
Swept Source OCT Takes Optical Medical Imaging to the Next Level
Swept source optical coherence tomography enables much faster imaging for comprehensive wide-field, 4D screening of large organs or dynamic processes.
WOLFGANG DREXLER, MEDICAL UNIVERSITY OF VIENNA, MICHAEL MINNEMAN AND JASON ENSHER, INSIGHT PHOTONIC SOLUTIONS
There are a number of serious eye maladies that can only be properly diagnosed if ophthalmologists can visualize the vasculature of the eye in detail. It used to be that eye vasculature imaging required the injection of a contrast agent such as fluorescein and, just as it properly infused in the eye, the capture of a blue light picture. If the ophthalmologist knew to run the test, and if the patient didn’t react to the agent, and if the image was taken at the precise moment, then the vasculature could be observed. Changes in the vasculature could be monitored and abnormalities could be identified, allowing for the diagnosis of serious diseases such as diabetic retinopathy and early macular degeneration. These diagnoses are critical to affect the best treatment.
Today, thanks in part to third-generation swept source lasers, imaging the vasculature is as easy as taking two almost identical normal eye images and doing some signal processing.
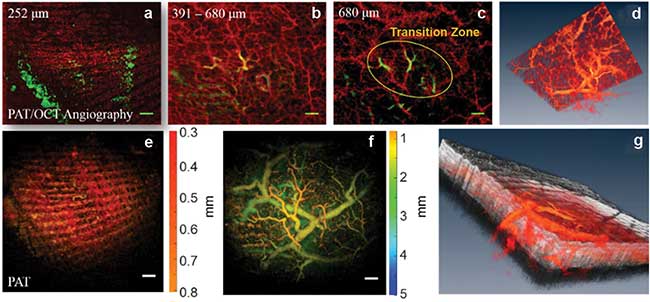
Skin angiography, with additional information and comparison to photoacoustic tomography (PAT), showing different depth ranges and a 3D visualization. Gray color maps optical coherence tomography (OCT) while red hot color map is used for blood vessels from OCT angiography (OCT-A) and PAT. Scale bar = 1 mm. Courtesy of the Medical University of Vienna (MUW).
Such changes are the reason why so many experts in the field are excited about the significant technical advances happening at the core of systems that perform medical imaging. It is likely that medical imaging systems will be able to run two to 20 times faster than they do today, and that the contrast, resolution, imaging depth and repeatability will all show noticeable and important improvements. And, as these core lasers transition from discrete component mechanical systems to entirely semiconductor systems, the costs decline substantially.
OCT evolution
State-of-the-art biomedical imaging relies on noninvasive, compact, maintenance-free, cost-effective technologies that perform quickly and reliably to reduce patient waiting time and ideally not use any exogenous contrast agents. In this context, optical coherence tomography (OCT) is one of the most innovative and successfully translated imaging technologies of the past two decades with substantial clinical and economic impact and acceptance in ophthalmology, cardiology and oncology in numerous organs. OCT’s high scanning and data acquisition speeds are important prerequisites for enabling the reduction of motion artifacts, such as those from breathing and eye movements, increasing the field of view, enabling wide-field imaging, detecting dynamic processes, as in 4D imaging, and finally enabling proper sampling to not miss subtle morphologic changes.
OCT consists of shining light, typically near-infrared (NIR), into an interferometer where one path illuminates tissue, referred to as the sample arm, and the other path is a reference reflection, known as the reference arm. The interferogram provides amplitude and phase information about the tissue. In a traditional OCT image, a depth profile at a particular point on the tissue is formed from the interferogram by computing the amplitude components of the Fourier transform. An optical system then steers the sample beam across a region of tissue to create a 3D map, or tomogram, of the tissue. In the 1990s, the first-generation OCT systems were time-domain OCT (TDOCT) that used broadband light sources and created interferograms by sweeping the mirror of the reference arm. Fourier/spectral domain OCT introduced in the early 2000s ushered in new levels of performance and also acceptance, increasing sensitivity/efficacy by two orders of magnitude over TDOCT by reading out a spectrometer in combination with a CCD line camera instead of a moving mirror and employing a pin detector.
