Single-strand DNA sequences bind with a target and produce a fluorescent response that can be read through a smartphone.
Najeeb Khalid, Two-Photon Research
Aptamer-based diagnostics is a new field in the development of technologies for instantly identifying disease. Aptamers are short sequences (oligonucleotides) of DNA or RNA that bind to a specific target molecule. They are typically 20 to 60 nucleotides in length and are equivalent to antibodies in their ability to specifically bind to their targets. Scientists have recently capitalized on these features by creating an aptamer-based photonic beacon that can interface optically with a smartphone or any computing device with an LED and sensor.
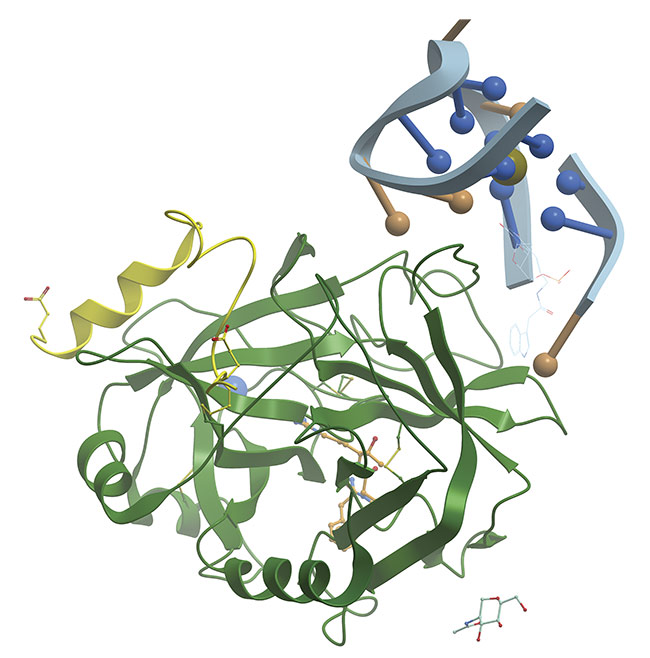
A thrombin-binding aptamer is a DNA sequence that folds into a G-quadruplex structure. Adapted with permission from Reference 1.
When compared to antibodies, which are traditionally used for disease detection, aptamers have a shorter generation time, lower manufacturing cost, high modifiability — meaning they only take a few weeks to develop, whereas antibodies take a year — better thermal stability, higher target potential, and, most importantly, no batch-to-batch variability. In general, aptamers demonstrate high sensitivity, meaning they have a high affinity for the intended target, and high specificity, meaning they have low interaction with unintended targets. Aptamers have been developed to bind with myriad targets, including small inorganic molecules, circulating proteins, viruses, and whole cells.
For diagnostics, specifically, aptamer-based beacons can identify and bind to viruses, bacteria, and biomarkers for cancer and other diseases. The beacons are also effective in binding with enzymes and many other types of molecules. Aptamer-based photonic beacons can be developed within a few months and tested through clinical trials. One example is the development of the diagnostics for detecting very low levels of the troponin2 enzyme in human blood. The time from the description of the sequence of the aptamer to the delivery from the manufacturer took 10 days, and then to initial testing took another two weeks, mainly waiting for the delivery of the antigen for testing.
An example of how aptamer-based photonic beacons can alter the health diagnostic landscape is through their incorporation into instant diagnostic tests that require no deciphering in a laboratory or by trained personnel. The results can then be transferred into the cloud, so no translation of paper-based data into digital data is required, which enables swift response to a potential pandemic or other urgent condition, providing valuable input into data-driven decisions that can lead to positive patient outcomes. Two cases that are particularly pertinent for this type of capability are the recent COVID-19 pandemic and cardiovascular disease2, which affects millions around the world.
The emergence and spread of COVID-19 spurred the discovery and development of aptamer-based photonic beacon technology. A team from Two-Photon Research was in South Asia during this time. The researchers realized that a simple, instant test — one that would take only seconds while connected to the internet — could help public health authorities to respond faster and track the pandemic. The CAST testing technology developed by the team involved small strands of DNA that would act like antibodies and could be engineered into molecular beacons that could easily be measured without a biochemical lab.
Selecting the right beacon
Aptamers can be selected for use as beacons via an in vitro process called SELEX (systematic evolution of ligands by exponential enrichment)3. The method can be refined through iterative testing or molecular modeling. Full in silico methods of designing aptamers are currently in development.
