Beyond the infrared, but just before you arrive at the microwaves, awaits an emergent area of opportunity
for biomedical research: the terahertz region. Spanning from 0.3 to 3 THz (equivalent
to 100 to 1000 µm), this wavelength range allows imaging and analysis of tissues
that otherwise are opaque to visible and infrared wavelengths, yet it doesn’t
have the risks associated with x-ray scanning.
Terahertz imaging is used for proteomics and general drug discovery
efforts, including defining the 3-D structures of proteins. It also is helpful for
viewing the myriad ways in which proteins fold into various configurations, which
affects their biophysical properties.
Spectroscopy within the terahertz range piles on the information.
It can reveal the different crystalline forms of molecules commonly used –
or under consideration – for pharmaceuticals. The same molecule of interest,
but with a variant form, may not be as stable; it may solubilize differently, or
it may not be absorbed by a patient in the same way as its counterpart. Terahertz
spectroscopy can help define these differences, which affect the overall mechanism
of action of a promising drug.
Terahertz imaging was first demonstrated in the mid-1990s, but
despite showing early promise didn’t expand in use quickly because there have
been a limited number of sources that could readily and consistently produce pulses
at terahertz frequencies. Using these wavelengths for spectroscopy has lagged even
more.
Primarily used in pharmaceutical research at first, the terahertz
range only recently has been investigated for medical imaging and spectroscopy.
At University College London, Caroline B. Reid and her colleagues are working with
TeraView Ltd. of Cambridge, UK, to explore potential medical applications for terahertz
radiation. Reid noted several potential benefits from using this wavelength range.
“Terahertz penetrates through many materials, has low scattering
compared to visible and near-IR wavelengths, is nonionizing and appears to provide
spectral signatures for anything from explosives to cancers,” she said.
Reid’s colleagues include Jan G. Laufer, Adam P. Gibson
and Jeremy C. Hebden of University College London, Emma Pickwell-MacPherson of the
Hong Kong University of Science and Technology in Clearwater Bay, and Vincent P.
Wallace of the University of Western Australia in Crawley. They published their
findings in the Aug. 21, 2010, issue of Physics in Medicine and Biology.
Reid’s group put to the test three terahertz reflection
spectroscopy techniques: linear spectral decomposition method (LSDM), spectrally
averaged dielectric coefficient method (SADCM) and Debye relaxation coefficient
method (DRCM). They tested each method using the same set of reflection data, which
was gathered using TeraView’s TPI Imaga1000 (imaging) and TPI Spectra1000
(spectrometry) instruments.

The accuracy and resolution of water and lipid concentrations as
measured via LSDM, SADCM and DRCM terahertz spectroscopy methods. Reused with permission
of Physics in Medicine and Biology.
Based on the Beers-Lambert law, LSDM provides a measurement of
the concentration of chromophores that react to the emitted terahertz radiation.
The concentration is estimated from the wavelength-dependent total attenuation that
occurs. The downside of the technique is that it requires previous knowledge of
the materials being scanned, especially their absorption coefficient spectra.
SADCM is a more empirical method that determines chromophore concentrations
by averaging the dielectric coefficients across a specific frequency range within
the terahertz band. Terahertz time-domain spectroscopy is well-suited for determining
the dielectric coefficients within a sample because it is a phase-sensitive technique.
However, Reid said, “SADCM can only be applied to a maximum of three solutes
[and] it returns poor resolutions, limiting its potential as a tool for tissue spectroscopy.”
Like SADCM, DRCM is an empirical technique. It measures the dielectric
relaxation among irradiated solutes within a sample, comparing them to measurements
previously taken from phantoms of various compositions. Reid said that DRCM could
be used to resolve the concentrations of more than two solutes in a sample, “provided
that material-specific variations in the Debye relaxation coefficients can be determined,
which are dependent on intramolecular interactions between the solutes.”
Reid and her colleagues tested samples comprising various concentrations
of lipids in water. Being a polar molecule, water is strongly absorbed – thus
easily distinguishable from other molecules – by terahertz radiation. They
found that the accuracy of the determined concentrations was strongly dependent
on which method was used. Nonetheless, all three proved capable, in principle, of
determining material composition through the concentration of the lipid solutes.
