hole from the environment both physically and electronically. Additionally, a second shell of organic molecules (capping ligands) are attached to the outside of the shell to give further protection and impart solubility to the quantum dots.
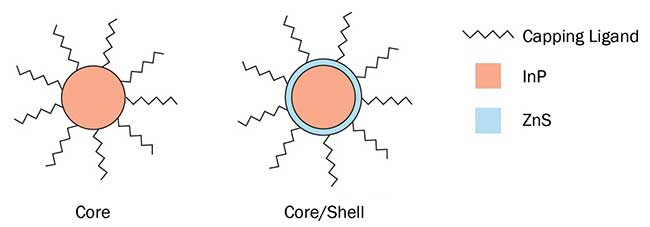
Figure 2. Illustration of simple core and core-shell quantum dot architectures. Image property of Nanoco Technology.
Synthetic methods for the manufacture of colloidal quantum dots have been developed over the past 20 years to finely control the size dispersion of the crystals, along with tuning the emission wavelengths. There are a number of materials that, when prepared on the nanoscale, can emit visible light and therefore are useful as down-converting materials in LED lighting applications. Examples include CdTe, CdSe, CdS, InP, CuxIn(Zn)1-x(S/Se)2, AgIn(S/Se)2 and ZnSe. Spectral windows where particular quantum dot materials emit, depending on their size, are illustrated in Figure 3. In each case, the shortest wavelength of emission represents that emitted from the smallest size of crystals investigated.
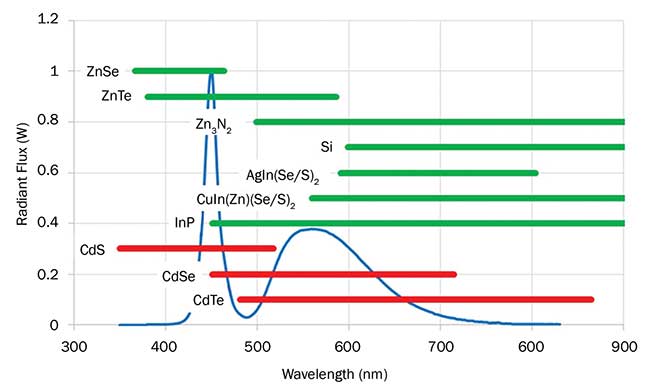
Figure 3. Emission tunability of a selection of visible light-emitting quantum dots (QDs) overlaid with a spectral power distribution of a commercially available 6500-K white LED (blue line). Red bars indicate QDs contain toxic elements, while green bars indicate nontoxic elements. Image property of Nanoco Technology.
The correct combination of quantum dots and phosphors lends itself well as a solution to the current issues facing lighting luminaire manufacturers. Utilizing the broad emission of a phosphor or phosphor blend, combined with a more narrow optimized emission of a quantum dot, provides a simple way to produce bright, desirable light that is both efficient (high LER/LES) and provides good color rendering (high CRI).
We demonstrated this in a luminaire, in which the tunability of the quantum dots was successfully utilized to finely optimize the performance of the lamp. The target specification for the lamp was 6500-K cold white light with CRI>90, maximized R9 and efficacy >100 lm/W. The R9 is part of the special CRI developed to provide a measure of how well saturated reds are rendered by a light source. This is of particular interest to the retail industry, where the correct rendering of items with bright, vibrant red colors is of great importance. It’s also a metric where light sources using broad-emitting phosphors will often underperform.
To test the performance of the luminaire, a red down-converting quantum dot film was paired with a blue-green light engine. The blue-green LEDs and red film were prepared in a complementary fashion such that when combined, the light of the correct specification was produced. The initial trial used quantum dots that emitted red light with a peak wavelength of 627 nm; the resulting lamp had the following performance: CCT 6430 K, LER 274 lm/W, LES 91 lm/W, CRI 88, R9 37.5. Although the performance was acceptable, a computer simulation of spectral gap analysis suggested the use of red quantum dots with slightly shorter emission wavelength — in this case 623 nm — would produce superior results in terms of both LES, because of better overlap with the photopic eye sensitivity curve, and CRI, because of the higher relative intensity of red light over the most sensitive wavelength region for a 6500-K light source.
Considering this information, a new batch of quantum dots with shorter emission wavelength were synthesized via subtle changes to the preparation chemistry. Subsequently, a new red film was developed. When this new film was combined with the same blue-green LED light engine, the resulting lamp had an improved performance of CCT 6735 K, LER 281 lm/W, LES 101 lm/W, CRI 96, R9 94. Comparative spectra are presented in Figure 4, demonstrating how one can see that by shifting the red peak to a shorter wavelength positively affects the final luminaire spectrum and subsequent performance metrics.
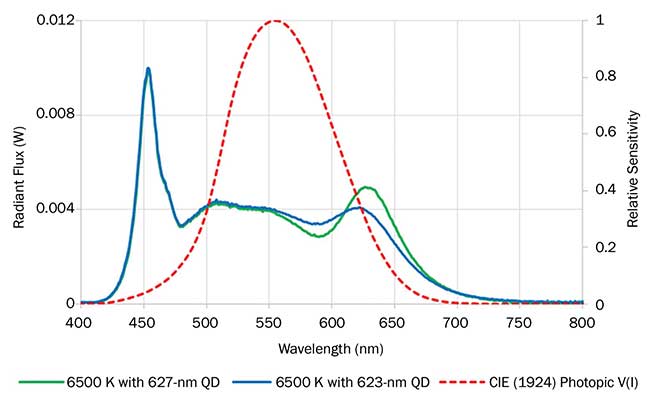
Figure 4. Spectral power distribution of a 6500-K luminaire using 623-nm red Cadmium-Free Quantum Dot (QD) film (blue), and spectral power distribution of a 6500-K luminaire using 627-nm red Cadmium-Free Quantum Dot film (green). Photopic eye sensitivity curve (red). Image property of Nanoco Technology.
A bright future
The use of quantum dots as down converters in white LEDs or luminaires offers a remarkable level of tunability of the resulting spectral output. This enables a finer level of optimization than can be achieved with rare-earth phosphors currently available. It also minimizes the compromises needed to achieve a higher quality of lighting while maintaining an acceptable level of efficiency.
Cadmium-Free Quantum Dots open up new possibilities even further. Recent advancements in the materials science underlying quantum dots has enabled them to be developed at high scale without the use of cadmium, a highly regulated and toxic heavy metal. With this breakthrough and the new availability of Cadmium-Free Quantum Dots, this technology now offers an ideal platform for the lighting industry, specifically in application areas such as retail, architecture and horticulture.
The future for this new class of materials applied to LED lighting for industry advancements in color performance and energy efficiency has never looked so bright.
Meet the authors
James Harris is section head at Nanoco Technologies Ltd.; email: [email protected]. David Webb is team leader for QD-LED testing at Nanoco Technologies Ltd.; email: [email protected].