The results of computer simulations depicting various laser wavelengths aimed at virtual bacterial colonies buried in gum tissue suggest that use of lasers in oral debridement is effective in killing disease-causing bacteria and promoting dental health. Selective killing of pathogens by laser is possible due to the difference in absorption of photon energy by the pathogens and the host tissues.
Researchers at New York Institute of Technology (NYIT) created mathematical models based on optical characteristics of gum tissues and bacteria, then simulated the use of three different types of lasers and their effects on two types of bacterial colonies of various sizes and depths within the gum models.
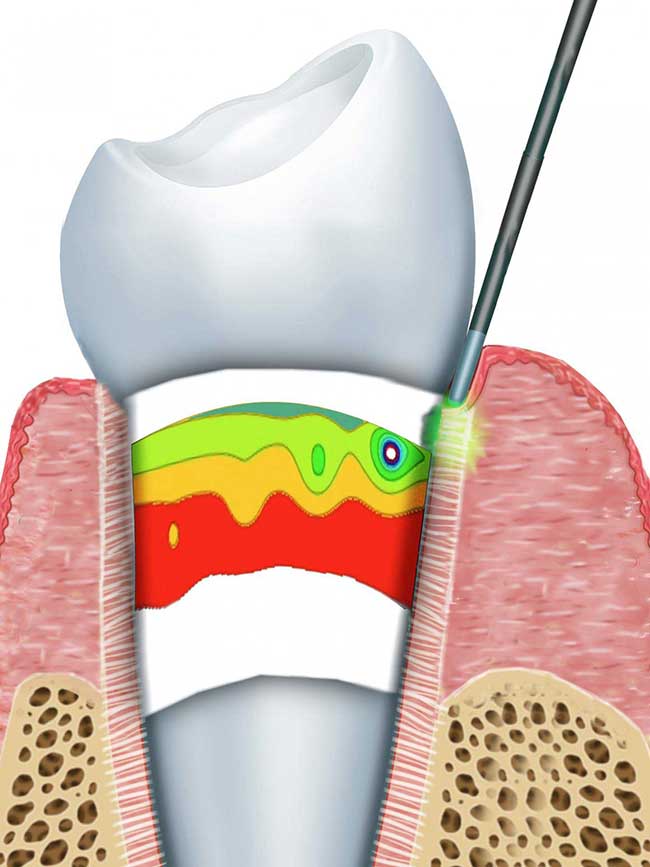
This is the cylindrical geometry of the periodontal ligament. Courtesy of Lou Reinisch/NYIT.
"One of the questions we asked is how deep could the bacteria be and still be affected by the laser light,” said Lou Reinisch, associate provost for academic affairs at NYIT.
The researchers created a “virtual periodontium,” a computational model of the optical and time-dependent thermal properties of infected periodontal tissues and the known optical properties of soft tissues. Their objective was to define the laser parameters that optimize pathogen destruction and the depth of the bactericidal effect.
The model simulated laser sulcular ebridement, a periodontal procedure. Virtual pathogen colonies were placed at different depths in the virtual periodontium to determine the depth for effective bactericidal effects given various laser parameters including wavelength, peak power, pulse duration, scan rate, fluence rate and differences in pathogen sensitivities.
Accumulated background heat from multiple passes increased the depth of the bactericidal effect. In visible and NIR wavelengths the large difference in absorption between normal soft tissue and Porphyromonas gingivalis (Pg) and Prevotella intermedia (Pi) resulted in selective destruction. Diode laser efficacy and depth of the bactericidal effect were variable and dependent on hemin availability. Both pulsed-Nd:YAG and the 810 nm diode lasers achieved a 2 to 3 mm deep damage zone for pigmented Pg and Pi in soft tissue without surface damage. The model predicted no selectivity for the Er:YAG laser . Depth of the bactericidal effect was highly dependent on pathogen absorption coefficient.

Short pulse durations enabled confinement of the thermal event to the target. Temporal selectivity was achieved by adjusting pulse duration based on target size. In the case of both 810-nm diode and 1064-nm Nd:YAG lasers, the simulations showed minimal heating of the surrounding tissue.
“Minimizing the thermal damage leads to faster healing,” said Reinisch.
“The findings are important because it opens up the possibility of tweaking the wavelength, power, and pulse duration to be the most effective for killing bacteria,” said Reinisch. “The doctors will look at this and say, 'I see there is a possible benefit for my patients in using the laser.'”
The cost of dental lasers can range from $5,000 to over $100,000, according to Reinisch, and health care professionals require extra training to use them. These costs are passed on to the patient so, he notes, there must be a definite benefit for the patient to justify these costs.
“The study reveals what's going on in the tissue, so I hope that we're educating the medical professionals by demonstrating that you can do a good job of killing bacteria with certain lasers,” said David Harris, director of Bio-Medical Consultants, Inc. “When you do this treatment, you remove an infection and allow tissue to regenerate. Getting rid of the infection means the tissue can heal without interference.”
The study's methodology of simulating how laser light interacts with tissue has implications beyond dentistry; physicians and surgeons use lasers in various treatments, including vocal cord procedures and dermatological treatments, including those for toenail fungus.
Guided by the results presented in the study, Reinisch and Harris expect that clinical trials will be designed to validate the findings.
The research was published in Lasers in Surgery and Medicine (doi: 10.1002/lsm.22568).