Atomic force microscopy answers important fundamental and clinical questions.
David L. Shenkenberg, Associate News Editor
Some of the latest atomic force microscopy (AFM) research demonstrates that the technique can provide information about the molecular composition of protein complexes and can show how something as tiny as a retrovirus, such as HIV, replicates in real time. Using chemically modified probes, the method can elucidate the chemical characteristics of a pathogen’s surface, information that can be used to develop antibiotics against it.
Since the invention of the first AFM in 1986, the microscopy technique has proved an advantageous alternative. It produces images with spatial resolutions three orders of magnitude greater than those achievable with optical microscopy, enabling imaging of DNA and viruses. Although scanning electron microscopes (SEMSs) have comparable spatial resolution, they require a vaccuum chamber that can dehydrate biological samples. They also use an electron gun that can fry living cells, they require time-consuming sample preparation that can alter the natural state of biological samples, and they produce only 2-D images. AFMs can produce 3-D images of living cells in their natural environment.
Whereas AFMs provide only topological maps, light and scanning electron microscopy can show features beneath the surface. Also, the force of the probe tips used in AFM can damage living cells, and optical and scanning electron microscopy can image larger areas and in much less time compared with early AFMs. Nevertheless, recent improvements have enabled AFMs to produce images in real time.
AFM measures surfaces using a microscale cantilever with a probe on the bottom, and the fineness of the probe tip determines the spatial resolution. Forces between the tip and the surface deflect the cantilever, according to Hooke’s Law, which states that the force is equal to the change in position multiplied by a spring constant. Conventional probe tips measure surface topography and mechanics, and they also have been developed for measuring electricity, chemistry, magnetism and temperature.
To measure the deflection of the cantilever tip, the reflection of a laser beam off the cantilever is detected by two photodiodes housed in a photodetector. The difference between the photodiode signals indicates the position of the laser beam and, thus, the position of the cantilever.
Ties that bind
In a study performed by Yuri L. Lyubchenko and colleagues at the University of Nebraska Medical Center in Omaha and at the Institute of Biotechnology in Vilnius, Lithuania, electron microscopy provided the clues, and AFM revealed the mechanism by which a certain protein binds DNA. Lyubchenko said that the way specialized proteins bind the molecule of life has implications for many biological processes, including the recombination, integration and excision of genetic material. These DNA rearrangements happen on a regular basis, but mistakes in these events can cause diseases such as cancer.
When a protein binds DNA, it often binds the molecule at two sites and makes a single loop with it. Electron microscopy data from other researchers has shown that a protein can make two-loop structures, suggesting that these structures have a biological basis. Using electron microscopy, Lyubchenko’s group found that the EcoRII protein can make two-loop structures as well. Recent EcoRII studies led to the hypothesis that the protein binds three DNA sites to make the two-loop structure.
Lyubchenko said that he found this interesting because the three-site interaction could increase the probability of the formation of protein-DNA complexes. EcoRII is not only a protein but also an enzyme commonly used by molecular biologists and biochemists to cut DNA. Elucidating the three-site mechanism may enable the development of more efficient enzymes by other research groups that specialize in that field, he said.
The researchers studied the EcoRII-DNA complexes using an AFM system and silicon-etched tapping-mode probes from Veeco Instruments Inc. of Woodbury, N.Y. They performed AFM imaging in tapping mode, as opposed to contact or noncontact mode. Just as the names imply, the probe remains in contact with the surface in contact mode, it repeatedly taps the surface in tapping mode and it does not touch the surface in noncontact mode. Tapping and noncontact modes generally are used for delicate samples that might be damaged by continuous contact. DNA molecules with three EcoRII binding sites arranged almost symmetrically were used because three binding sites are needed for the enzyme to form two loops.
To perform AFM, the protein-DNA complexes must remain immobilized on the microscope stage; otherwise, the probe might pick up or drag the molecules instead of moving over them. Mica is commonly placed underneath samples on the stage, but its negative charge repels DNA. Therefore, the investigators made the mica surface positively charged with a 1-(3-aminopropyl)silatrane solution, a chemical treatment they developed, according to Lyubchenko.
Data analysis performed with Femtoscan Online software from Advanced Technologies Center of Moscow determined the length of the DNA fragments, the protein volume and the yields of the complexes. The researchers produced histograms showing the distribution of the length and volume measurements using Origin 6.0 from Originlab of Northampton, Mass.