The important characteristics of aptamers that make them useful for diagnostics are the affinity (Kd), which is a measure of the strength with which the aptamer binds to the biomarker, and specificity, which defines the degree to which the aptamer binds only to the target.
Aptamers primarily bind to their targets through hydrogen bonding, though other electrostatic and hydrophilic interactions often support binding. As a result, high-affinity aptamers cannot be developed for some potential targets due to limited hydrogen bonding sites present on the target molecule. Chemically modified nucleotides can be used to generate aptamers for these targets.
Aptamer-based beacon
Finding an aptamer that will serve this function is just the start of the process, however. When an aptamer binds to the target biomarker, the result must be detectable. To make it detectable, an aptamer molecular photonic beacon must be created (Figure 1). This beacon will emit photons to announce the binding with the targeted biomarkers. The photons thus become detectable.
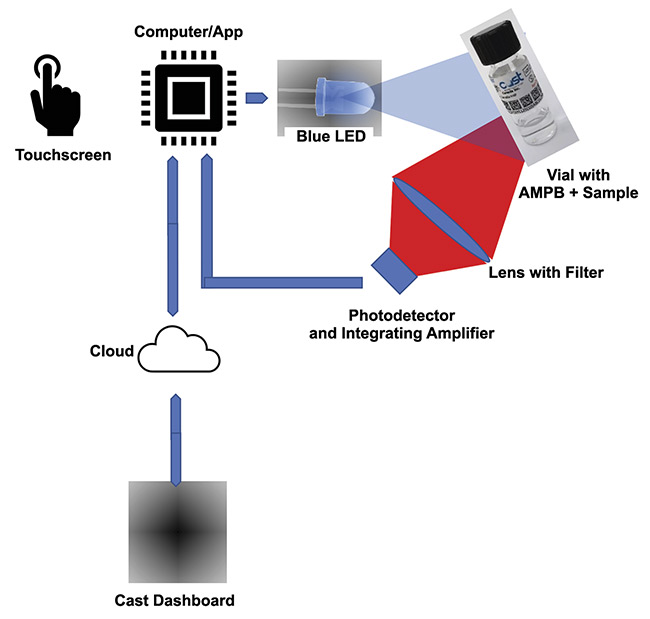
Figure 1. An illustration of how a fluorophore detection device works. AMPB: aptamer molecular photonic beacon. Courtesy of Two-Photon Research.
An aptamer molecular photonic beacon is made up of an aptamer that labeled with a fluorophore and an antisense (Figure 2) labeled with a quencher. The quencher and the fluorophore must be adjacent within the Förster radius (2 to 7 nm) and, when the aptamer binds to the target, they must separate. Unquenched fluorophores can thus provide a direct measure of the binding.
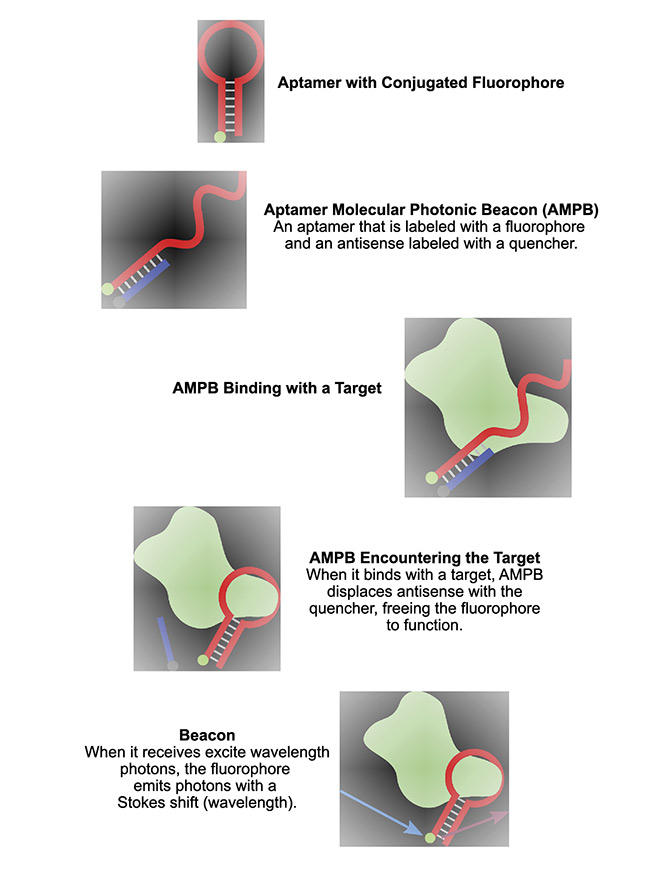
Figure 2. The construction and behavior of an aptamer molecular photonic beacon. Courtesy of Two-Photon Research.