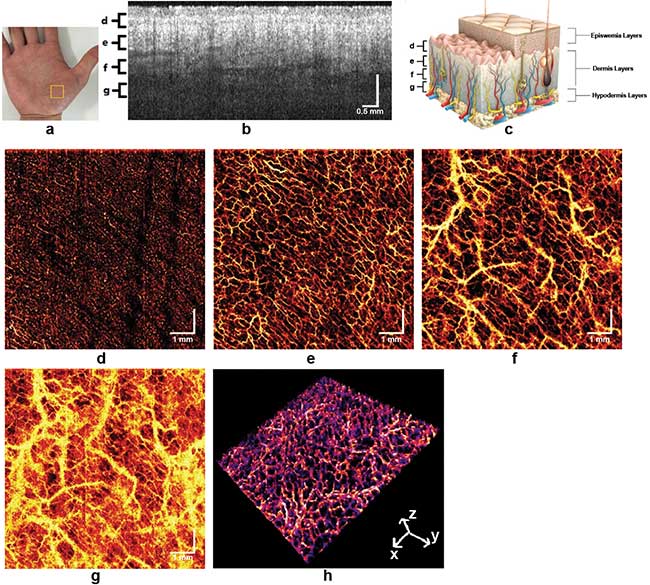
Skin angiography. Images taken using swept source OCT utilizing an akinetic all-semiconductor swept laser. Courtesy of the Medical University of Vienna (MUW).
A swept source light
The complexity and imaging speed was moved from a mechanically moving mirror in combination with a broad bandwidth light source and pin detectors to using the same light source but stopping the depth scanning mirror and changing the detector to a fast readout CCD camera. Consequently, with each exposure of the CCD line, an OCT-A scan was accomplished. The third generation of OCT — swept source OCT (SSOCT) — still does not need a scanning mirror but switches back to the cost-effective, dual balanced detectors. Additionally, the key technological element deciding the OCT scanning speed is the sweep speed of the light source. To a certain point the complexity of high-speed OCT is shifted to the swept source light source technology. SSOCT, on the other hand, becomes increasingly popular due to its potentially more compact fiber-based setup, its higher speed, better signal-to-noise ratio (SNR), and significantly less to no SNR decay with scanning depth. With spectral domain OCT (SDOCT) that range was 3-10 s dB per millimeter1.
While in frequency domain (FD) or SDOCT, scanning and data acquisition speed is mainly determined by the readout time of the CCD line camera in the spectrometer, the wavelength-tuning speed of swept sources over a broad bandwidth is the decisive factor in SSOCT. In the past decade, different swept source technologies have emerged to significantly improve imaging speed in commercial OCT systems — especially those using wavelengths greater than 1 μm. Also, SSOCT operates in the longer wavelength region, where scattering is less pronounced. That is why nonophthalmic applications benefited first from SSOCT. With the initiation of retinal OCT at 1050 nm, commercial ophthalmic OCT systems also benefited from this light source technology. Recent advancements of laser technology, such as vertical-cavity surface-emitting laser (VCSEL) sources, Fourier domain mode-locking (FDML) lasers operating in the MHz range, and akinetic lasers, further increase SSOCT’s attractiveness2,3,4.
The OCT technology of the future
Despite the emergence of new swept source technologies in recent years, it is noteworthy that none of them is, at the moment, even close in maturity and reliability to that of the superluminescent laser diodes (SLDs) that have been already employed in commercial OCT systems. Nevertheless, swept sources seem to be the OCT technology of the future since they have the potential to offer significantly higher scanning speeds, extended depth range with significantly reduced sensitivity roll-off, reduced fringe washout from sample motion or rapid transverse scanning, and improved light detection efficiency due to dual balanced detection. SSOCT avoids the need for spectrometers with line scan cameras, but it requires a high-speed, narrow line width swept source.

Swept lasers are being manufactured from wafer-scale semiconductors, dramatically reducing costs while improving performance. A single wafer carries over 2,000 lasers. Courtesy of Insight Photonic Solutions.
Spectrometers have limited spectral resolution due to the grating resolving power, beam spot size and finite pixel dimensions of the line scan cameras. A narrow line width swept source enables spectral resolution in SSOCT, which can be much higher than that of SDOCT. For OCT applications using wavelengths greater than 1 μm, SSOCT seems to be the most promising technology of choice because indium gallium arsenide (InGaAs) line cameras are limited in speed and number of pixels, and they are more expensive than silicon line cameras. SDOCT suffers from significantly more SNR roll-off with scanning depth than that achievable by using narrow line width swept sources.
Higher OCT scanning speed enabled by SSOCT has numerous advantages: 1. Motion artifacts will be avoided, especially in ophthalmic or cardiologic applications; 2. Comprehensive imaging of larger hollow organs enables wide-field imaging for the detection of early subtle morphological tissue changes over a larger field of view; 3. Wide-field-of-view retinal OCT angiography (OCT-A) similar to fluorescence or indocyanine green angiography; 4. 4D imaging of dynamic changes for intraoperative OCT in the retina or the brain; and 5. Flexibility for functional extensions such as Doppler OCT for blood flow monitoring5.