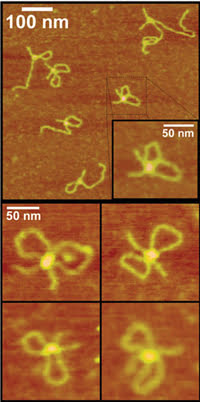
Figure 1. AFM revealed that a DNA-binding protein can form two loops of DNA. Reprinted with permission of Biochemistry.
As reported in the Oct. 2 issue of Biochemistry, analysis of the AFM data confirmed electron microscopy data by showing that two-loop complexes formed as well as single-loop complexes and nonlooped structures (Figure 1). Protein volume measurements directly showed that two EcoRII enzymes came together to form two loops with the DNA, whereas single enzymes made a single loop. Mutant enzymes with one end shortened, specifically the amino terminus, could not bind DNA as single units. On the other hand, mutant enzymes with a shortened carboxy terminus could bind as single units but could not form the double units needed to bind three sites and to make two loops.
Lyubchenko said that their results led them to hypothesize that a structural change within each enzyme creates a third site at the carboxy termini of the two enzymes, enabling the two-enzyme unit to bind a third site on the DNA (Figure 2). The researchers plan to investigate this hypothesis and to examine whether other DNA-binding proteins form two-loop structures. They also plan to measure the strength of the enzyme-DNA binding interactions using atomic force spectroscopy, also known as chemical force microscopy. To perform this technique, scientists coat the probe tip with certain chemicals. As the tip travels along the surface, it is deflected according to the relationship between the probe chemistry and the surface chemistry.
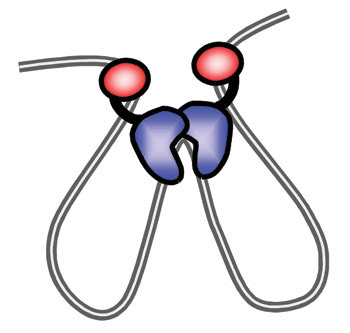
Figure 2. The AFM work led directly to this model of how the EcoRII enzyme binds DNA to form the two-loop structure. Reprinted with permission of Biochemistry.
Elucidating pathogenesis
Some situations call for chemical force. Researchers recently used chemical force microscopy to observe real-time changes in pathogenic microbes’ surface chemistry directly related to their ability to cause disease. The investigators hailed from Université Catholique de Louvain in Belgium and from the Paris and Lille sites of Institut Pasteur in France. They studied the mold Aspergillus fumigatus and the bacterium Mycobacterium bovis.
It is known that A. fumigatus spores travel through the air and adhere to the respiratory tract of the human body, where they germinate and cause disease, but exactly how they attach is not understood. Electron microscopy and biochemical data indicate that hydrophobic cell wall proteins possibly enable the mold spores to adhere to surfaces such as those of the respiratory tract. However, the data do not show how changes in the surface chemistry, particularly changes in the hydrophobicity and hydrophilicity of the cell wall surface, mediate the adhesion and the germination of the spores in real time, so the researchers turned to chemical force microscopy. Jean-Paul Latgé of Institut Pasteur in Paris said that this real-time surface chemistry information could be used to understand better the pathobiology of disease caused by A. fumigatus.
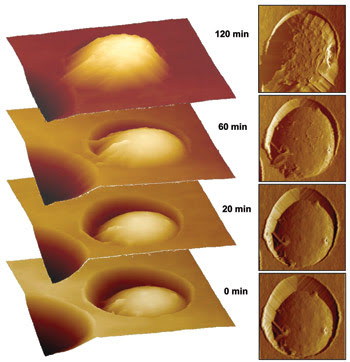
Figure 3. Using AFM, researchers observed the surface chemistry of the pathogenic mold Aspergillus fumigatus in real time as it germinated. Reprinted with permission of Biophysical Journal.
The researchers used Veeco instruments that included soft silicon nitride cantilevers and an AFM with a temperature-controlled stage, because A. fumigatus germinates at 37 °C.
To determine the surface chemistry of A. fumigatus, they used chemical force microscopy. Because they wanted to determine the relative hydrophobicity and hydrophilicity of the surface, they coated the probe tip with hydrocarbon chains, which are hydrophobic. In principle, hydrophobic regions of the surface will adhere strongly to the probe tip, whereas hydrophilic regions will adhere weakly. By doing so, chemical force microscopy can determine the hydrophobicity and hydrophilicity of molecules on the surface.