Fluorophores are small molecules that absorb light of a specific wavelength (λex in fluorophore specifications) and emit light of longer wavelength (λem). Fluorophore molecules can be proteins, small organic molecules (1 kDa), or larger quantum dots (2 to 10 nm).
Fluorophores are conjugated to the aptamer after DNA synthesis. The DNA strands are synthesized with a terminal amine group (the end of a protein sequence), with which the ester group (a class of organic compounds) on the fluorophore readily conjugates.
Fluorophores absorb a photon and emit a photon with a Stokes shift (wavelength). The exciting wavelength λex is lower than the emission wavelength λem, and the difference is the Stokes shift. The difference in wavelength indicates a loss of energy. The quantum efficiency, or the ratio of photons emitted to the ones absorbed, of this function is usually 75%.
The excited wavelength of the fluorophore must be such that it does not interfere with the collection of the emitted photons. In practical terms, this means filters must be chosen to reduce any component of the excited source that may manifest in the emitting wavelength.
Increasing sensitivity
Saliva and blood both emit autofluorescence. For the best sensitivity in test results, it is important to mitigate this autoflorescence, or, in other words, to increase the signal-to-noise ratio (SNR) in the system. For this reason, the emission wavelength needs to be separate from the excitation wavelength. Therefore, the Stokes shift has to be high so that the emission wavelength is separated on the spectrum from the autofluorescence of saliva and blood. Figures 3 and 4 show the autofluorescence of saliva and blood. Figure 5 shows the photon excitation and emission process.
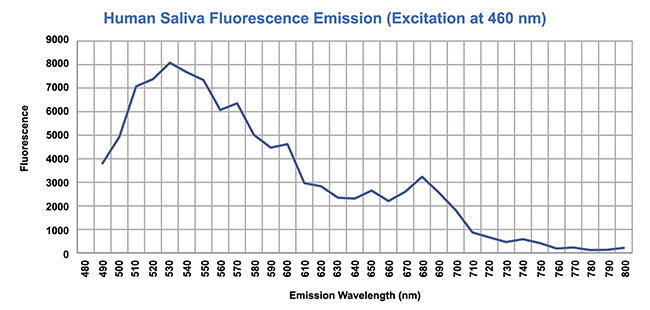
Figure 3. Mitigating the autofluorescence detected in human saliva. Courtesy of Two-Photon Research.
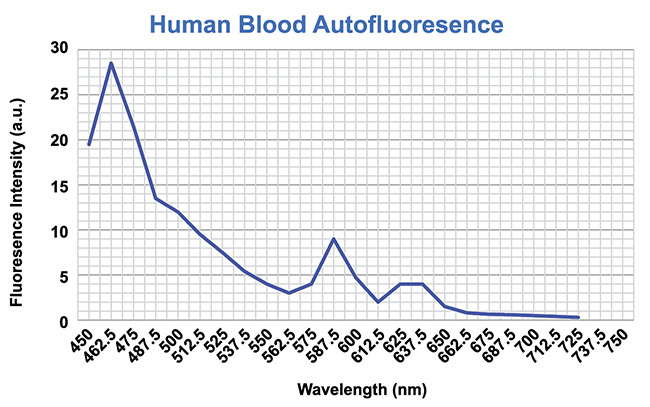
Figure 4. Mitigating the autofluorescence detected in human blood. Courtesy of Two-Photon Research.
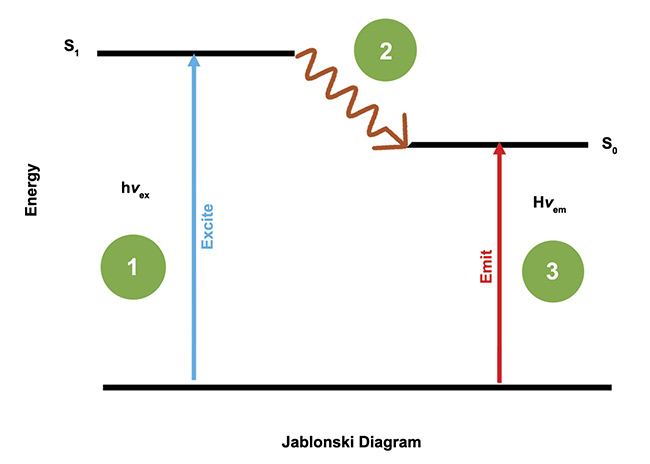
Figure 5. The process of exciting a fluorophore and the photons it emits as a result of the excitation. Courtesy of Two-Photon Research.