OCT angiography
OCT-A is a recently emerging functional extension of OCT, which enables noninvasive label-free visualization of tissue vasculature and hence perfusion exploiting the motion contrast of moving blood cells. Imaging vasculature is especially important for diagnosing certain conditions. For example, in diabetic retinopathy microaneurysms and irregular vasculature formation are all-important indicators of the disease and its progression. As another example, cancerous tumors often recruit vasculature to feed their growth.
The working principle of OCT-A is to sense dynamic structural changes in-between successive tomograms taken at the same location. The easiest way is to calculate the average of pairwise or, in what is known as intensity-based OCT-A, sets of differences of a set of intensity tomograms rapidly taken at the same position. Implementation on a graphics-processing-unit-based system allows real-time imaging of 3D angiograms in vivo. Otherwise, in what is known as phase-sensitive OCT-A, one can use phase information of the OCT signal and the motion contrast is achieved by taking the average of pairwise phase differences across a set of phase tomograms at the same location.
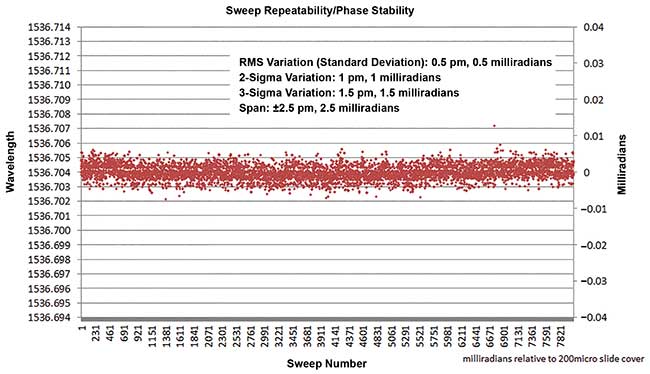
Intra- and inter-sweep repeatability becomes critical for angiography with OCT-A. New lasers exhibit order-of-magnitude or more increase in repeatability compared to traditional mechanical lasers. From W. Drexler and J. G. Fujimoto (2015). Optical Coherence Tomography — Technology and Applications. 2nd ed., Switzerland: Springer International Publishing.
On the other hand, the OCT signal phase is highly sensitive to motion in the subwavelength region, which results in stripe-like motion artifacts that obstruct vasculature. Consequently, it is necessary to correct bulk motion prior to motion contrasting, especially for in-vivo measurements that increase the post-processing effort. The advantage of phase-sensitive OCT-A is its independence from backscattering intensity changes, yielding potentially better vascular contrast for highly scattering tissue. A hybrid of both methods is to calculate differences of the full complex OCT signal or of spectral data6,7,8,9,10.
If SSOCT is used for phase-sensitive OCT-A, dedicated hardware such as fiber Bragg gratings, reference interferometers and sophisticated post-processing routines are necessary to ensure a wavelength-stable sampled signal. Any trigger jitter could deteriorate the signal phase and result in errors in depth dependent phase, which are hard to eliminate in post-processing alone. In one new laser source, the akinetic all-semiconductor laser, the intrinsic phase stability of the sweep virtually eliminates the need for additional components and post-processing required by other swept source technologies. The all-semiconductor sweeps are highly repeatable, and stable to vibrations or environmental changes, leading to extremely phase-stable tomograms near the shot-noise limit without additional parts or computations. Currently, most OCT-A systems are based on spectral OCT because of the reduced system complexity11,12,13.
The road to affordable, clinically important imaging
SSOCT will become much less expensive as the core lasers transition from mechanically based elements to all-semiconductor akinetic systems. Because of wafer-scale fabrication, it is likely that these all-semiconductor laser systems will be available in volumes for a few thousand dollars. System integration of detection, data acquisition and computing platform will only add incrementally to this cost due to both photonic and electronic integration, yielding a low-cost OCT engine. The combination of performance advantages and rapidly reduced cost will allow SSOCT to enable OCT-A to realize its full potential as a clinically important and commercially successful medical imaging technique.
Meet the authors
Wolfgang Drexler is the head of the Center for Medical Physics and Biomedical Engineering at the Medical University of Vienna in Austria; email: [email protected]. Michael Minneman is the president of Insight Photonic Solutions in Lafayette, Colo.; email: [email protected]. Jason Ensher is the CTO and vice president of strategic marketing at Insight Photonic Solutions; email: [email protected].
