Coherent anti-Stokes Raman scattering (CARS) microscopy historically has struggled to break into a marketplace already crowded with popular imaging techniques such as fluorescence microscopy and with new superresolution techniques such as STORM (stochastic optical reconstruction microscopy) and STED (stimulated emission depletion microscopy).
Although fluorescence microscopy may still be the most commonly used biological imaging tool, it is not without its drawbacks: It is severely limited by the artifacts, photobleaching and cytotoxicity caused by the labels it requires.
Not that CARS is without drawbacks. It cannot provide the clarity offered by many established imaging methods. There is also the huge issue of cost: On average, a CARS microscope is several times more expensive than any other advanced optical microscope system currently on the market. In a tough environment for research funding, it can be a difficult stretch to purchase a CARS microscope.
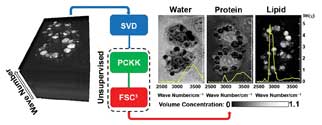
Sketch of CARS hyperspectral image analysis as reported in Analytical Chemistry 2013, Vol. 85, Issue 22, pp 10820-10828. The method provides an unsupervised quantitative chemical analysis of the most significant cellular components with spectra given by water, lipid and proteins, and it reveals the spatially resolved chemical structure of cells. SVD = singular value decomposition; PCKK = phase-corrected Kramers-Kronig method; FSC3 = factorization into susceptibilities and concentrations of chemical components. Courtesy of Paola Borri.
But CARS does possess one crucial advantage: label-free imaging – meaning, the sample remains unaffected.
This makes CARS most suitable for applications where labeling is not possible (pharmaceutical research, food processing, living organisms) as well as for many medical applications where labeling is highly undesirable (tissue engineering, large-scale neuronal imaging), and there are even places where labeling is impossible (imaging of water).
It is these areas of research that are beginning to embrace CARS microscopy and, in turn, make improvements to the technique. So much so that many believe it to be on the verge of mainstream use by researchers and expected to extend to clinical use.
“CARS microscopy is a mature technology. It is ready for mass-market adaptation. It should be available in all optical imaging facilities,” said Dr. Thuc Le, assistant research professor at Desert Research Institute in Las Vegas. “It is important to stress that a CARS microscope is able to perform simultaneous vibrational imaging, two-photon fluorescence imaging, second-harmonic generation, third-harmonic generation and others.”
The multimodality of a CARS microscope expands the number of biomolecules that can be visualized simultaneously, which is critical for interrogation of complex, multivariable biological systems.
Top applications of CARS microscopy
CARS microscopy has a number of fascinating applications, ranging from materials science to in vivo investigations of small animals. But perhaps most interesting of all, CARS has proved particularly successful in imaging unstained lipids; from artificial membrane model systems, living cells and tissues to whole organisms.
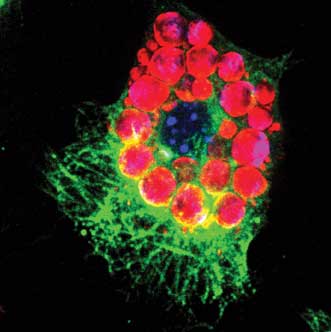
A murine stromal embryonic stem cell OP9 is differentiating into a fat cell. Image was obtained with CARS visualization of lipid droplets (red) and multicolor two-photon fluorescence visualization of nucleus (blue) and α-tubulin (green). Courtesy of Dr. Thuc Le, Desert Research Institute, Las Vegas.
For neurodegenerative applications, CARS turns out to be a very powerful tool because it offers a perspective on the spinal cord and brain not previously possible. Since the myelin sheath that coats nerve axons is lipid rich, it provides a strong CARS contrast.
“It is possible to monitor degeneration in a live animal over an extended period of time, something that is impossible otherwise because of the need for staining, which is only moderately compatible with in vivo imaging (lipophilic dyes) and not really compatible with longitudinal imaging (imaging the same animal over time),” said Dr. Daniel Côté of Laval University in Quebec City. “The high spatial resolution and high specificity of CARS imaging for myelin, because of its lipid content, is a game-changing technology.”

Transverse section of mouse spinal cord from the cervical region. Imaging modalities are reflectance (green) and CARS (red). Nerve fibers in the white matter are surrounded by myelin, a lipid-rich substance that appears as numerous ringlike structures in the image. The spinal cord mosaic on the left side is the result of having stitched more than 400 images together. On the right side, three enlarged regions are shown as a means to put emphasis on the ringlike structures. Size = 2 mm. Courtesy of Erik Bélanger, Sophie Laffray and Daniel Côté.
CARS microscopy also has been used to investigate the effect of cholesterol-lowering drugs, and to image the intestine, the fat droplets in intact liver tissue, and the breast and lung tissue for cancer diagnosis.
An upcoming area of biomedical interest is the study of nanoparticle uptake in cells and tissues, according to a Progress in Lipid Research review article by Paola Borri and colleagues at Cardiff University in Wales (doi: 10.1016/j.plipres.2013.07.003). The motivation for these studies is either an interest in the toxicity of nanoparticles or in their pharmaceutical application.