Incorporating antisense
An antisense strand, also called a probe strand or an aptamer complementary element, is a short sequence of nucleic acids that is partially complementary to an aptamer in the binding process. Antisense strands are typically 8 to 16 nucleotides in length.
The antisense strand’s affinity for the aptamer is modulated by decreasing its length or introducing mismatched nucleotides to its sequence. The strand is designed to have lower affinity to the aptamer than the intended target. As a result, the target molecule displaces the antisense strand. This displacement can be used for signal transduction — that is, the consequence of binding generates the signal.
Fluorescence quenchers are molecules that decrease the fluorescence intensity of light-emitting molecules. For this purpose, scientists often use dark quenchers, which absorb light across a relatively broad range of wavelengths and then release the energy as heat. The fluorophore and quencher must be in proximity, approximately 2 to 7 nm apart, to achieve high quenching efficiency.
How binding works
As discussed, aptamers primarily interact with their targets through hydrogen bonding. Hydrogen bonds are weaker and less stable than covalent or ionic bonds. An individual hydrogen bond has a lifetime on the order of picoseconds. However, large numbers of hydrogen bonds acting collectively are stronger and more stable than a single bond.
In a double-stranded DNA fragment, the spontaneous rupture of hydrogen bonds between the two strands temporarily opens the helix and exposes one or several nucleotides to the surrounding solvent. While the helix is open, the exposed nucleotides can bind to another available molecule. These fluctuations of the DNA helix occur at a timescale on the order of milliseconds. This phenomenon is referred to as DNA breathing, or fraying. In the case of an aptamer/antisense duplex, the antisense typically occupies the aptamer’s binding site. DNA breathing allows the target molecule to interact with the binding site, forming new hydrogen bonds. As the remaining hydrogen bonds between the aptamer and antisense break, the aptamer binds completely to the target molecule and the antisense is displaced.
The opposite can also occur: The new hydrogen bonds between the aptamer and its target break, allowing the antisense strand to rebind to the aptamer and displace the target molecule. However, since the aptamer’s affinity to the target is greater than its affinity to the antisense, it is more likely that the antisense will be displaced.
The binding of the antisense to the aptamer and the replacement of the antisense with the target molecule both involve hydrogen binding and thus quantum mechanical paradigms. The dynamic nature of the binding leads to an integrated strength of the binding as well as the manner in which these bindings are broken, and the target replaces the antisense. This frees the fluorophore to emit photons because the quenching action of the quencher conjugated to the antisense is no longer close enough to quench the fluorophore.
Photons are emitted by the chosen fluorophore at a rate of 400 million per second. A state-of-the-art detector will not have difficulty detecting these photons. Since the fluorophores are revealed in a liquid solvent, they will show the effects of Brownian motion, or the changes in the directions of particles due to collisions. Thus, over a second or more, the fluorophore will emit photons in all directions. By integrating the result from a static detector, or a large area or cell array, an accurate quantification of the total photons emitted can be calculated.
A single fluorophore will emit 2 × 109 photons every second. At 75% quantum efficiency, this is 1.5 × 109 photons per second. The emission λem = 655 nm. The energy of the photon is 3 × 10−19 J. Over one second, this is 4.5 × 10−19 J or 4.5 × 10−10 W. In an advanced CAST testing device, the emission of photons is omnidirectional, and a 20° solid cone collected by the optics and detector (out of 4π) translates to a sensitivity of one fluorophore in under a millisecond.
The detection process
If a cell array is used in detecting the emitted photons from the free fluorophores, image processing is required. Therefore, a photo detector cell array is framed as a camera. Several calculations are needed to establish the number of photons detected. These include the removal of subpixels not covering the emission photon wavelength and the quantification of static and dynamic dark current per cell.