References
1. W. Drexler and J. G. Fujimoto (2015). Optical Coherence Tomography — Technology and Applications. 2nd ed., Switzerland: Springer International Publishing.
2. D. D. John, et al. (2015). Wideband electrically pumped 1050 nm MEMS-tunable VCSEL for ophthalmic imaging. Journal of Lightwave Technology, Vol. 33, pp. 3461–3468.
3. R. Huber, et al. (2006). Fourier domain mode locking (FDML): A new laser operating regime and applications for optical coherence tomography. Optics Express, Vol. 14, pp. 3225–3237.
4. M. Bonesi, et al. (2014). Akinetic all-semiconductor programmable swept-source at 1550 nm and 1310 nm with centimeters coherence length. Optics Express, Vol. 22, pp. 2632–2655.
5. Drexler (2015).
6. I. Grulkowski, et al. (2009). Scanning protocols dedicated to smart velocity ranging in spectral OCT. Optics Express, Vol. 17,
pp. 23736–23754.
7. A. Mariampillai, et al. (2008). Speckle variance detection of microvasculature using swept-source optical coherence tomography. Optics Letters, Vol. 33, pp. 1530–1532.
8. K. K. C. Lee, et al. (2012). Real-time speckle variance swept-source optical coherence tomography using a graphics processing unit. Biomedical Optics Express, Vol. 3, pp. 1557–1564.
9. D. Y. Kim, et al. (2011). In vivo volumetric imaging of human retinal circulation with phase-variance optical coherence tomography. Biomedical Optics Express, Vol. 2, pp. 1504–1513.
10. L. An, et al. (2010). Ultrahigh sensitive optical microangiography for in vivo imaging of microcirculations within human skin tissue beds, Optics Express, Vol. 18, pp. 8220–8228.
11. W. Choi, et al. (2013). Phase-sensitive swept-source optical coherence tomography imaging of the human retina with a vertical cavity surface-emitting laser light source, Optics Letters, Vol. 38, pp. 338–340.
12. B. Braaf, et al. (2011). Phasestabilized optical frequency domain imaging at 1-μm for the measurement of blood flow in the human choroid, Optics Express, Vol. 19, pp. 20886–20903.
13. B. Vakoc, et al. (2005). Phase-resolved optical frequency domain imaging. Optics Express, Vol. 13, pp. 5483–5493.
/Buyers_Guide/Insight_Photonic_Solutions_Inc/c19785
Published: September 2016
Glossary
- optical coherence tomography
- Optical coherence tomography (OCT) is a non-invasive imaging technique used in medical and scientific fields to capture high-resolution, cross-sectional images of biological tissues. It provides detailed, real-time, and three-dimensional visualization of tissue structures at the micrometer scale. OCT is particularly valuable in ophthalmology, cardiology, dermatology, and various other medical specialties.
Here are the key features and components of optical coherence tomography:
Principle of...
- swept source optical coherence tomography
- Swept-source optical coherence tomography is an imaging technique used in medical diagnostics, particularly in ophthalmology, to visualize and analyze the internal structures of biological tissues, especially the eye's retina.
Key aspects of SS-OCT are:
Optical coherence tomography (OCT): OCT is a non-invasive imaging technology that provides high-resolution, cross-sectional images of biological tissues. It uses low-coherence interferometry to measure the echo time delay and magnitude of...
- optical coherence tomography angiography
- Also known as OCT-A, optical coherence tomography angiography is an imaging technique that uses light waves to measure changes in the reflectance characteristics, enabling the the quantitative analysis of vasculature. Primarily used in ophthamology, OCT-A is a noninvasive alternative to dye-based techniques for blood vessel imaging, such as indocyanine-green angiography and fluorescein angiography.
- ophthalmology
- Ophthalmology is a branch of medicine that focuses on the anatomy, physiology, and diseases of the eyes and visual system. Ophthalmologists are medical doctors who specialize in the diagnosis, treatment, and prevention of eye disorders and diseases. They are trained to provide comprehensive eye care, including medical, surgical, and optical interventions.
Key areas within ophthalmology include:
General eye care: Ophthalmologists perform routine eye examinations to assess visual acuity,...
camerasCCDImagingsemiconductor lasersBiophotonicsLasersoptical coherence tomographyOCTswept source optical coherence tomographyssoctoptical coherence tomography angiographyoctaWolfgang DrexlerMichael MinnemanJason EnsherInsight Photonic Solutionsophthalmologyvasculaturefluorescence angiographyFAindocyanine green angiographyicgaakinetic all-semiconductor laserretinal diseaseFeatures