An optoacoustic tomography platform for imaging neural activation deep in the brain has enabled researchers to observe the activation of large neural circuits, currently up to the size of a small-animal brain, in real time and 3D. The platform can record 100 volumetric frames per second across scalable fields of view ranging between 50 and 1000 mm with respective spatial resolution of 35 to 200 µm. To date, optical imaging of large distributed neural populations has been limited to modest scanning depths and volumes.
The technique, called functional optoacoustic neuro-tomography (FONT), applies the principles of optoacoustics to fluorescent calcium markers typically imaged by light-sheet microscopy and similar modalities during neural activity.
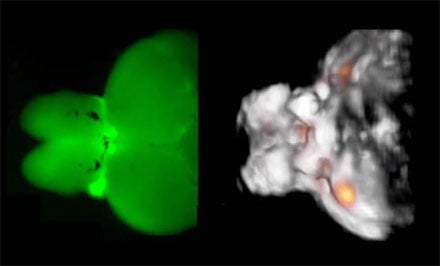
The figure shows typical images acquired from a highly scattering structure of an adult zebra fish brain. The fluorescence image on the left has a very blurry appearance indicating that the intense light scattering in large brains makes them inaccessible by optical microscopy methods. In contrast, functional optoacoustic tomography (image on the right) is able to provide high-resolution three-dimensional information regarding real-time neuronal activity (orange dots) in the entire scattering brain. Courtesy of Helmholtz Zentrum München.
"We discovered that optoacoustics can be made sensitive to the differences in calcium ion concentrations resulting from neural activity and devised a rapid functional optoacoustic neuro-tomography [FONT] system that can simultaneously record signals from a very large number of neurons," said Xosé Luis Deán-Ben.
The experimental FONT platform, developed by a research team at Helmholtz Zentrum München and the Technical University of Munich, was used to image calcium activity in both immobilized and free-swimming zebra fish larvae, bred to exhibit expression of a particular calcium sensor in a large fraction of neurons. It was also applied to isolated brains from larger adult zebra fish.

Experiments on brains of adult zebra fish expressing genetically encoded calcium indicator GCaMP5G demonstrated the ability to directly track neural dynamics, overcoming the longstanding penetration barrier of optical imaging in opaque brains. The FONT technique was also used to trace neural activity during unrestrained motion of the animals.
"We hypothesized that some fluorescent genetically encoded calcium indicators (GECIs) could provide a suitable calcium-dependent optoacoustic contrast," commented the team in its paper. "Our study is, to the best of our knowledge, the first to examine the optoacoustic signature of modern GECIs, showing that the strong changes in fluorescence are directly related to that signature."
"So far we have demonstrated real-time analysis on whole brains of adult animals of roughly 24 mm3," said Daniel Razansky. "Thanks to our method, one can now capture fast activity of millions of neurons simultaneously.”
Current state-of-the-art optical microscopy methods are limited to volumes well below a cubic millimeter when it comes to imaging of fast neural activity, according to the researchers. The team believes the FONT method to be capable of visualizing volumes of more than 1000 mm3 with temporal resolution of 10 milliseconds.
Large-scale observation of neural activity is one of the keys to understanding how the brain works, both under normal and diseased conditions. The ability of FONT to simultaneously track movements and neural activation in 3D from living unrestrained organisms could form a basis for behavioral studies not possible with current optical neuroimaging techniques. FONT may also accelerate development of novel therapies targeting neurological and neuropsychiatric disorders.
The research was accepted for publication in Light: Science & Applications (doi:10.1038/lsa.2016.201).