When a large-area photon detector is used, the interference of the dark current is eliminated and no filters are used on the detector. This removes the image processing requirement. Nevertheless, other computing functions, such as integration, are still required to quantitate the photons emitted.
Test devices
Two test devices for detecting biomarkers by using aptamer molecular photonic beacons have been designed and tested. Both devices require the beacons, along with the sample, to be inserted in the test device for evaluation.
The first portable test device is cellphone-based and uses the cellphone LED as the exciting source and the CMOS camera as the emission detection source. This design uses integration, both spatial and temporal, to evaluate the fluorophore activity and determine whether the binding has occurred. Fourier transforms are used to extract the signal from the background optical noise. The cellphone performs all communication functions, captures the subject’s data, and stores the results in the cloud. The cellphone also mitigates human errors and reads the GS-1 data matrix on the vial. The sensitivity of the test device is 1 nW/mL.
For higher sensitivity (5 pW/mL), a second test device was developed that also uses a cellphone, but it operates through a Bluetooth connection. The device itself uses a large-area photon detection circuit and has a very high SNR.
To conduct a test, a 5-mL vial (Figure 6), containing 2 mL of the aptamer molecular photonic beacon in a phosphate buffered saline solution and 100 μL of the sample, is inserted into the test device (Figure 7). The type of aptamer molecular photonic beacon that is used may change from one test to the other.

Figure 6. A vial containing a COVID-19 aptamer molecular photonic beacon. Courtesy of Two-Photon Research.

Figure 7. A testing device that analyzes the sample that is bound with the aptamer. Courtesy of Two-Photon Research.
Through the cellphone, the device will request subject identification information and perform mitigation for errors, such as the lack or presence of a vial, the type of aptamer molecular photonic beacon in the vial, the expiration date of the vial, any light leak, and motion during the test. The results will be computed, combined with user and subject data, and stored in the cloud, and they will also be presented on the cellphone display. A dashboard that connects the user to the cloud is provided so that the user can access the data by single result or as part of a large demographic of information over time, geographical region, or class or subclass of subject by disease.
The system design can be enhanced by adding multiple special-purpose CMOS or single-photon detector arrays and LEDs. Mitigation of errors can also be accommodated in the software, alerting the user to the absence of contents, any light leak, thermal runaway, and motion in the fluid contained in the vial. Sensitivity can be increased by using multiple cell arrays, higher LED power, and optics.
Advantages in diagnostics
Every diagnostic test carried out on the platform is instantly recorded in the cloud. Access to the information can be controlled by using databases that authenticate ownership of the test devices. As such devices propagate in the world, informetric and pandemic demographics would be available instantly, identifying populations that are facing increased risk.
Moving diagnostics from the lab to the point of need — whether to the doctor’s office or the pharmacy, or to airport check-in counters or homes — offers the potential to immediately record and report a prevalence of viral threats along with the progress or inhibition of diseases. Armed with this information, authorities could rapidly tailor health policies to respond to evolving threats.
Aptamer molecular photonic beacons are currently in development to detect breast and lung cancers, and if the tests are successful, diagnostics could also become available for a host of illnesses that plague populations worldwide.
Meet the authors
Najeeb Khalid is CEO of Two-Photon Research; email: [email protected].
Ilias Hurley is the designer of the aptamer molecular photonic beacon. He is a biochemist at Two-Photon Research; email: [email protected].
Alexander Novikov is chief scientist, quantum engineer, and the designer of the CAST test device at Two-Photon Research; email: [email protected].
Acknowledgments
The authors would like to thank B.L. Hu of Two-Photon Research for providing the quantum calculations, A. Khalid of Two-Photon Research for providing software design and implementation, and professor Maureen McKeague of McGill University and professor Maxim Berezovski of the University of Ottawa for their advice and direction.
References
1. H.M. Berman et al. (2000). The protein data bank. Nucleic Acids Res, Vol. 28, pp. 235-242.
2. C. Varounis and A. Beshiri (2021). The future of cardiovascular disease prevention: a risk registry approach. Sci Am, www.scientificamerican.com/custom-media/nature-outlook-heart-health/the-future-of-cardiovascular-disease-prevention-a-risk-registry-approach.
3. C. Chai et al. (2011). SELEX (systematic evolution of ligands by exponential enrichment), as a powerful tool for deciphering the protein-DNA interaction space. Methods Mol Biol, Vol. 754, pp. 249-258, www.pubmed.ncbi.nlm.nih.gov/21720957.