The researchers performed AFM imaging in contact mode because it achieves the best resolution with their cellular systems. The mold spores have hard cell walls that resist damage by contact with the probe. They also used gentle imaging forces of less than 0.1 nN.
Before germination, the investigators detected strong adhesion forces across the entire surface of the cell wall, indicating that the surface was hydrophobic. After two hours, they observed both strong and weak forces scattered across the surface, indicating a mixture of hydrophilic and hydrophobic compounds. After three hours, the surface had become almost totally hydrophilic. The results are detailed in the Sept. 21 online edition of Biophysical Journal.
The researchers believe that the spores probably remain hydrophobic in air but likely become hydrophilic as they germinate within the respiratory tract. Further experiments confirming these views are detailed in the October issue of Nano Letters, which also contains a description of how an antibiotic changes the surface chemistry of Mycobacterium bovis BCG, a weakened form of the tuberculosis-causing M. bovis virus. The BCG variant is used as a vaccine for the disease.
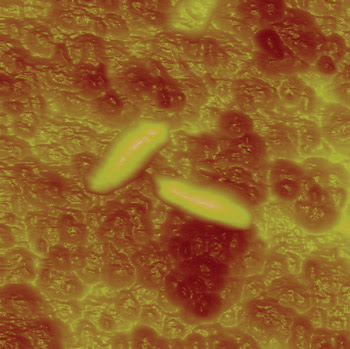
Figure 4. The technique enabled researchers to study the effect of an antibiotic on the surface chemistry of Mycobacterium bovis BCG.
The scientists treated M. bovis BCG with the antibiotic isoniazid and examined the surface of the bacterium with the AFM, using probe tips coated with hydrocarbon chains. They found that the surface was hydrophobic prior to treatment, whereas it became hydrophilic afterward. Yves F. Dufrêne, a researcher at Université Catholique de Louvain, said that this result indicates that AFM possibly could be used to screen for tuberculosis drugs routinely. Indeed, his group already has begun experiments using the instrument to investigate the effects of various drugs on M. bovis BCG. He said that, with 2 million deaths per year caused by tuberculosis, there is a need to develop novel approaches to define or screen new drugs.
Although his group successfully investigated microbial germination and the effect of an antibiotic in real time, he said he would like to see faster AFMs because the instruments currently take about 30 s to record data, whereas many biological changes can occur in milliseconds. Because AFM involves repeatedly moving a probe across a surface, the mechanics of this movement inherently limit the speed of the technique.
Imaging viruses in real time
Although faster instruments are always desirable, the speed of AFM certainly did not prevent researchers from investigating viruses exiting cells in real time with high resolution. Micha Gladnikoff and Itay Rousso from Weizmann Institute of Science in Rehovot, Israel, used the technique to monitor mouse and rat Moloney leukemia virus particles.
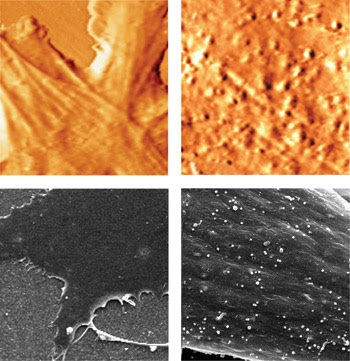
Figure 5. The top left AFM image shows a healthy cell and the top right, a cell infected with the mouse and rat Moloney leukemia virus. The bottom left and bottom right images were taken with an SEM for comparison and also show a healthy and an infected cell, respectively. Reprinted with permission of Biophysical Journal.
As with HIV, the Moloney virus is a retrovirus that exits the cell by a budding mechanism, which involves its assembly on the cell surface and subsequent release. All viruses take over the machinery of cells to make more viruses, and these virus copies must leave the cells once they are produced. The more virus copies there are in the body, the more the organism infected with the virus becomes sick.
Rousso said that AFM could be used to monitor any virus with a cell-surface budding mechanism. In contrast, electron microscopy requires fixation methods that preclude real-time monitoring, and fluorescence microscopy lacks the resolution to show viruses as much more than fluorescent blobs.