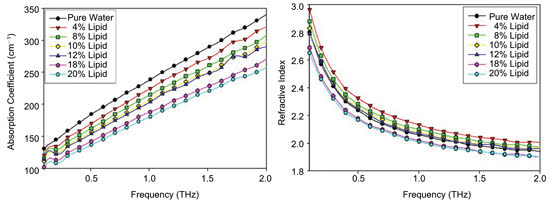
Shown are the absorption coefficients (left) and indices of refraction (right) garnered
from terahertz spectroscopy of various concentrations of lipids in water. Reused
with permission of Physics in Medicine and Biology.
The sensitivity of all three analytical methods is fixed, Reid
said. “In order to improve resolution,” she added, “only the standard
deviation of the measurement system may be improved, which may be achieved by increasing
the signal-to-noise ratio of the system.”
TeraView has developed new generations of its spectroscopy system,
which may further the method’s capabilities.
“Continued technological advances in the field which increase
the output power and signal-to-noise ratio of the systems,” Reid said, “may
enable smaller resolutions … and increase the potential of the technique
in tissue analysis.”
Seeking the cirrhotic liver
Imaging techniques such as MRI and ultrasound often can help locate
atypical tissue structures, but a biopsy may be required to diagnose the problematic
structure. Waiting for the results of a biopsy can be troubling for both patient
and physician alike; for example, biopsies are inherently invasive and leave patients
subject to complications of surgery.
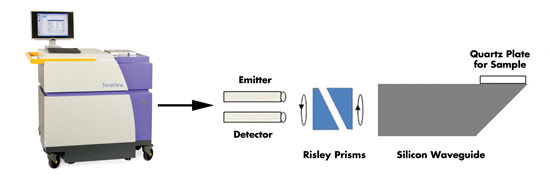
Disease such as liver cirrhosis can be determined using terahertz
spectroscopy setups coupled to imaging probes. Photo courtesy of TeraView Ltd.
One of Reid’s colleagues, Emma Pickwell-MacPherson, leads
her own terahertz research group at the Hong Kong University of Science and Technology.
Her team is interested in investigating the contrast mechanisms of various diseases,
including liver cirrhosis.
In liver cirrhosis, normal tissue is damaged and replaced by scars,
fibrosis and nodules, unhealthy features that adversely affect the liver’s
ability to function properly. As with many diseases, liver cirrhosis is best treated
as soon as possible, which requires early diagnosis. However, Pickwell-MacPherson’s
group reported in the Dec. 21, 2010, issue of Physics in Medicine and Biology that
current methods are not sensitive or specific enough to detect occult liver injury
at either early or even intermediary stages.
Using a TeraView TPI Imaga1000 combined with a handheld probe,
the researchers tested samples comprising healthy and cirrhotic rat liver tissue.
They measured reflections off both samples freshly excised from the animals and
from some of the same tissues after fixing them in formalin.
They found that both the fresh and the fixed cirrhotic samples
exhibited higher refractive indices and absorption coefficients than their healthy
counterparts. The formalin-fixed tissues, however, had lower absolute values for
both healthy and diseased tissues. Furthermore, they found that cirrhotic liver
tissue had a higher water content than healthy tissue.
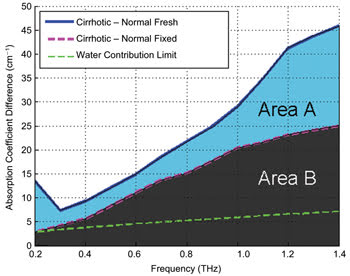
The blue line indicates the absorption coefficient
of freshly excised cirrhotic rat tissue, minus that of the fresh healthy tissue.
The pink line represents the same calculation for samples that were later fixed
in formalin. Thus, the area labeled “A” predominantly represents the
difference in absorption due to water, whereas area “B” represents the
difference due to structural changes, such as nodules and fibrosis.
They also discovered that structural changes in cirrhotic tissues
are picked up by the terahertz technique. The diseased rats “had evidence
of biliary cirrhosis and nodular liver with intense ductular proliferation, fibrotic
bridges and more pronounced inflammatory infiltration,” Pickwell-MacPherson
said. “These changes in the diseased tissue structure are the origin of the
structural differences that we are seeing.”
The investigators concluded that terahertz spectroscopy shows
promise for disease diagnosis, especially as a complement to other techniques.
“Terahertz imaging and spectroscopy could ultimately be
used intra-operatively to help surgeons determine the margins of [a] tumor to be
removed,” Pickwell-MacPherson said. “Perhaps, as the technology develops,
it will be possible to use a terahertz endoscope for diagnosis of diseases such
as colon cancer.”