
2-Level Optogenetic Device Could Broaden Options for Large-Mammal Studies
An implantable microLED optrode array, developed by researchers at the University of Strathclyde and the University of Utah, is capable of exciting below-surface neurons in large mammal brains both by structured-light delivery and by large-volume illumination. Through its depth of access, heat control, and an electric delivery system that will be compatible with future wireless applications, the new array could advance the use of optogenetics in studies of large mammal brains.
To build the device, the researchers integrated a glass microneedle array with a custom-fabricated microLED device. The array of 181 80 μm × 80 μm2 microLEDs was fabricated on a 150-μm-thick GaN-on-sapphire wafer, coupled to a glass needle array on a 150-μm-thick backplane. To reduce stray light, the researchers patterned a pinhole layer on the sapphire side of the microLED array. An evaluation of the device’s light delivery and thermal properties showed that the device was capable of peak irradiances > 80 mW/mm2 per needle site.
The electrically addressable optrode array can provide 181 individually addressable illumination sites — 81 for surface illumination and 100 for illumination in intracortical layers. Each site can provide either high-resolution stimulation or larger-volume stimulation through control of the drive current, allowing a range of intensities. Several sites can be operated simultaneously to extend the illumination volume further or allow complex illumination patterns. The ability to produce spatiotemporal patterns of light at two layers within the cortex could help studies aimed at linking the neural activity of small groups of neurons to specific cognitive functions.
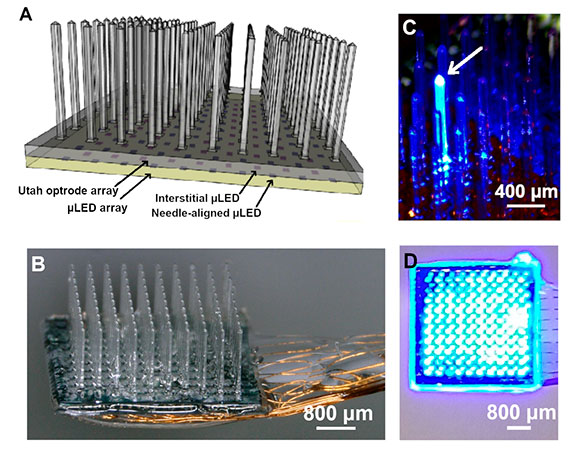
A) Schematic of the integrated device with the glass Utah Optrode Array (UOA) bonded to a microLED array. Illumination is from the microLED, through the sapphire, which is bonded to the glass UOA, and delivered to tissue either by the glass needles or through interstitial sites. A pinhole layer (not shown) is patterned onto the sapphire substrate of the microLED array before bonding to reduce optical crosstalk. B) Completed device with polymer-coated wire bonds permitting independent control of the microLEDs. C) Illumination of a single microLED delivering high-intensity light to the needle tip. D) All 100 sites simultaneously illuminated. Courtesy of Niall McAlinden et al.
This large array for multisite optical stimulation could enable stimulation of multiple cortical sites simultaneously or sequentially. It could also open the possibility of stimulating cortical sites with different wavelengths — and therefore different cell types — at selected locations.
The research was published in Neurophotonics, a publication of SPIE, the international society for optics and photonics (https://doi.org/10.1117/1.NPh.6.3.035010).
Published: September 2019