The researchers employed a Veeco AFM mounted on a Carl Zeiss inverted optical microscope to find the cells infected with viruses. They used fluorescence microscopy and positioned the AFM tip over them. Optical microscopy also enabled them to show that the AFM probe did not alter the cell morphology or harm the cells. Rousso said that the high temporal resolution of optical microscopy can complement AFM, although the temporal resolution of AFM was sufficient for real-time imaging in this case.
They performed AFM in tapping mode in liquid using pyramidally shaped silicon nitride cantilevers. They began with a 10 × 10-μm scanning area and used smaller scanning areas to zoom in on discrete features. Images were rendered with software from Nanotec Electronica of Tres Cantos, Spain. They maintained the cells in an incubator from Harvard Apparatus Inc. of Holliston, Mass., with a Newport temperature controller and a Carl Zeiss CO2 controller.
As reported in the Sept. 7 online edition of Biophysical Journal, the researchers found that budding took place either in more than 45 min or in less than 25 min. “The two kinetics phases may represent different viral assembly pathways,” Rousso said. They also observed structures that may aid in viral budding. Budding occurred from random sites, refuting earlier work with mutants that suggested that it occurs from unique sites.
“In the case of retrovirus budding, what has been done so far is to measure the overall amount of virus particles released to the medium as a function of time,” Rousso said. “Such measurement can’t pick up the two kinetics pathways we observed in our single virus particle analysis.”
He said they plan to examine whether the slow and fast kinetics pathways of budding events represent two different assembly pathways and that they will investigate the assembly structures that they observed. He added that preliminary results from his lab indicate that the assembly structures may be associated with the cytoskeleton.
Combining AFM with Infrared ATR Spectroscopy
Infrared attenuated total reflectance (ATR) spectroscopy can reveal overall changes in the chemistry of a sample, and AFM can show its topography. Combining the techniques permits simultaneous monitoring of the overall chemical changes and topography, enabling direct comparisons between the types of information gained from the techniques. The combination likely will provide further information about processes that are of broad importance to cell biology, biochemistry and medicine, such as protein folding, cell signaling and molecular interactions.
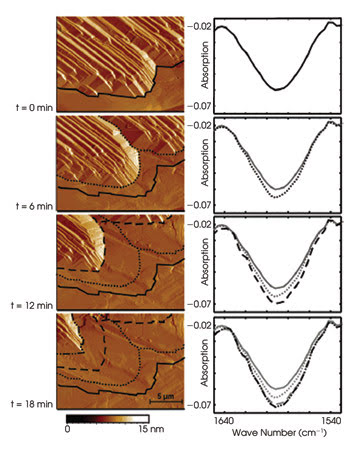
Changes in topology measured by AFM correlated well with spectral changes measured by ATR spectroscopy. Reprinted with permission of Analytical Chemistry.
Researchers from Georgia Institute of Technology in Atlanta combined AFM and infrared ATR spectroscopy into one system and used the system to observe the model process of solid urea dissolving in an organic solution. In ATR spectroscopy, a crystal waveguide typically is placed below a sample. In this case, the sample was in a chamber, and the crystal waveguide was at the bottom of the chamber. Radiation from a Fourier transform infrared spectrometer is focused onto the crystal such that the beam is reflected totally within the crystal, generating an evanescent field at the waveguide surface that can interact with sample molecules. The beam ultimately travels to a detector.
Fourier transform infrared spectroscopy often employs a Michelson interferometer for modulating the radiation interrogating the sample in the time domain. In evanescent field absorption spectroscopy, light waves passing through a sample are compared with light waves passing through a reference, which is usually the solvent without the sample. A Fourier transform is a mathematical method of transforming the obtained data from the time domain into the frequency domain, so that the spectra from the sample of interest are clearly resolved.
The researchers used a modular Fourier transform infrared spectrometer from L.O.T.-Oriel GmbH of Darmstadt, Germany, a liquid nitrogen-cooled photoconductive mercury cadmium telluride detector from Infrared Associates Inc. of Stuart, Fla., and an Agilent Technologies AFM.
As reported in the Nov. 15 issue of Analytical Chemistry, the investigators recorded infrared spectra at a rate of 60 per minute while using AFM to scan an area of 20 × 15 μm at a speed of 83 μm/s, for a total time of 20 min. They achieved a spectroscopic signal-to-noise ratio of >50 per scan and obtained spectral peaks matching the known peaks of urea. The AFM and IR-ATR spectral data correlated well. Therefore, they demonstrated the feasibility of the combined system.