Borri and colleagues are currently investigating hyperspectral CARS imaging in which a CARS spectrum is measured at each spatial position either by taking a series of spatially resolved images at different vibrational frequencies or by acquiring a spectrum at each spatial point following simultaneous excitation of several vibrations.
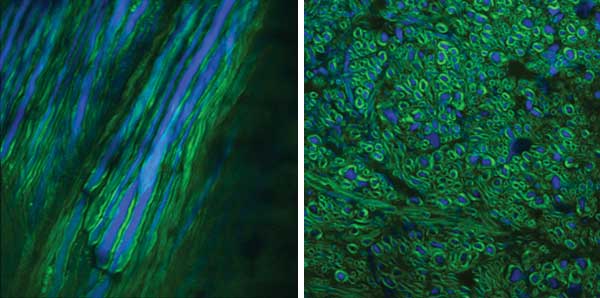
Images of spinal cord in a transgenic mouse. Nerve fibers (blue) express yellow fluorescent protein, and myelin surrounding these fibers generates a strong CARS signal (green). Two perspectives are shown: a longitudinal view after surgical exposure for an in vivo measurement (a) and a transverse section (b). Courtesy of Amy Daradich, Erik Bélanger, Benoit Aubé and Daniel Côté.
CARS technology progress
Dr. Herman Offerhaus at the Optical Sciences group at Twente University in Enschede, Netherlands, believes that CARS has not found widespread acceptance because it is still relatively complex, requiring multiple sources (or a chain of sources) and some advanced electronics. “Nothing earth-shattering, but it is a bit more complex.”
But in the past five years, a lot of effort has been spent on simplifying the technology, improving sensitivity and increasing its speed. Spectral labeling and high-speed detectors are making CARS much more useful, and arrays that allow for spectral lock-in detection of many channels simultaneously also have been developed.
“The sensitivity issue has two sides: the improvement of detection of the signal and the rejection of the nonresonant part of the signal,” Offerhaus said. “The latter, sometimes referred to as selectivity, is especially important when looking at low-density compounds (minority species).”
Perhaps the most notable advance to have emerged in the past five years is stimulated Raman scattering (SRS), either stimulated Raman gain or stimulated Raman loss. SRS is proving a very interesting variant of CARS that is nominally free of the nonresonant background that plagues CARS.
Although SRS is free of the purely nonresonant CARS signal, it is not truly background-free because other processes occur that cause a background. “SRS techniques probe changes in the intensity of the excitation laser beams due to energy transfer from the laser fields to the vibrational states instead of probing the vibrational coherence rising from lasers-sample interaction,” Le said.
But CARS has an advantage over SRS, in that the signal contains the phase of the molecular vibration. This phase can be used to reject the nonresonant signal, but it also can be used to enhance the selectivity and extract other features. Offerhaus believes that the advantages of having access to the phase has made a number of people come back to CARS and might be an important feature in the future. (For more on CARS-related technological advances, see the sidebar.)
What’s next for CARS?
The steady increase in sensitivity and imaging speed that we have seen over the past five years is expected to continue, but the push for more user-friendly systems also will eventually lead to more widespread application, particularly for chemometrics (spectral decomposition).
Either fiber sources, or simply better, more-robust laser sources are set to make a real difference to CARS users, and integration in clinical systems via CARS endoscopy is a clear directional trend.
“Challenges are in the development of more user-friendly setups and data analysis methods, to make the technology really applicable to biologists, but we are getting there,” Borri said. “I think biomedical applications will [benefit] more and more from CARS microscopy in the next few years.”
Other notable technical advances
CARS has seen a number of advances over the years, including:
• Hyperspectral imaging (to obtain information over a broad range to better identify multiple species):
• Fast spectral scanning of picosecond pulses.
• The use of broadband pulses.
• The use of chirped femtosecond pulses.
• Large-area CARS microscopy:
• Professor Sunney Xie’s group at Harvard University has shown brain imaging over several centimeters.
• Increased speed (smarter modulation and detection):
• Professor Ji-Xin Cheng of Purdue University in West Lafayette, Ind., has developed spectral labeling with corresponding multiwavelength detection that allows the acquisition of many frequencies simultaneously.
• Video-rate image acquisition at a single frequency:
• Assistant professor Daniel Côté of Laval University in Quebec City demonstrates live imaging for medical applications in neurodegenerative disorders.
• Vibrational photoacoustic (VPA) imaging:
• VPA imaging uses light for vibrational excitation, then detects the resulting changes in sound waves to provide millimeter-scale penetration depth.
• Endoscopic CARS microscopy:
• Could be possible due to microendoscopy and lensless endoscopy.
• Pulse shaping:
• Spatial shaping improves penetration depth and the selective excitation of specific molecules.
• Fiber laser sources:
• Developments in fiber lasers offer the potential for robust and cheaper commercial systems that could greatly extend applications.